Duly noted, that’s watch plan. I am on a simple therapeutic dose of Rapamycin, but without the side effects. Will dial back when key markers go south.
Mac’s report is a fascinating collection of information - I hope you keep us informed how you are doing.
Having said that: it may be difficult for most people using Sirolimus off-label to get access to weekly blood tests. I do wonder how big of a risk there is when dosing so high. I know of another person (not a member) that for a while took extremely high doses of intranasal Sirolimus. And I can imagine people may read here (also people that don’t necessarily post here) and ‘try this out at home’.
Rivas mentioned some important points when it comes to thrombocytopenia and leukopenia, that imho deserve more attention. Could there be a risk to end up with internal bleedings, for example? How much of a risk would there be to end up with an invasive infection?
Weekly blood tests do seem very important as Rivas suggested. But again, I think most people don’t have access to that. This short study suggests that most cases of sirolimus-induced thrombocytopenia and leukopenia in transplant patients resolved on its own - but then again they may have lower Cmax levels of Sirolimus.
"Sirolimus-induced thrombocytopenia and leukopenia in renal transplant recipients: risk factors, incidence, progression, and management
Abstract
Background: Our study assessed the factors that predispose renal transplant recipients to the occurrence of thrombocytopenia and leukopenia, as well as the severity and the time- and concentration-dependence of these side-effects, after administration of sirolimus (SRL) in combination with a cyclosporine (CsA) and prednisone (Pred) regimen.
Methods: The clinical courses of two cohorts of renal transplant recipients were compared over 1 year: 119 patients received SRL in addition to CsA and Pred, and 65 demographically similar, concurrent patients received only CsA and Pred. Using an analysis of variance, pretransplant laboratory values and SRL trough concentrations (C0) were correlated with the occurrence, severity, and persistence of drug-induced thrombocytopenia (platelet count <150x10(3) cell/mm3) and/or leukopenia (white blood cell count <5,000/mm3).
Results: Neither the ethnic background nor the pretransplant cytomegalovirus serological status was associated with the occurrence of hematological complications. Thrombocytopenia was usually observed during the first 4 weeks of treatment (P=0.004). The occurrence, but not the severity or the persistence, of both thrombocytopenia and leukopenia correlated significantly with SRL trough concentrations > or =16 ng/ml (P=0.001 and 0.0001, respectively). A significant correlation is evident between the occurrence of the two adverse effects (P=0.001). In 89% of patients, the first episode of either type of cytopenia resolved spontaneously. Among the remaining 11%, 7% responded to SRL dose reduction, and 4% to temporary suspension. No patient required permanent cessation of SRL therapy. Most patients experienced repeated, but self-limited, episodes of toxicity.
Conclusion: Thrombocytopenia and leukopenia are not infrequent occurrences with SRL treatment, and they generally resolve spontaneously."
Thanks for digging that. Actually makes me feel better about it since his trough was 11 and didn’t hit that critical 16 number.
Also good to know that it’s self limited regardless.
@MAC Have you seen this patent yet? Its the Wyeth patent for injectable rapamycin / sirolimus:
of course, this is IV and not IM administration. I don’t know enough about this area to know if you can use an IV solution for IM administration - I’m sure you and others will chime in and correct me if I’m way off base here.
I would still love for more human clinical research to be done on IV, IM, IN and transdermal patch-based rapamycin delivery approaches…
The preferred method of injection is by intravenous injection.
The injectable rapamycin composition of this invention provides a therapeutically effective amount of rapamycin in the blood of a mammal and also provides a therapeutically effective amount of rapamycin in the brain, liver, kidney, lung and spleen of a mammal.
The invention also provides a method of making the rapamycin-containing nonionic surfactant injectable compositions. These injectable compositions of rapamycin are prepared by (a) dissolving rapamycin in an organic solvent, which is capable of dissolving rapamycin and is miscible with the nonionic surfactant, (b) adding the nonionic surfactant, (c) if required, removing the organic solvent, and (d) adding water or an aqueous solution containing pharmaceutically acceptable excipients commonly used.in pharmaceutical formulations.
The injectable rapamycin composition comprises the following three main components: rapamycin, a nonionic surfactant and water.
The therapeutically effective amount of rapamycin in the injectable composition is usually in the range of about 1.0 to about 1.0 to about 20 mg/ml of solution. This range represents a range of about 0.1 to about to about 2.0 per cent by weight of rapamycin in the injectable composition.
I really do think you may be on to something here MAC! Honestly, I prefer injections to IV. If injections can beat oral in terms of efficacy and side-effects, I think many people would adopt it. I wish you the best of luck for a great success!
Had a brief once read through. Not an easily replicable retail hacker formulation. Conceptually you could give IV as IM but there are some differences. I’ve mostly seen IV formulations parenteral delivery, but it could be redeployed for IM with different dosing.
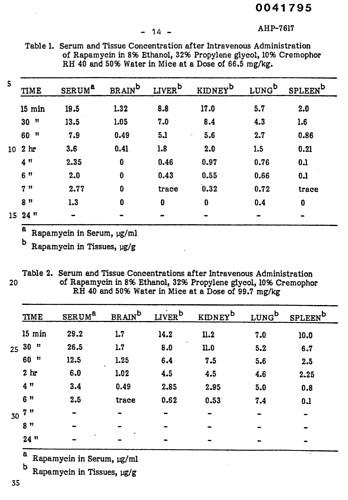
Some very interesting select organ tissue rapamcyin concentrations, shown in table below. Reminder these are for SINGLE dose administrations:
-
These are a VERY high human equivalent doses (not surprising as IM or IV or IP), and even so, not much partitions into the brain, with very rapid dropoff to non measurable after 4-5 hrs. We clearly see the metabolic differences in metabolism vs human.
-
It would take vey high and continuous AUC to get constant Rapamycin bathing into the brain vs a single dose, especially oral.
-
If MB has tweeted or commented that he upped his weekly/intermittent dose to a 20 mg single ORAL dose route to get Rapamycin into the brain, he is sadly mistaken. If he’s taking a chronic daily oral dosing, then depending on the trough level, there might be some passage into the brain. There has only been one paper that I have come across that showed human brain tissue Rapamycin partitioning, but only with 2 or 5 mg daily dosing. So that may be his 2mg/day rationale.
-
This is yet another stark demonstration of why I have added IN to my protocol, to add to my brain uptake WITHOUT adding to even higher current systemic levels delivered via the parallel IM. At my therapeutic level of Rapamycin dosing, there is certainly Rapamycin crossing the BBB. My trough of 11 ng/L and high AUC is equivalent to transplant daily oral dosing, similar to thousands + patients over many decades. The IN is a more targeted and brain specific route of delivery, which is additionally to IM vs oral delivery, is the real uniqueness of this paradigm.
Hopefully I could get a radionuclide tagged Rapamycin PET scan of my dosing protocol to track tissue concentrations across all my organs and brain. ![]()
I was able to get the 0.5mL IM dose split into just two x 0.25mL doses!
This is yet another practical milestone vs original 5 x 0.1mL doses at beginning of protocol. Recall 1st doses, I literally was in so much pain in my legs, it significantly impacted my gait for week+.
There was still a brief bout of intense stinging when 1st liquid entered tissue, but with slow injection was able to push through to 0.25mL without much additional stinging. Post injection, just a mild soreness (10 minutes later at time of writing), but don’t expect any lingering effects re pain/mobility. It seems my tissue is fully sensitized now to the base level of Rapamycin, and “hormesis” dynamic in play.
Have you noticed any cognitive changes? I recall preventing dementia is a high priority for you? I’m also interested in whether this mode of administration might benefit neuroplastic pain…
Nothing scientific, hard to say. I felt amazing going into this, not feeling any “wow” improvements, and perhaps more importantly, no negative symptoms.
I have another online cognitive test opportunity coming up, will let you know if any significant signal outcome.
Absolutely zero post injection pain or symptoms rest of day.
From what I understood the DMSO results in stinging rather than the Rapa. So I’m surprised you say the stinging is getting less after repeated injections.
I did a blank DSMO vehicle injection…zero symptoms. It’s the Rapmycin.
The “less” from repeated injections is the existing steady state Rapamycin tissue concentration blunting the new bolus.
Your experience may differ. In several forums that I frequent, members inject/use intranasal DMSO as a carrier commonly and the comment that DMSO stings, is almost without exception expressed.
Not all, I ran the n=1 blank pure DSMO experiment, no pain. The 1st doses were extensive massive pain! Current doses, NO pain. How is that DSMO?
Well as I said in my previous post: your experience may differ. Reactions to DMSO are not uncommon though: Adverse reactions of dimethyl sulfoxide in humans: a systematic review - PMC
Amazingly well. Zero symptoms, no impact normal routine, no change dosing. I did skip my last blood donation just out of an abundance of caution as my last iron panel (clinically iron deficient anemia) was screaming danger, lol, although zero symptoms. Will post next labs.
3rd week of just two x 0.25mL injections. Pretty strong stinging at injection, but very rapid dissipation and NO lingering effects.
The gastro effects are indeed real and persistent…sure sign of the metabolic effects of high dose Rapamycin.
Good to see new members joining.
Amazing site, kudos. I would not have ventured into these waters without the support and knowledge exchanges of this forum.
I’ve been upping my daily running to 10km/day, I have a half marathon in 2 weeks. Still doing daily resistance exercise + endurance running.
Excellent bud!
Like your rational for injecting RAPA, and following your journey.
All good here too! Ageless
@MAC , just checking in. Still dosing at the same level? Still going well? Hope you are working on the paper to get published - would love to see this approach get broader attention and testing.
All good! Still on same exact weekly dosing protocol, with full adaptation to two injections of 0.25mL, one per thigh. The post injection stinging seems to be lessening as time passes. Certainly, not bothersome at all post injection (dissipation within minutes). The IN dosing still feels the same, slight stinging at time of nozzle spray, but very rapidly dissipates. The IN dose does seem to give me a “buzz”, hoping some rapamycin is getting in!
Feeling normal, no side effects. My sleep, which “seemed” off a bit early days (waking earlier), seems to have recovered to baseline normal for me. The one consistent noticeable effect is gastro centric…very regular, much softer, smaller poops. I also feel I am “less” gassy than historical. Have not ventured into diarrhetic. There is most definitely some HUGE fundamental change in my gut biome.
Just completed a half marathon 2 days ago, shaved 10 minutes from last same event/course (pre rapamycin). Arguably, perhaps in somewhat better endurance fitness leading into the race, but more importantly, NO discernible ill effects of my rapamycin protocol. I would think I would have noticed effects by now as I am a high daily exerciser, combination resistance + endurance, and not seeing any impact in reps, endurance, recovery. Reminder, I am NOT a high performance athlete, so not pushing high performance limits (nor tracking detailed performance metrics). But I am full on taking therapeutic dosing (11.9 ng/L weekly trough last check) of rapamycin, and clinically iron deficient anemic.
Been travelling and busy, but still working on the paper. Hoping the editors will give it a shot at publication. None of the rapa gods has replied to my sending them my case study, save for MK (“very interesting”). Will keep you posted.
Plan to do a next full blood panel in a few weeks, would mark 4 months on this protocol. Will be curious to see how markers are trending. I have paused my regular 8 week blood donations until I see my next full iron panel.