@MAC - All well thought out. I knew you had your reasons and that they were probably backed up by data. Many physicians are starting to use TRT in SQ dosing and getting great responses much of which you have already covered, but trying to avoid the peaks and troughs that may be more valuable in Rapa longevity dosing to avoid immunosuppression. Keep us posted and good luck!
Please define “great responses” re TRT/SQ?
Sure!
Compliance of SQ over IM. Not everyone is up or an IM thigh shot.
Slower and more steady response due to a more even release into the blood stream IM injections tend to get in much faster due to higher vascularity, as you pointed out. Many are dosing twice a week, but some are even dosing daily with small doses.
Less chance of infection causing serious adverse events due to the muscle not being pierced over and over. If an infection occurs sub Q, it will tend to open the skin and not create an issue deep into the muscle.
Overall, most find a very similar response ( some better and some not) with SQ dosing, but with small needles and more likely to self inject vs. have a spouse of other do it for them. Its just alternative option that some people have not been exposed to.
I can see that benefit, especially as I’m currently doing 2 TRT injections + 5 IM rapa injections PER WEEK.
My next iteration is to make an even more super concentrated rapamycin/DSMO liquid, say 60 mg/mL, so I can reduce dose volume/# injections, still deliver mg dose, and thus reduce penetrations.
Mac,
How are you delivering the
intranasal (IN) solution?
A device or?
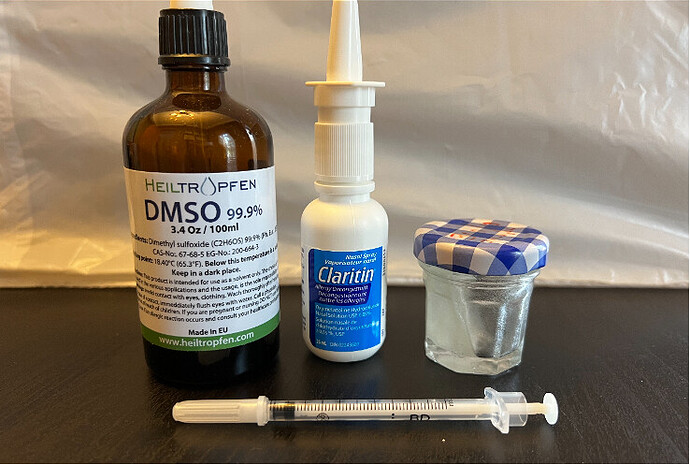
Page 48 of my study. Inside the nasal sprayer is a 30 gm/mL solution of rapamycin in 100% DSMO. You can of course make different concentrations for different volume/dose delivery. You cannot use just “any” nasal spray device…anything that touches DSMO should be INERT to the solvating capability of DSMO. The main storage compartment can be glass. Check for the recycle number under the plastic container and/or ensure the materials used in the mechanical delivery.
Sterilize everything prior to any filling. Keep in freezer. Thaw out prior to use (DSMO solidifies at 19C/65F).
DMSO compatibility with plastics and what kind of jars, sprayers to use
Can DMSO be stored in plastic? Well, yes, but not all plastic types are compatible with DMSO. It is okay to use the below-listed types of plastic.
Dimethyl Sulfoxide is non-reactive to these types of plastic:
- PETE (1)
- HDPE (2)
- LDPE (4)
- PP (5)
- Nylon
- PTFE
These containers are safe to use with dimethyl sulfoxide:
- Glass containers
- Glass jars
- Stainless steel
It seems we should get all the issues and unknowns around dosing approaches, tissue specificity, etc. into this new online database being developed:
A comprehensive online compilation of open problems in longevity science
Professor Joao Pedro Magalhaes (University of Birmingham, UK)
In this project, Professor Pedro Magalhaes & team will create and populate an online framework of open problems in longevity and aging research. He will engage and collaborate with the scientific community to develop this project.
Another big thanks to MAC for this update. His experiment into Rapa dosing protocols is truly on the cutting edge and I hope everyone realizes it. We may never find another individual who is willing to do this and report back in such detail on side-effects, labs, etc. He is destroying many of the myths about Rapa side-effects especially when Rapa is delivered IM and IN. One thing is for sure, Cancer will have a difficult time establishing a presence in his body. Of course, there are many who have health issues and they would be advised to go very slowly into this new frontier.
That’s very true. I wonder if a compounding pharmacy could save us alot of trouble and just prepare an injectable solution of rapamycin.
Maybe 10 mg’s rapa into 1 ml of solution.
I bought some Pep-C on a lark. When I realized it needed to be injected I bought all of the accoutrements for that. I believe it will never leave my freezer.
Do these repeated freeze-and-thaw-cycles of the Rapa-DMSO-solution not degrade the Rapamycin?
@MAC You’ve gone dark on us… hope all is well. Please provide update when you have a chance.
I’ve shared your injection protocol with many researchers (animal researchers with a background in rapamycin studies) and have gotten surprisingly little feedback so far. Part of the issue is that human clinical (and non-clinical) studies are pretty distant from the experience of mouse studies I suspect.
I still think that this pathway of IM and IN delivery for rapamycin may prove to be the optimal long term to get the higher doses we want while minimizing side effects, but we need to get the medical professionals interested in testing it further. Perhaps this is something that Healthspan might support or AgelessRX. I noticed today that Healthspan is getting into the business of microneedle delivery of rapamycin for hair growth (see this quote from another forum I visit:
My name is Daniel Tawfik and I am the founder of Healthspan. We prescribe rapamycin as an online telemedicine service. We will have a topical Rapamycin + EGCG solution delivered through microneedles product starting in October.
Living the man rodent dream!
Massive sirolimus levels, no symptoms, minor biomarkers dysregulation, and smaller and softer poops.
Still on same IM/IN weekly dosing.
Seriously, all is good. Nothing untoward re how I feel. No impact on DAILY intense workout regiment or recovery. Can do HIIT no issues. So much for rapamcyin and mTOR inhibition and exercise blunting. Smashed that theory. Half marathon coming up.
Since I am OMAD, hunger signal starts and continuous all day until dinner. Rapamcyin seems to have upped this signal noticeably. I am feeling a “tightening” to my body, a slow weight loss and leanness feeling. Weight loss modest so far but trending downwards.
The smaller, softer poops is helpful as being keto can be a bit constapiationagenic. My constipation symptoms are gone. Sleep seems a bit perturbed…no issues falling asleep, but waking a bit earlier (per my smart watch tracking). This is a common side effect, but not impacting my daily routine.
I am busy writing a paper for submission to an Aging journal of my case study. Hoping my outlier intervention will get some researcher group to take me on as a lab rat…that’s my objective. They can scan and tissue assay all they want. No euthanasia though😉
I have another rx for labs in couple months, will update any transiency either way.
I’ve got the 0.5mL injections down from 5 x 0.1mL to 3 equal injections. Hoping to get it down to two injections of 0.25mL. Pushing the dose volume can produce intense stinging. Recall early days massive stinging and post injection pain from the injections, which has now completely abated.
Just think, I am taking perhaps 20x+ what most people on this site are dosing.
Great to hear things are going well, and Fantastic news on the paper. Submit it to Blagosklonny and I bet he’d work with you to get it into his journal.
From your descriptions - this sounds like the best method so far to get the systemic levels of rapamycin high enough for significant lifespan / heathspan effects, without the side effects. Please do blood testing sometimes to validate the key blood metrics are also good.
He has not replied at all. Maybe my slight dissing him in my case study intro, he didn’t take it too well. ![]() Actually none of the mTOR gods has replied, except MK who said “very interesting study”.
Actually none of the mTOR gods has replied, except MK who said “very interesting study”.
I don’t know about longevity, but from a dose/side effect, apparently yes.
Yes will update next labs, with much deeper immuno markers. Fascinating to see where I am trending.
As you know , I think your experiment is great, but some of the high dose rapamycin studies reported neutropenia and thrombocytopenia.
I’d approach this like we do with clozapine. Check your CBC weekly for maybe the first 6 months or so and then monthly. If your neutrophils or platelets really drop then you need to quickly react and notify your PCP.
All of the other labs wouldn’t be particularly urgent.
Duly noted, that’s watch plan. I am on a simple therapeutic dose of Rapamycin, but without the side effects. Will dial back when key markers go south.
Mac’s report is a fascinating collection of information - I hope you keep us informed how you are doing.
Having said that: it may be difficult for most people using Sirolimus off-label to get access to weekly blood tests. I do wonder how big of a risk there is when dosing so high. I know of another person (not a member) that for a while took extremely high doses of intranasal Sirolimus. And I can imagine people may read here (also people that don’t necessarily post here) and ‘try this out at home’.
Rivas mentioned some important points when it comes to thrombocytopenia and leukopenia, that imho deserve more attention. Could there be a risk to end up with internal bleedings, for example? How much of a risk would there be to end up with an invasive infection?
Weekly blood tests do seem very important as Rivas suggested. But again, I think most people don’t have access to that. This short study suggests that most cases of sirolimus-induced thrombocytopenia and leukopenia in transplant patients resolved on its own - but then again they may have lower Cmax levels of Sirolimus.
"Sirolimus-induced thrombocytopenia and leukopenia in renal transplant recipients: risk factors, incidence, progression, and management
Abstract
Background: Our study assessed the factors that predispose renal transplant recipients to the occurrence of thrombocytopenia and leukopenia, as well as the severity and the time- and concentration-dependence of these side-effects, after administration of sirolimus (SRL) in combination with a cyclosporine (CsA) and prednisone (Pred) regimen.
Methods: The clinical courses of two cohorts of renal transplant recipients were compared over 1 year: 119 patients received SRL in addition to CsA and Pred, and 65 demographically similar, concurrent patients received only CsA and Pred. Using an analysis of variance, pretransplant laboratory values and SRL trough concentrations (C0) were correlated with the occurrence, severity, and persistence of drug-induced thrombocytopenia (platelet count <150x10(3) cell/mm3) and/or leukopenia (white blood cell count <5,000/mm3).
Results: Neither the ethnic background nor the pretransplant cytomegalovirus serological status was associated with the occurrence of hematological complications. Thrombocytopenia was usually observed during the first 4 weeks of treatment (P=0.004). The occurrence, but not the severity or the persistence, of both thrombocytopenia and leukopenia correlated significantly with SRL trough concentrations > or =16 ng/ml (P=0.001 and 0.0001, respectively). A significant correlation is evident between the occurrence of the two adverse effects (P=0.001). In 89% of patients, the first episode of either type of cytopenia resolved spontaneously. Among the remaining 11%, 7% responded to SRL dose reduction, and 4% to temporary suspension. No patient required permanent cessation of SRL therapy. Most patients experienced repeated, but self-limited, episodes of toxicity.
Conclusion: Thrombocytopenia and leukopenia are not infrequent occurrences with SRL treatment, and they generally resolve spontaneously."
Thanks for digging that. Actually makes me feel better about it since his trough was 11 and didn’t hit that critical 16 number.
Also good to know that it’s self limited regardless.
@MAC Have you seen this patent yet? Its the Wyeth patent for injectable rapamycin / sirolimus:
of course, this is IV and not IM administration. I don’t know enough about this area to know if you can use an IV solution for IM administration - I’m sure you and others will chime in and correct me if I’m way off base here.
I would still love for more human clinical research to be done on IV, IM, IN and transdermal patch-based rapamycin delivery approaches…
The preferred method of injection is by intravenous injection.
The injectable rapamycin composition of this invention provides a therapeutically effective amount of rapamycin in the blood of a mammal and also provides a therapeutically effective amount of rapamycin in the brain, liver, kidney, lung and spleen of a mammal.
The invention also provides a method of making the rapamycin-containing nonionic surfactant injectable compositions. These injectable compositions of rapamycin are prepared by (a) dissolving rapamycin in an organic solvent, which is capable of dissolving rapamycin and is miscible with the nonionic surfactant, (b) adding the nonionic surfactant, (c) if required, removing the organic solvent, and (d) adding water or an aqueous solution containing pharmaceutically acceptable excipients commonly used.in pharmaceutical formulations.
The injectable rapamycin composition comprises the following three main components: rapamycin, a nonionic surfactant and water.
The therapeutically effective amount of rapamycin in the injectable composition is usually in the range of about 1.0 to about 1.0 to about 20 mg/ml of solution. This range represents a range of about 0.1 to about to about 2.0 per cent by weight of rapamycin in the injectable composition.