17 Alpha Estradiol is probably the second best-performing longevity drug after rapamycin. We wish it was a supplement, but you can only buy it as drug or chemical right now. We’ve identified sources and pricing / cost below but its hard to get. Apollo ventures has announced that it is seeking to develop 17 alpha estradiol as a human drug which is great, but it will likely greatly increase the cost.
In the National Institute on Aging’s ITP life extension studies it was shown that:
- It Extends healthy lifespan (in mice) by up to 28%
- 17α-Estradiol seems to work just as well in late middle age as if you start it at an earlier age
- 17α-Estradiol only works in males for lifespan extension
- It resulted in their muscles getting stronger, improved glucose tolerance, and positive changes in muscular structure
While 17 alpha estradiol is not yet available as an FDA-approved drug, it is almost identical to the estradiol / estrogen that women take for hormone replacement therapy. But, 17 alpha estradiol is different in some very important aspects.
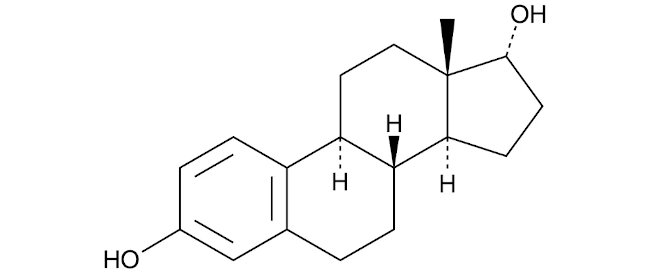
17α-estradiol is a naturally-occurring isomer of 17β-estradiol (commonly referred to as estrogen or oestrogen, the primary female sex hormone in women. In contrast to 17β-estradiol, 17α-estradiol is thought to be a non-feminizing estrogen - so if taken by a male, it will not cause the male to develop female-like physical characteristics. All human studies done to date have been completed safely, with no adverse events reported.
A small phase 1 clinical study of 17α-Estradiol (see full paper below) suggested it was safe in people, with no reported side effects. Moreover, it has been reported that the regular estrogen hormone pills that women take contain a significant amount (typically around 5%) of 17α-Estradiol (in addition to the 17β-estradiol that is the primary active ingredient) so it is a compound that has already been in use by millions of people for the past 20+ years.
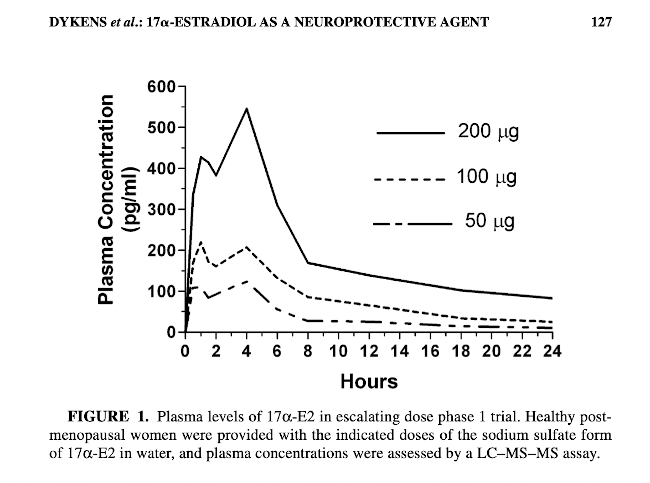
Also, the phase 1 clinical study noted:
In addition to the phase I trial described below, there are several published studies of the administration of 17α-E2 to humans by oral, sublingual, s.c., intravenous, or topical routes. These include studies with single oral doses ranging up to 70 mg, and a 12-week study at 2 mg p.o. per day. No adverse events have been reported. (See a list and links to all these earlier 17a-estradiol human studies at bottom of this post)
Note: One chemistry professor friend I spoke to regarding 17-alpha estradiol as an anti-aging compound suggested that the only concern he would have about it is that since 17-alpha and 17-beta estradiol are usually manufactured together (they are just chiral, i.e. right hand/left hand versions, of the same molecule), in any purchase of 17-alpha estradiol there will be some 17-beta estradiol, so the issue is how much there is of the beta version. 17-Beta estradiol is 10 fold more active than the 17-alpha estradiol. So, even with small percentages (e.g. 1 to 3% of the total purchase is 17-beta estradiol, you could see some feminizing effects from taking what is purchased as 97% to 99% “17-alpha estradiol”. So, I suspect that this would be the key side effect to watch for if taking this compound longer term. Also, the this also suggests that the greater purity of the 17-a estradiol sample, the lower the likelihood of excess 17-beta estradiol. The other key unknown with 17-alpha estradiol is the optimal dose for humans / males. Given the lack of side-effects and the wide range of dosing tried so far (albeit in a limited and short-term fashion) it seems like a reasonably strategy for biohackers might be to start low (at 50 μg/day), and slowly increase. The highest dose tested that I’ve seen is the 70mg dose.
17α-Estradiol is also sold in some countries for topical use for hair regrowth. Galderma has a product called Ell-Cranell that it sells around the world for his purpose. It doesn’t seem to be particularly effective in this application, similar to minoxidil.
17α-Estradiol in NIA ITP Program, Discussion with Richard Miller
The following information is sourced from the Peter Attia / Richard Miller podcast:
What was the genesis behind the plan to investigate 17α-Estradiol?
Richard Miller tells Peter Attia that a pharmacologist named James Simpkins has studied estrogen receptors and suggested investigating 17α-Estradiol. He had tried to make a compound that was like estrogen—17β-estradiol—which is generically crudely referred to as estrogen. Estrogens are widely used as medications, mainly in hormonal contraception, hormone replacement therapy, and to treat gender dysphoria in transgender women and other transfeminine individuals as part of feminizing hormone therapy.
James Simpkins synthesized 17α-Estradiol—very much the same compound except for one of the bonds tilts up instead of tilting down. And in his studies mostly in tissue culture cells, he found that yes, it was tenfold less active compared to 17β-estradiol (the compound which most people refer to as the estrogen used in hormone replacement therapy).
When he gave it to mice, he found it was non-feminizing: So when you give it to male mice, it did not induce the secondary sexual characteristics that the stronger estrogens, like 17 beta- estradiol, estrogen would do. His idea was that giving estrogen to male mice might make them live as long as female mice (female mice live 5-10% longer than males) AND this compound called 17α-Estradiol will be good for males because “no man wants to look feminine” even if he lives as long as a women.
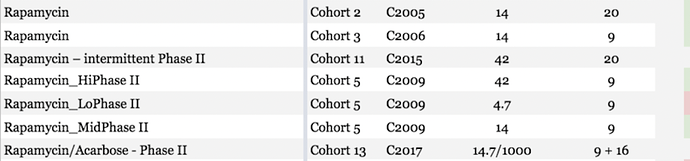
National Institute on Aging, ITP studies – 17α-Estradiol
The first attempt—cohort 5—used a low dose (4.8 ppm) and it extended median survival in males, but not maximum lifespan. In the next ITP study using 17-Alpha Estradiol —cohort 7—they gave a dose 3 times higher (14.4 parts per million) and it extended the measure of maximum longevity , as well as median longevity at all three sites for male mice, but not for females. In this study the males not only lived longer than regular old males, they lived longer than regular females. Dr. Rich Miller suggests that they have some clues as to why it works only in males, but they don’t know for sure. He said that their hope is that they can try to figure out a way to get around that obstacle and to get it to work in females as well. 17-Alpha Estradiol was also studied by the ITP in cohort 12 but the results have not yet been published.
Peter Attia asked if there an IND (Investigational New Drug application) for this compound?
Rich Miller suggested that there is a lot of attention being devoted to developing novel estrogenic compounds and “I’ll bet a great chunk of it is kept quiet by pharmaceutical firms”.
How to Buy / Purchase 17α-Estradiol:
17α-Estradiol is easily available from Lab / Reagent supply resellers at low cost (for people who work in biology / chemistry labs or academic institutions or companies). I hear people are being successful in acquiring this via friends who are academics where they can order these materials, or own smaller companies that can order chemicals like this). Please post your personal results with 17-alpha estradiol below in the comments. I see people are posting below about their acquisitions of this exciting new compound already…
CAS Number: 57-91-0
Cayman Chemical: 17α-Estradiol / CAS 57-91-0, $160/gram
Sigma Aldrich: 17α-Estradiol / CAS 57-91-0, $650 per gram
BioMol: 17α-Estradiol / CAS 57-91-0, €200 Euros/gram
The Most Interesting Research Studies on 17α-Estradiol
https://www.ncbi.nlm.nih.gov/labs/pmc/articles/PMC6052402/#!po=2.17391
National Institutes on Aging, ITP Life Extension Study on 17α-Estradiol
acel.12496.pdf (1.2 MB)
Development of 17α-Estradiol as a Neuroprotective Therapeutic Agent Rationale and Results from a Phase I Clinical Study
17AlphaEstradioldykens2005.pdf (801.1 KB)
Review of the Effects of 17-Alpha Estradiol in Humans: A Less Feminizing Estrogen With Neuroprotective Potential
17AlphaEstradiolmoos2009.pdf (213.3 KB)
Alzheimers Drug Discovery Foundation Report on 17α-Estradiol from 2021:
17α-Estradiol_UPDATE.pdf (415.6 KB)
Older Human Studies of 17-Alpha Estradiol
WILLIAMS, K.I. & D.S. LAYNE. 1967. Metabolism of 17-alpha-estradiol-6,7-3H by nonpregnant women. J. Clin. Endocrinol. Metab. 27: 159–164.
SCHROEDER, J., et al. 1997. Estradiol-17-alpha—a reasonable hormone replacement for the aging male? Maturitas 27(supplementary): 216.
HOBE, G., R. SCHON, N. GONCHAROV, et al. 2002. Some new aspects of 17alpha-estradiol metabolism in man. Steroids 67: 883–893.
BREUER H. & E. SCHOTT. 1966. Studies of the metabolism of 17 alpha-estradiol in man. J. Clin. Endocrinol. Metab. 26: 533–536.
LAURITZEN, C. 1969. Estrogenic activity of 17alpha-estradiol, equilin, equilenin, and their sulfoconjugates in experimental animals and in man. Symposion der Deutschen Gesellschaft fuer Endokrinologie 15: 142–144.
MEYER, W.J. 3rd, D.H. HENNEMAN, H.R. KEISER & F.C. BARTTER. 1976. 17 alpha estradiol: separation of estrogen effect on collagen from other clinical and biochemical effects in man. Res. Commun. Chem. Pathol. Pharmacol. 13:685–695.
[ORFANOS, C.E. & L. VOGELS. 1980. Local therapy of androgenetic alopecia with 17 alpha-estradiol. A controlled, randomized double-blind study (author’s translation)]. Dermatologica 161: 124–132.
OETTEL, M., M. DOREN, R. HELLER, et al. 1996. Estrogens and antioxidative capacity. Klimakterium und Hormonsubstituzion. Proceedings of Internationales Greifwalder Menopause Symposium, June 22-23: 109–118.
OETTEL, M. 1999. Estrogens and antiestrogens in the male. In Estrogens and Antiestrogens. II Pharmacology and Clinical Application of Estrogens and Antiestrogens. M. Oettel and E. Schillinger, Eds.: 505–571. Springer. Berlin.
Studies of Estradiol in Men - breuer1966.pdf (444.3 KB)
 .
.