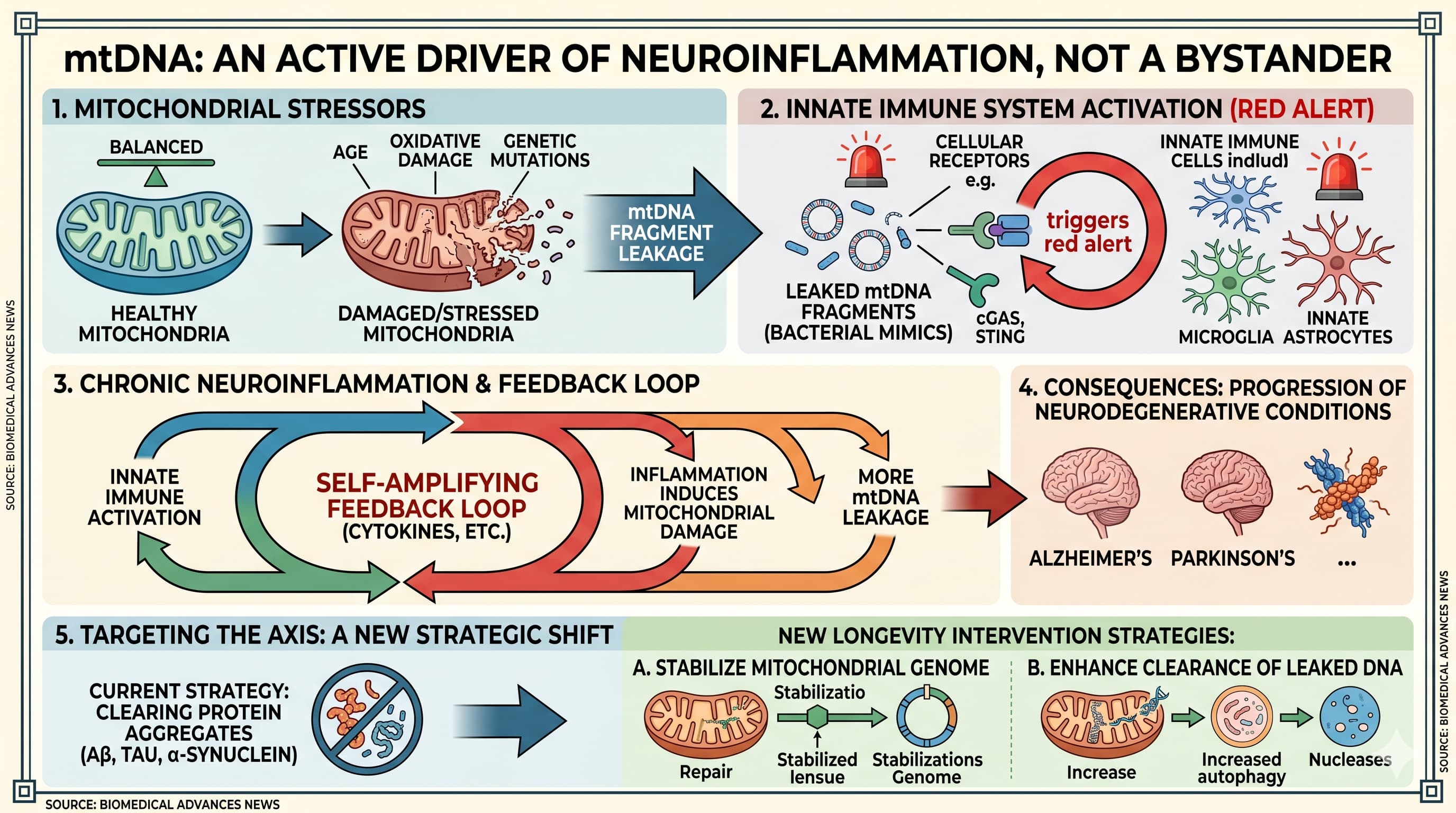
For decades, mitochondrial DNA (mtDNA) was viewed as a passive blueprint, a circular genome tucked safely within the “powerhouse” of the cell. This perspective is undergoing a radical shift. A comprehensive review published in Ageing Research Reviews (2026) by researchers at the University of Coimbra, Portugal , positions mtDNA as an active driver of chronic neuroinflammation rather than a mere bystander of cellular injury. When mitochondria are stressed—by age, oxidative damage, or genetic mutations—they leak fragments of their DNA into the cytosol or the extracellular space. To the cell’s innate immune system, these fragments look suspiciously like bacterial invaders, triggering a “red alert” that fuels the progression of Alzheimer’s, Parkinson’s, and other neurodegenerative conditions.
The mechanism of this “betrayal” is the mitochondria-immune axis. Under physiological conditions, a protein called TFAM coats and protects mtDNA. However, when quality control mechanisms like mitophagy (the recycling of damaged mitochondria) fail, oxidized and fragmented mtDNA escapes. These fragments exit through specialized channels like the voltage-dependent anion channel (VDAC) or the mitochondrial permeability transition pore (mPTP). Once in the cytosol, mtDNA is detected by sensors such as cGAS-STING and NLRP3. This detection triggers a cascade: the production of type I interferons and pro-inflammatory cytokines like IL-1β and IL-18. This is not a localized event; the review details how mtDNA can be packaged into mitochondrial-derived vesicles (MDVs) or exosomes and shipped to neighboring cells, effectively “spreading” the inflammatory signal throughout the brain’s microenvironment.
This process creates a self-amplifying feedback loop. Inflammation induced by leaked mtDNA further damages mitochondria, leading to more leakage. In Alzheimer’s disease models, Amyloid-β and Tau fibrils have been shown to directly promote this mtDNA release, which then drives synapse loss and cognitive decline. By reframing neurodegeneration as a disorder of a dysfunctional mitochondria-immune axis, the authors suggest that the most effective “longevity” interventions may not be those that simply clear protein aggregates, but those that stabilize the mitochondrial genome or enhance the cellular machinery responsible for clearing leaked DNA before it can trigger an immune response.
Actionable Insights
The practical takeaway for longevity optimization is the prioritization of mitochondrial quality control and mitophagy to prevent the accumulation of “pro-inflammatory” mtDNA.
-
Mitophagy Induction: The paper highlights natural compounds like Urolithin A and Nicotinamide Riboside (NR) as promising enhancers of mitochondrial dynamics and mitophagy. These compounds help ensure that damaged mitochondria are recycled before they can leak DNA.
-
Pharmaceutical Interventions: The review notes several drugs currently in trials that target these pathways. Metformin and Rapamycin are noted for their ability to induce autophagy/mitophagy and reduce oxidative stress.
-
Antioxidant Support: Reducing reactive oxygen species (ROS) is critical, as oxidized mtDNA is more likely to escape and is less easily degraded by the cellular “clean-up” enzyme TREX1. Resveratrol and mitochondrial-targeted antioxidants are identified as potential tools for preserving mtDNA integrity.
-
Biomarker Monitoring: While still in the research phase, monitoring cell-free mtDNA (cf-mtDNA) in blood or cerebrospinal fluid may eventually serve as a “smoke detector” for early-stage neuroinflammation.
Source:
- Open Access Paper: Linking mitochondrial DNA release to neurodegeneration and cognitive decline
- Institution: Center for Neuroscience and Cell Biology (CNC-UC), University of Coimbra.
- Country: Portugal.
- Journal Name: Ageing Research Reviews.
- Impact Evaluation: The impact score (CiteScore) for this journal is 25.7 (based on 2024-2026 trends), therefore, this is a High impact journal.
Related Reading:
- Role of mitochondria in physiological activities, diseases, and therapy
- Hazel Szeto, SS-31 peptide, the World's First FDA-Approved Mitochondria-targeted Drug (Longevity Summit, 2025)
- A Masterclass in Mitochondria | Martin Picard, PhD | Uncommon Living 17
- Melatonin as a Guardian of Mitochondria: Mechanisms and Therapeutic Potential in Neurodegenerative Diseases (Jan 26)
- Mitochondrial Medicine, Podcast with Tom Benson of Mitrix Bio
- Longevity Treatments for a 91-Year-Old? A Bold Bet in Silicon Valley’s Immortality Race (Mitrix)