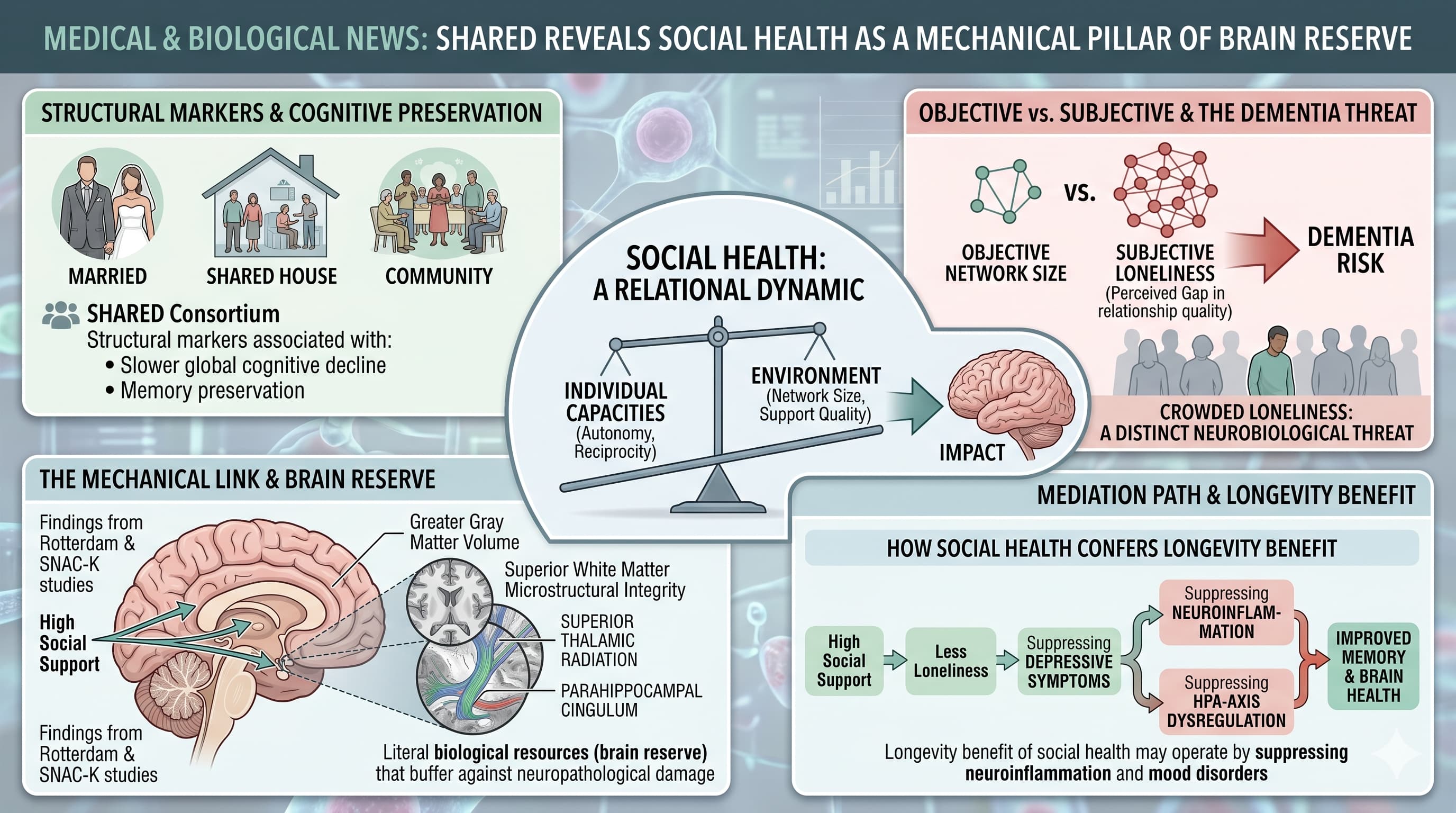
The biomedical model of dementia has long prioritized amyloid plaques and tau tangles, yet a massive global consortium study now suggests that “social health” may be just as vital for neuroprotection. The Social Health and Reserve in the Dementia Patient Journey (SHARED) consortium, analyzing data from over 150,000 participants across 40+ global cohorts, has finalized a multidimensional framework that positions social connection not as a “lifestyle perk,” but as a structural determinant of brain reserve.
The “Big Idea” is that social health is a relational dynamic between an individual’s capacities (autonomy, reciprocity) and their environment (network size, support quality). Unlike previous studies that focused narrowly on social isolation, SHARED reveals that structural markers—such as being married, living with others, and frequent community engagement—are consistently associated with slower global cognitive decline and memory preservation. Crucially, the research moves beyond correlation into the mechanical. Findings from the Rotterdam and SNAC-K studies link high social support to greater gray matter volume and superior white matter microstructural integrity, particularly in regions like the superior thalamic radiation and parahippocampal cingulum. These are the literal biological resources (brain reserve) that buffer against neuropathological damage.
However, the findings also highlight a critical nuance: the “subjective” often outweighs the “objective.” Loneliness—the perceived gap in relationship quality—was found to be a more potent predictor of dementia risk than mere social network size in several cohorts. This suggests that “crowded loneliness” is a distinct neurobiological threat. Furthermore, the consortium identified that depressive symptoms partially mediate the link between social support and memory, suggesting that the “longevity benefit” of social health may operate by suppressing neuroinflammation and HPA-axis dysregulation associated with mood disorders. For the longevity-focused professional, this paper marks a transition from viewing “socializing” as leisure to viewing it as a core protocol for maintaining the structural integrity of the aging brain.
Actionable Insights
To maximize cognitive health and longevity based on the SHARED findings, individuals should transition from passive social existence to active “Social Health” management:
-
Prioritize Structural Complexity: Actively expand your social network size and frequency of interactions. Being in a relationship, living with others, and weekly community engagement are verified predictors of slower cognitive decline.
-
Target High-Integrity Regions: Leverage “Face Processing” and “Theory of Mind” through complex social interactions. These activities are linked to higher white matter microstructural integrity in regions like the parahippocampal cingulum, essential for memory.
-
Audit for Reciprocity: Move beyond receiving support. The qualitative data identifies “reciprocity” (being helpful to others) and “maintaining dignity” as key unmeasured markers that support independence and brain resilience.
-
Mitigate Subjective Loneliness: Since loneliness predicts dementia risk independently of network size, prioritize relationship satisfaction and quality over quantity.
-
Mental Health as a Proxy: Monitor depressive symptoms. Because depression partially mediates the social-cognition link, managing mood is a mechanical necessity for preserving memory through social pathways.
Source:
- Open Access Paper: How does social health impact cognitive function and brain reserve? Findings from the SHARED Consortium
- Institutions: Centre for Healthy Brain Ageing (CHeBA), UNSW Sydney, Australia; Karolinska Institutet, Stockholm, Sweden; University College London, UK.
- Countries: Australia, Sweden, UK, Netherlands, Poland, Germany, Indonesia.
- Journal Name: Ageing Research Reviews, June 2026
- Impact Evaluation: The impact score of this journal is 13.1 (CiteScore 2024), therefore this is a High impact journal.