Looks like RapAdmin is on the cutting edge with this. I asked the free version of Grok what info was out there, and the answer was nothing:
Descovy (FTC/TAF), the HIV-1 PrEP medication, has been found to have age reversal qualities by researchers from the University of Colorado Anschutz Medical Center and UC San Diego. While it may be too early for other clinical results, some longevity focused biohackers are likely to have tried it and may have reported anecdotal evidence of its efficacy and its side effects.
Present a brief summary of such activity, followed by a more detailed survey and, finally, an evaluation. Although rigorous analysis of the same degree appropriate to clinical studies and lab research is not possible, do your best. The overarching view you should take is that of an elderly biohacker interested in optimal healthspan and longevity, and interested in trying this intervention.
Brief Summary
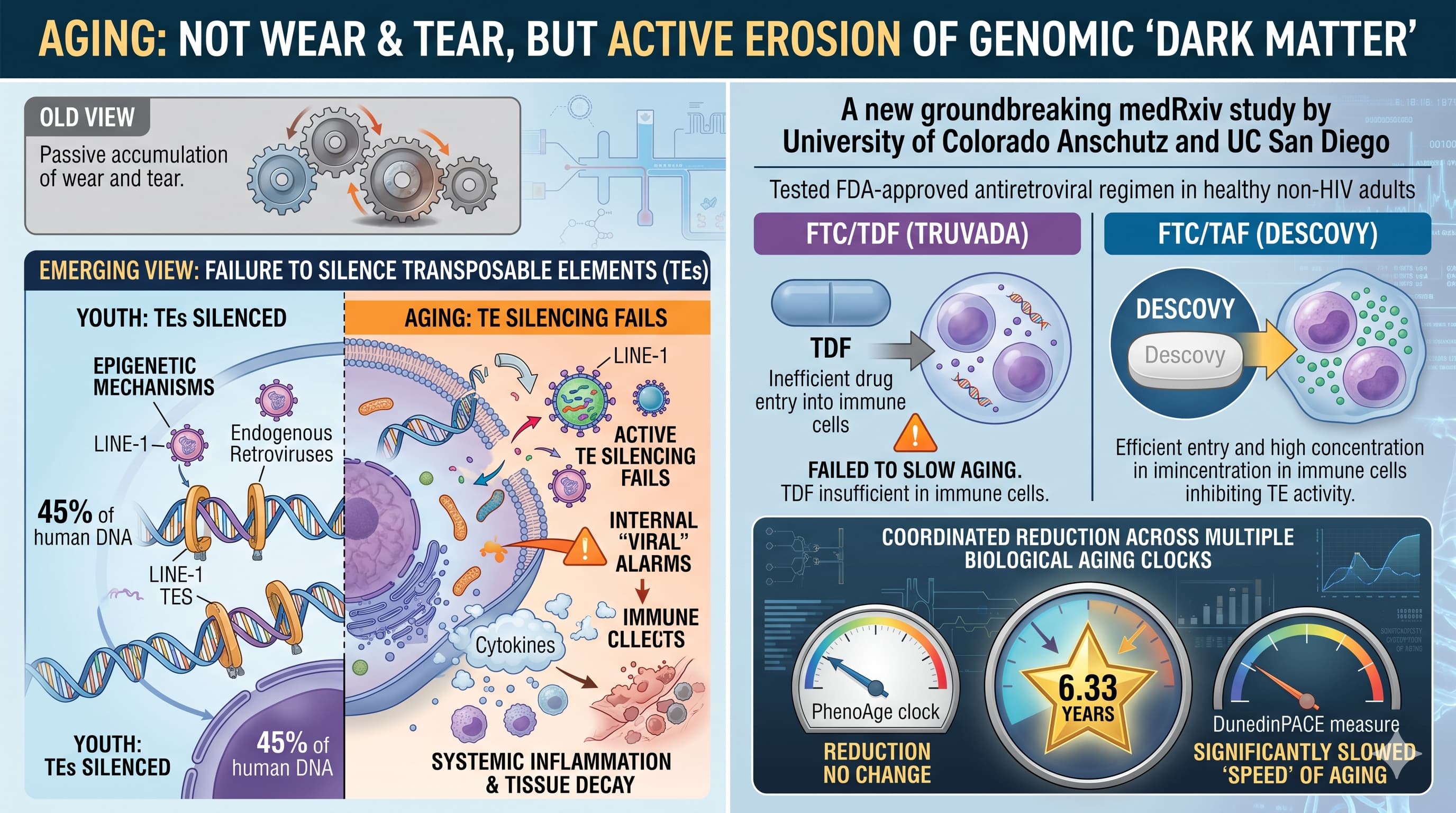
A March 2026 preprint from researchers at the University of Colorado Anschutz Medical Campus and UC San Diego reported that 12 weeks of FTC/TAF (Descovy, 200 mg/25 mg) produced coordinated reductions in multiple DNA methylation-based epigenetic aging clocks in healthy, HIV-negative adults (mean age ~28). Notable changes included a PhenoAge drop of 6.33 years and slowed DunedinPACE (a pace-of-aging measure). No similar benefits appeared with FTC/TDF (Truvada). The proposed mechanism involves superior intracellular penetration of tenofovir alafenamide (TAF), which inhibits age-associated retrotransposon (e.g., LINE-1) activity that drives inflammation via pathways like cGAS-STING.
This is the first human proof-of-concept for retrotransposon-targeted gerotherapeutics using an FDA-approved drug. Longevity communities (e.g., rapamycin.news forums) quickly discussed the findings, noting cheap generic availability and the intriguing surrogate-endpoint signal. However, because the preprint is only weeks old, there is essentially zero public anecdotal evidence from longevity-focused biohackers who have actually tried Descovy off-label for age reversal. Searches across web results, Reddit, X (Twitter), and longevity-specific sites turned up no “I took it and here’s what happened” reports—only study shares, mechanistic speculation, and PrEP-related side-effect chatter unrelated to longevity.Rapamycin
More Detailed Survey
The data come from a post-hoc re-analysis of biospecimens from two earlier randomized pharmacokinetic (PK) studies that tested directly observed dosing regimens (33 %, 67 %, or 100 % daily adherence for 12 weeks, with washouts) in HIV-negative volunteers. The FTC/TAF arm (N=36) showed statistically significant improvements across multiple clocks (PhenoAge, DunedinPACE, and several system-specific clocks including brain aging) plus declines in epigenetic proxies for IL-6 and a trend for C-reactive protein. The comparator FTC/TDF arm (N=43) showed none of these changes, consistent with TAF’s better ability to reach therapeutic levels inside immune cells where retrotransposons are active. No adverse events were reported in the original short PK protocols.Medrxiv
Key limitations (explicitly acknowledged by the authors):
- Not a dedicated anti-aging trial; no placebo arm in the re-analysis; young, healthy cohort; short duration.
- Epigenetic clocks are strong correlates of chronological age and mortality risk but are not yet validated as surrogates for clinical healthspan or lifespan extension.
- Mechanism is plausible (NRTIs have long been known to block LINE-1 reverse transcriptase in cell assays), but direct retrotransposon readouts were not measured here.
- Prospective, placebo-controlled studies in older adults with clinical endpoints are explicitly called for.
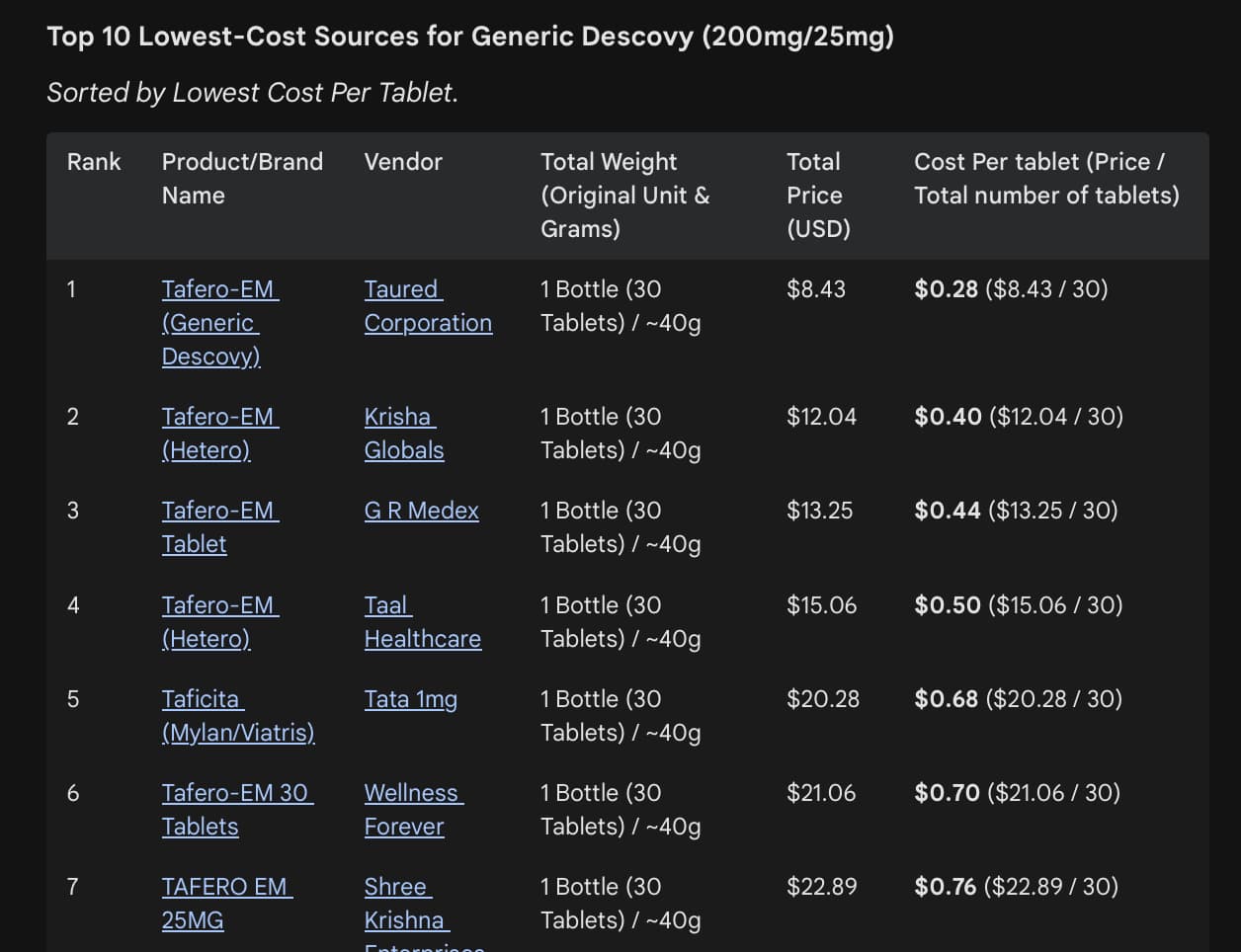
On the biohacker/anecdotal front, the landscape is silent. Longevity forums have noted the preprint and discussed sourcing low-cost Indian generics (e.g., Tafero-EM or Tafmune-EM, ~$30–60 for 12 weeks), but no users have posted self-experiment results, before/after epigenetic tests, side-effect logs, or subjective “youthfulness” reports. PrEP user communities (primarily on Reddit) discuss standard Descovy side effects—mild GI upset (diarrhea, nausea, abdominal pain), headache, fatigue—but these are in the context of HIV prevention, not longevity dosing, and most resolve quickly. Established PrEP safety data (DISCOVER trial and post-marketing) show Descovy is generally well-tolerated, with a better bone/kidney profile than Truvada but potential for modest weight gain and lipid shifts in some users. Rare serious risks (kidney impairment, lactic acidosis, liver issues, HBV flare if unscreened) require baseline and periodic lab monitoring. No long-term data exist for chronic use in healthy older adults.
Evaluation (from the perspective of an elderly biohacker optimizing healthspan)
At my stage of life, I’m pragmatic: I want interventions with a favorable risk–benefit ratio, measurable real-world payoff (function, disease-free years), and minimal downside. This Descovy signal is genuinely exciting on mechanistic grounds—retrotransposon derepression is an emerging hallmark of aging, and a cheap, oral, already-approved drug hitting it in humans is rare. The effect size on surrogates (roughly 20–25 % “reversal” relative to baseline age in just 12 weeks) would be eye-catching if it held in older people and translated to harder outcomes. The differential pharmacology versus TDF is clean and biologically coherent.
That said, this is still very early-stage. I would not rush to self-experiment. Reasons:
-
Evidence gap — Surrogate clocks in young volunteers do not equal proven healthspan extension in the elderly. I’ve seen plenty of “promising” biomarkers that failed to deliver clinically.
-
Safety in my demographic — My kidneys, liver, and mitochondria are not what they were at 28. Even though TAF is the “gentler” tenofovir, chronic NRTI exposure carries theoretical mitochondrial and metabolic risks; lipid/weight shifts could matter more when baseline cardiovascular risk is higher.
-
Practical hurdles — Requires a prescription (off-label use), reliable sourcing if going generic, and serial labs (kidney function, lipids, liver, possibly repeat epigenetic testing). Drug–drug interactions with common elderly polypharmacy are uncharted.
-
Opportunity cost — I already have higher-confidence levers (resistance training, Zone 2 cardio, protein timing, sleep optimization, rapamycin or metformin if tolerated). Adding an unproven NRTI would need to clear a higher bar.
Bottom line for me personally: I’m glad this paper exists and I’ll follow the inevitable follow-up trials closely—especially any that recruit older adults or measure functional outcomes (grip strength, gait speed, cognition, frailty indices). If a well-designed RCT ever shows durable clinical benefits with an acceptable safety margin, Descovy (or a refined retrotransposon inhibitor) could join my stack as a low-cost, low-burden add-on. Until then, it stays in the “intriguing but not yet actionable” column. For any fellow elderly biohacker considering it: get a physician partner, baseline everything, and treat it as an N=1 experiment only after weighing the unknowns. Right now, the data are too preliminary—and the real-world anecdotes too nonexistent—to justify routine use for longevity.