Here is an old (2004) and interesting study on dosing protocols on an angiogenic cancer mouse model.
Dosing of rapamycin is CRITICAL to achieve an optimal antiangiogenic effect against cancer
https://onlinelibrary.wiley.com/doi/epdf/10.1111/j.1432-2277.2004.00026.x
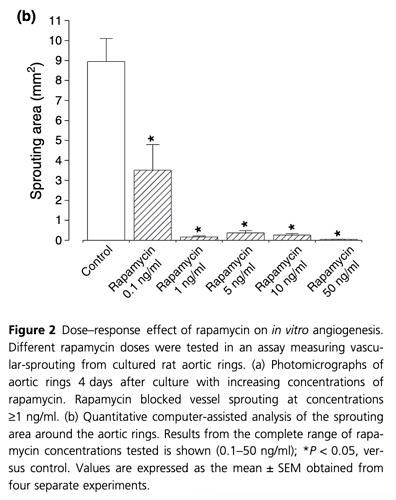
“Rapamycin has antiangiogenic activity against tumors. This has been discussed while addressing the problem of cancer in organ transplantation. Here we investigated effective dosing schedules against tumors and angiogenesis. Growth of established CT-26 colon adenocarcinoma tumors was measured in Balb/cmice treated with total equivalent rapamycin doses (1.5 mg/kg/day) given once a day, once every 3 days, or by continuous infusion. Tumors were MOST inhibited with continuous rapamycin infusion, and LESS BY BOLUS dosing. Interestingly, however, continuous dosing produced the lowest rapamycin blood levels(15 ng/ml). As rapamycin sensitive p70S6-kinase intracellular signaling is critical for angiogenesis, p70S6-kinase activation was measured in endothelialcells by Western blotting. Maximal p70S6-kinase inhibition occurred from1–5 ng/ml rapamycin. These same rapamycin concentrations optimally blocked vessel-sprouting from cultured aortic rings. Therefore, low-level rapamycin dosing most effectively controls tumors in mice. Importantly, antiangiogenic rapamycin levels are compatible with immunosuppressive doses, supporting its potential use in transplant patients with cancer”
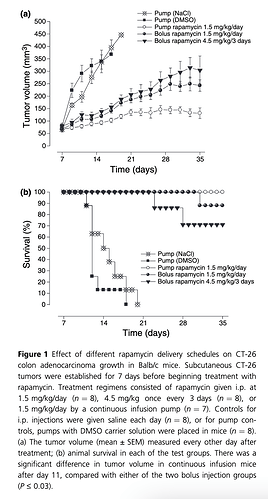
Rapamycin treatment (ME: interesting, no dietary delivery) was initiated for the remainder of the experimental period under three different dosing schedules that delivered the same total amount of drug over the test period:
(i)1.5 mg/kg/day, intraperitoneally (i.p.)
(ii) 4.5 mg/kg once every 3 days i.p.,
(iii) 1.5 mg/kg/day by continuous infusion using an i.p. placed Alzet pump
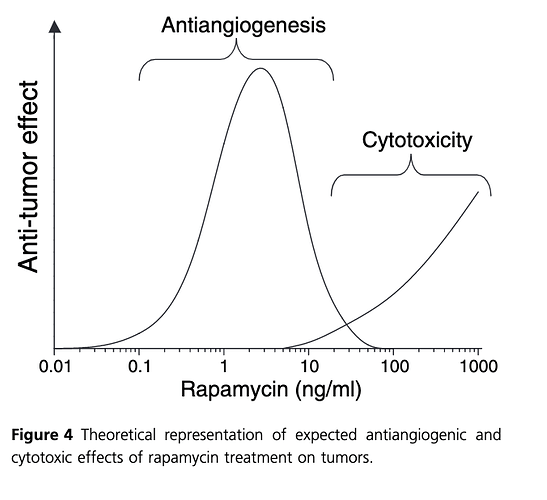
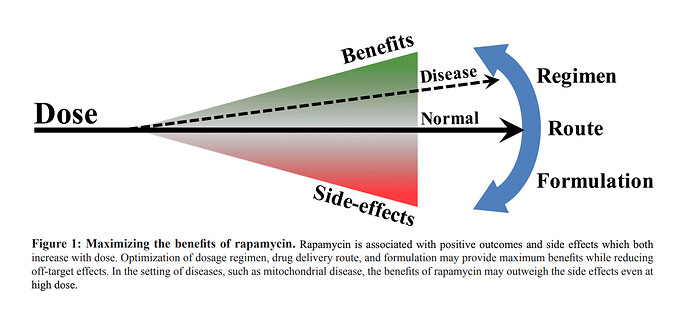
Taken together, we suggest that the effectiveness of rapamycin as an anticancer agent falls into two primary dose ranges, where low ng/ml concentrations of drug produce a potent antiangiogenic or potential anti-proliferative effect, and higher doses which can serve to have a direct cytotoxic effect on tumor cells (Fig. 4). Importantly, with regard to the use of the drug in trans-plant patients, the antiangiogenic dosing profile of rapamycin appears most compatible with normal immunosuppressive therapy.
Finally, it is interesting to consider the observation that continuous delivery of rapamycin provided the most effective in vivo treatment regimen.
This finding is similar to what has been observed with other known antiangiogenic agents, where low-level continuous-delivery methods induce the greatest anti-tumor effect [17–19]. The advantage of this type of therapy is that side effects tend to be low, making it possible to use this approach over along period of time to maintain constant ‘pressure’ against tumor expansion. It is notable that as antiangio-genic effects are directed against normal vessel cells, and not cancer cells, drug resistance to this form of therapy is less likely.
However, an antiangiogenic approach to tumor therapy is not without pitfalls. In particular, if the antiangiogenic agent is not also cytotoxic at the concentrations used, nests of cancer cells not large enough to require angiogenesis will continue to exist"
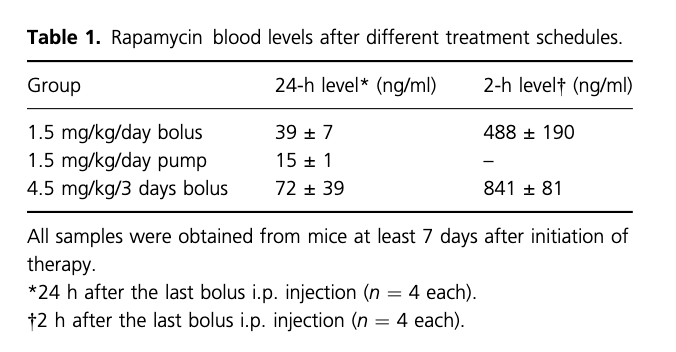
Surprised at the significant effects of the dosing protocols. The high dose every 3 days produced the poorest result compared to constant LOWER dosing? Namely, higher peak and higher likely trough. No measurements of TOR1 or TOR2 blunting, would have been very beneficial.
Meaning for translation in humans? Take rapamycin smaller doses higher frequency for lower peaks/trough, vs higher doses spaced father apart but more side effects? How about taking Everolimus, with shorter half life, smaller doses more frequently? Perhaps more potency, less side effects? Or just maintain a MIN trough level, however the rapalog/modality, but without side effects? And which side effects acceptable? Minor and/or lipid/CBC dysregulation?
I wish we had a readily available assay for mTOR inhibition: “70S6-kinase:rapamycin inhibits this signaling pathway because mTOR, which is blocked by rapamycin, is an upstream regulator of p70S6-kinase. Phosphorylation at the Thr389 site correlates with p70S6-kinase activity”
These findings could be a metabolism translation issue…mice have super high metabolism (ie cancer proliferation), and clear rapamycin very quickly, so any “gap” in rapamycin gives cancer cells a chance to proliferate (1). But even transient treatment of mice with rapamycin mid life (albeit continuous), can increase lifespan 60% (MK, 2016)
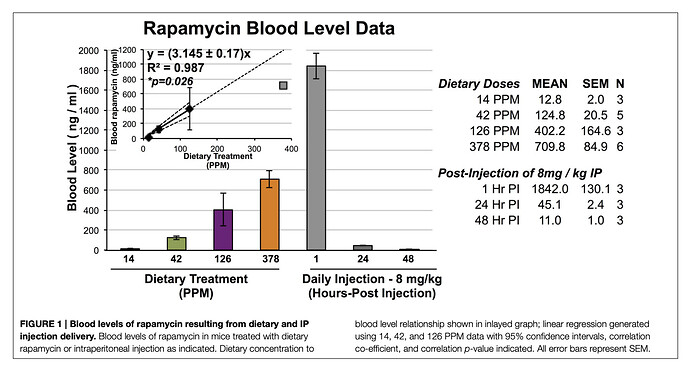
(1) Blood levels of rapamycin in IP injected mice goes from 1800 ng/L to 45 ng/L in 24 hrs.
An MK paper (2016)…6 YEARS AGO, he postulates about rapamycin regimen, dose, formulation
Rapamycin in aging and disease: maximizing efficacy while minimizing side effects
“Moving forward, we believe an emphasis should be placed on understanding how to achieve maximal efficacy from rapamycin treatment while minimizing side effects. While eRAPA and injected rapamycin are not directly comparable, as the delivery methods will have different pharmacokinetic parameters and may result in dramatically different tissue distributions, these results provide an initial foray into examination of dosing and delivery of rapamycin in a preclinical model of a medically relevant class of disease”
Yet what has changed, what have we learned in human translation since then? Pretty much nada…simple oral dosing to “max tolerable side effects”, and we don’t even know which side effects are acceptable long term?
“dramatically different tissue distributions”…indeed, but as yet unexplored.
We have no readily available tissue or blood or other assays for TOR or p70S6-kinase, or fully validated/translatable epigenetic aging, one of Peter Attia’s rapamycin concerns…“we have no good clinical biomarker”
Although they did measure an mTOR signal in the seminal cancer/rapamycin/GFJ study: “mTOR phosphorylates p70S6 kinase (p70S6K) at threonine 389 which most closely correlates with activity in vivo(23). p70S6K phosphorylation at Thr389 was measured in peripheral blood lymphocytes (PBL) as a potential biomarker of rapamycin activity. Only subjects in the sirolimus alone study underwent determination of phosphorylated p70S6K”