I have written my first ITP proposal (see below) for testing the cocktail Rapamycin, Acarbose and 17-alpha-estradiol. I sent it to Richard Miller to get some feedback and this was what I got back. Can we together as a community improve this proposal because it is an important step in the longevity field to take. Things go also very slow forward so every year that goes by we decrease our chances of living longer. So let’s do this as a community.
Richards feedback:
Some comments on our proposal.
I would not spend much time on general suggestions for ITP strategy on drug selection. If you do have such suggestions, you are welcome to make them (e-mail, for example), but they do not enhance your proposal, i.e. for justification of this three-agent mixture.
It does not help to invent new terminology. Rapa-plus-Aca is easy to understand. A new term like “longevity intervention cocktail v1” is not easy to understand. It’s jargon, and you should avoid jargon.
A proposal to test a specific mixture on male mice only will be rejected – we test all agents (and combinations) on both sexes.
The ITP has decided to test the combination of Rapa at 14 ppm plus 17aE2 at 14 ppm; decision was made yesterday.
The most important part of an application to test three agents is the part where you provide a justification for testing them in combination – your section on “why test this type of cocktail?” In fact, you don’t want to address “this type of cocktail,” but want to justify this specific combination of agents. Your section on this short and fairly superficial. You say “it would be interesting to see if they may have good complementary effects” and that’s true, but you provide no supportive arguments – why this specific set of drugs? What is the evidence for possible complementarity? Will side effects of the combination (like weight loss, for example) be more severe?
The second element in your justification – advice for people who already take all three agents – is not useful. We have no idea if benefits, or harms, seen in mice will also be seen in people. And in any case such a goal is not the focus of the ITP.
Hope these comments help you as you revise your proposal.
ITP PROPOSAL: RAPAMYCIN + ACARBOSE + 17-ALPHA-ESTRADIOL
Background
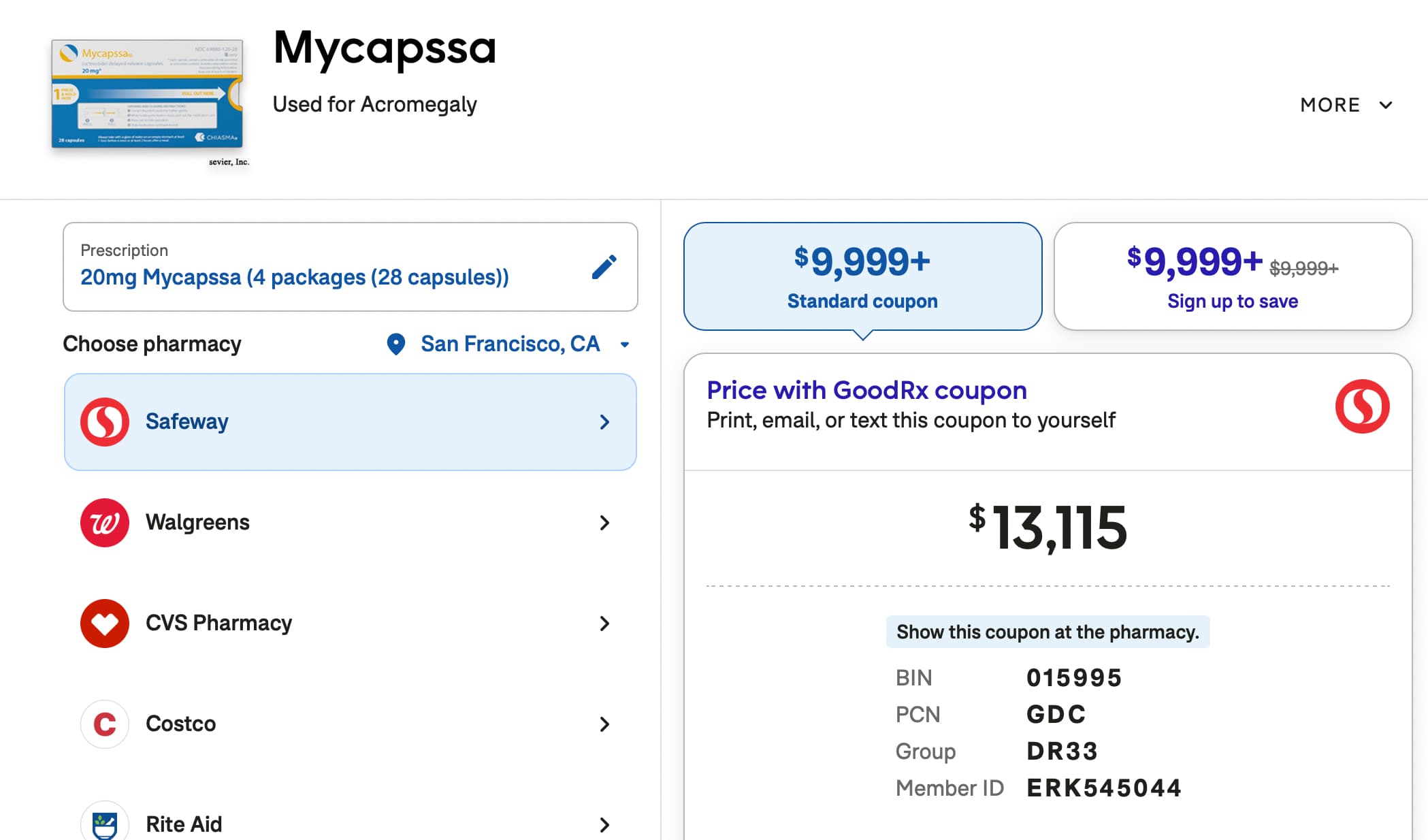
The ITP has done more than 150 intervention tests throughout the years and the best lifespan effect for both genders has been seen when Rapamycin and Acarbose was combined. In this proposal I will call that intervention for the “Longevity Intervention Cocktail v1.0 young-aged edition”.
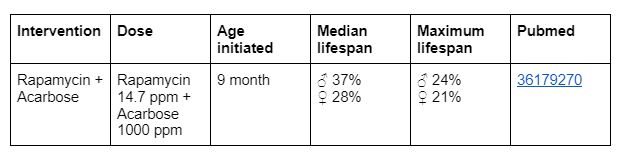
There is something unique with the discovery of the “Longevity intervention cocktail v1.0” because it seems not to be gender-specific like for example the compound 17-a-estradiol. Therefore one interesting thing to do is to further develop this cocktail and see if it’s possible to improve the lifespan effects even more. A suggestion is to dedicate every year at least one intervention for this purpose. Which specific intervention or dose regime which is tested may differ between genders. This is because the more advanced longevity intervention cocktails most likely are gender-specific.
To get the most valuable data from the ITP investment which is around 450k dollars per intervention for both genders then a suggestion is to skip the middle step test where Rapamycin is tested with another compound. Like the test which is done in cohort 2023 where Rapamycin and 17-a-estradiol. This is of course very interesting data to get but most likely it will not lead to any huge lifespan effects which outperform the “Longevity intervention cocktail v1.0”. For that reason a suggestion is instead to directly test to add a compound to the cocktail and see if the ITP lifespan record can be beaten or not. This way the longevity needle can be moved much faster and in a cost effective way. It will half the ITP investment. So instead of 900k (= 450k for Rapamycin and 17-a-estradiol + 450k for Rapamycin, Acarbose and 17-a-estradiol) it will land on 450k (= 450k for Rapamycin, Acarbose and 17-a-estradiol). If the ITP wants in the future to get that middle step data point then such an investment can be done later on but it should not be a mandatory first step to take. We should instead aim first for the cocktail improvement.
One last suggestion to decrease lead time in ITP even more is to do the cocktail tests on middle aged mice between 16 - 20 months old. The ITP did a successful test on such mice and that type of cocktail I will call in this proposal for the “Longevity Intervention Cocktail v1.0 middle-aged edition”.

Suggested treatment protocol
This proposal is about improving the male version of the cocktail. So the proposal is to add the compound 17-a-estradiol to the “Longevity intervention cocktail v1.0 middle-aged edition”. This compound has shown really good lifespan effects on male mice in the ITP but not so big ones on females.
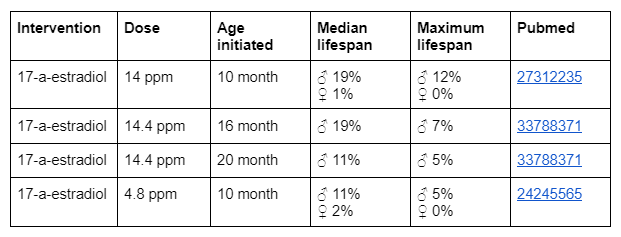
In the cohort 2023 there exists already a cocktail test on Rapamycin and 17-a-estradiol. Therefore the proposal for the next new cocktail is to use the same dose regime of 17-a-estradiol as in the cohort 2023 and to start the treatment at the 16 month age. So the proposed longevity intervention cocktail is:
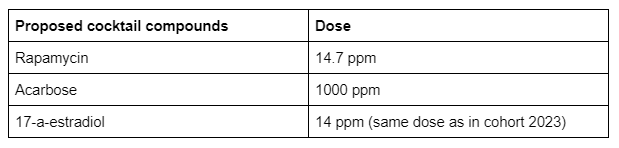
Why test this type of cocktail?
-
Complementary mechanism of action
The three compounds may have complementary mechanisms of action. Rapamycin is a mTOR inhibitor. Acarbose is a glucose regulator. The mechanism of action around 17-a-estradiol is not yet fully understood but seems to have a more broad effect than the two former ones. There exists overlaps between the compounds but it would be interesting to see if they may have good complementary effects. -
Test the “best of the best” approach
There are some people in the longevity community who take Rapamycin, Acarbose and 17-a-estradiol. For that reason there is a value to see if stacking the “best of the best” really leads to the best effects or if things are a bit more complicated than that approach.
Probably this cocktail will not lead to detrimental lifespan effects in mice or lower lifespan than control but it may lead to lower lifespan effects than the “longevity intervention cocktail v1”. So even if the experiment shows this it would be very valuable and educational data for the general public. It will highlight the complexity of that it’s not that simple to just take the best interventions and combine them. It will show that more of something good is not always better. This is also something the researcher Brian Kennedy has tried to point out throughout the years. In a podcast interview he raised for example this concern: "If you hit the same thing from too many different pathways you’re going to go from good to bad. I think that’s a concern right now.” So it would be good to get more data on the topic to see if this concern is valid or not and how strong it is.
Safety information
All three compounds have been tested in the ITP before and therefore the safety data is quite well known when it comes to single compound interventions. There exists also ITP safety data on the combination of Rapamycin and Acarbose. In cohort 2023 when Rapamycin and 17-a-estradiol was tested then too big weight loss was seen according to Richard Miller. This was one reason why the dose of 17-a-estradiol was lowered and this changed dose is also the one that is proposed to be used in this new suggested cocktail with Rapamycin, Acarbose and 17-a-estradiol.
References
Strong R, Miller RA, Cheng CJ, Nelson JF, Gelfond J, Allani SK, et al. Lifespan benefits for the combination of rapamycin plus acarbose and for captopril in genetically heterogeneous mice. Aging Cell. 2022. Lifespan benefits for the combination of rapamycin plus acarbose and for captopril in genetically heterogeneous mice - PubMed
Strong R, Miller RA, Antebi A, Astle CM, Bogue M, Denzel MS, et al. Longer lifespan in male mice treated with a weakly estrogenic agonist, an antioxidant, an α-glucosidase inhibitor or a Nrf2-inducer. Aging Cell. 2016. Longer lifespan in male mice treated with a weakly estrogenic agonist, an antioxidant, an α-glucosidase inhibitor or a Nrf2-inducer - PubMed
Harrison DE, Strong R, Reifsnyder P, Kumar N, Fernandez E, Flurkey K, et al. 17-a-estradiol late in life extends lifespan in aging UM-HET3 male mice; four other drugs do not affect lifespan in either sex. Aging Cell. 2021. 17-a-estradiol late in life extends lifespan in aging UM-HET3 male mice; nicotinamide riboside and three other drugs do not affect lifespan in either sex - PubMed
Harrison DE, Strong R., Allison DB, Ames BN, Astle CM, Atamna H, et al. Acarbose, 17-α-estradiol, and nordihydroguaiaretic acid extend mouse lifespan preferentially in males. Aging Cell. 2014. Acarbose, 17-α-estradiol, and nordihydroguaiaretic acid extend mouse lifespan preferentially in males - PubMed