Loyal for Dogs
Celine Halioua did a fairly basic / high-level presentation on her company “Loyal”. No new data was presented.
She is taking a very “consumer friendly” approach in marketing her company. Generally this seems like a good approach that, if the drug is effective, will help tie the benefits of longer lifespans for people’s pets, to the broader cause of improving lifespans and healthspans.
Some people might argue about whether life extension is a good idea for people and society in general, but it doesn’t seem like extending the lifespan of dogs is at all controversial. This would be a great benefit for the longevity community as a whole, so I do hope she’s successful.
Loyal has raised something in the neighborhood of $50 Million so far, and investors I’ve spoken to are bullish on its prospects going forward, so I’m sure we’ll see more investment rounds if the clinical / vet trials of their drugs are successful.
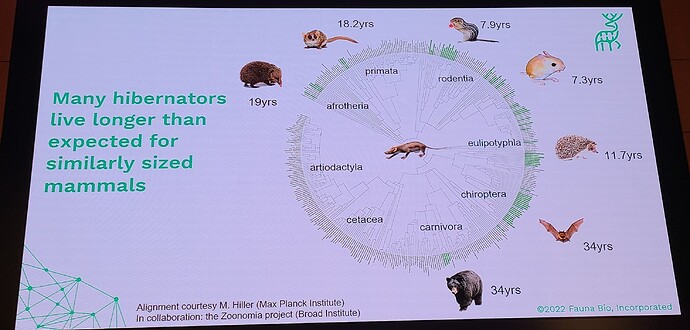
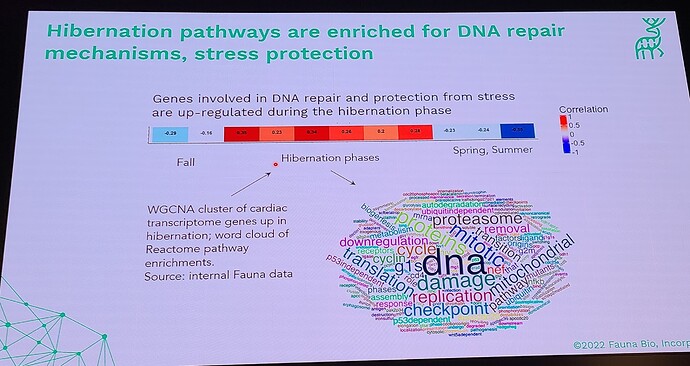
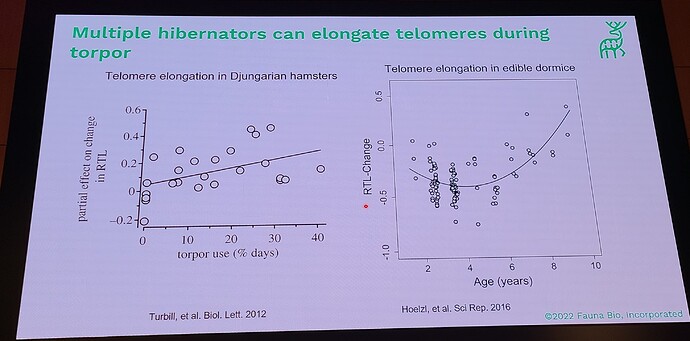
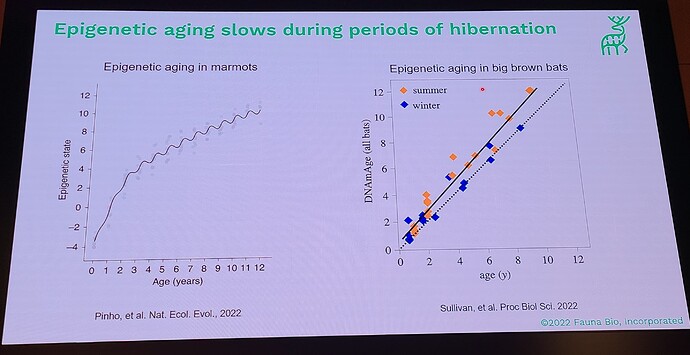
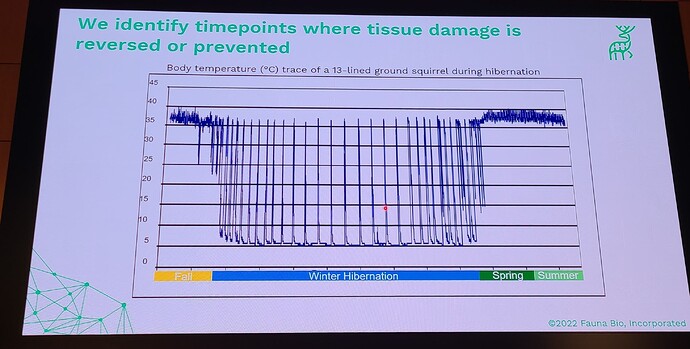
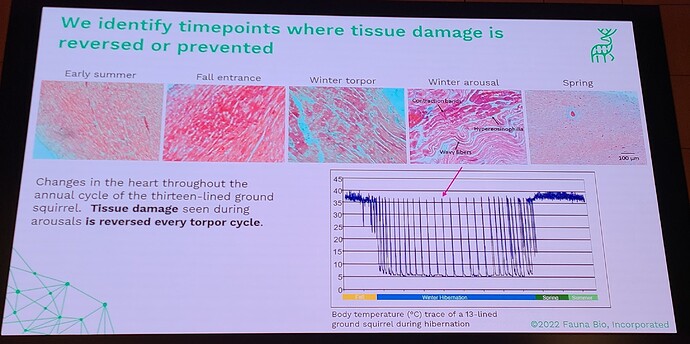
Coming out with a longevity drug for dogs is quite different than doing this for people, because unlike with people, you can actually do a longevity study with dogs (giving middle-aged, large dogs the drug and then watching them for a few years and gathering data on disease incidence in the dosed group, vs the control group). This means that the first FDA-authorized longevity drug will likely be for animals… in fact long before any drug will be FDA approved for humans for increased longevity. All the other longevity biotech companies that are only targeting human applications must first choose a disease indication to first demonstrate effectiveness, and only much later (after repeatedly showing that it reduces incidence of age-related diseases) would the FDA ever consider letting it claim that it slows aging (increases longevity). All of which means that we will likely see numerous FDA approved longevity drugs over the coming decade, and people will likely have a decade or two to get used to the idea of longevity drugs (being used in dogs), before they will see any drugs that claim longevity for humans.
Loy001 - first drug, targeted at the shorter lifespan of large dogs. This suggested to me that their first drug targeted the IGF-1 pathway, and she confirmed this when I spoke to her later in the day.
She said they use the “statins” metaphor a lot in their discussion of their drug. Preventative medicine is always better for the patient. Like statins, their drug will (it is hoped) reduce risk of future disease.
Celine says that they are very focused on this being a low side effect, low risk drug for the dogs.
First drug targeted to be released in 2024, to be sold through veterinarians from the sounds of it. At some point later they hope to take their drugs to human clinical trials, and FDA approval for people.
I’ve not dug deep into the research literature on IGF-1, but perhaps someone who has can comment on this question: Will a drug that inhibits IGF-1 only increase the lives of large dogs? Or, would it also work (but just to a lesser extent) in small and medium sized dogs? I’m assuming this would be the case, but it would also likely be a case of diminishing returns; the small dogs wouldn’t benefit much from having an even lower IGF-1 level?
I have to believe that many people (perhaps many people here) will be interested in trying this drug sooner rather than later. I told Celine that I expect lots of people will be getting prescriptions for their new anti-aging drug for their dogs, but then taking it themselves. She said she thinks that maybe the FDA is also expecting that… the FDA is asking her for human data on their drug even though they are only asking for dog FDA approval at this time.
Unlike most of the other longevity pharma companies like BioAge, etc Celine Halioua has been very quiet about the exact drug / molecule that they are using as their drug, and has not identified whether they in-licensed it from another company or not. I find this a little unusual, because I assume any patent applications would have been filed long ago.
There has been some research on longevity and lower IGF-1 levels in animals and humans: Lower Insulin-like Growth Factor-1 and Live Longer? | Natural Medicine Journal
And there are a number of drugs that seem to inhibit, or lower IGF-1 already on the market (used to treat people which excessively high IGF-1 levels, diagnosed as Acromegaly) , and I wonder if Loyal is just repurposing one of these drugs. Does anyone have thoughts on this?
MayoClinic suggests:
- Drugs that reduce growth hormone production (somatostatin analogues). In the body, a brain hormone called somatostatin works against (inhibits) GH production. The drugs octreotide (Sandostatin) and lanreotide (Somatuline Depot) are man-made (synthetic) versions of somatostatin. Taking one of these drugs signals the pituitary gland to produce less GH, and may even reduce the size of a pituitary tumor. Typically, these drugs are injected into the muscles of your buttocks (gluteal muscles) once a month by a health care professional.
- Drugs to lower hormone levels (dopamine agonists). The oral medications cabergoline and bromocriptine (Parlodel) may help lower levels of GH and IGF-1 in some people. These drugs may also help decrease tumor size. To treat acromegaly, these medications usually need to be taken at high doses, which can increase the risk of side effects. Common side effects of these drugs include nausea, vomiting, stuffy nose, tiredness, dizziness, sleep problems and mood changes.
- Drug to block the action of GH (growth hormone antagonist). The medication pegvisomant (Somavert) blocks the effect of GH on the body’s tissues. Pegvisomant may be particularly helpful for people who haven’t had good success with other treatments. Given as a daily injection, this medication can help lower IGF-1 levels and relieve symptoms, but it doesn’t lower GH levels or reduce tumor size.
Related: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6721736/