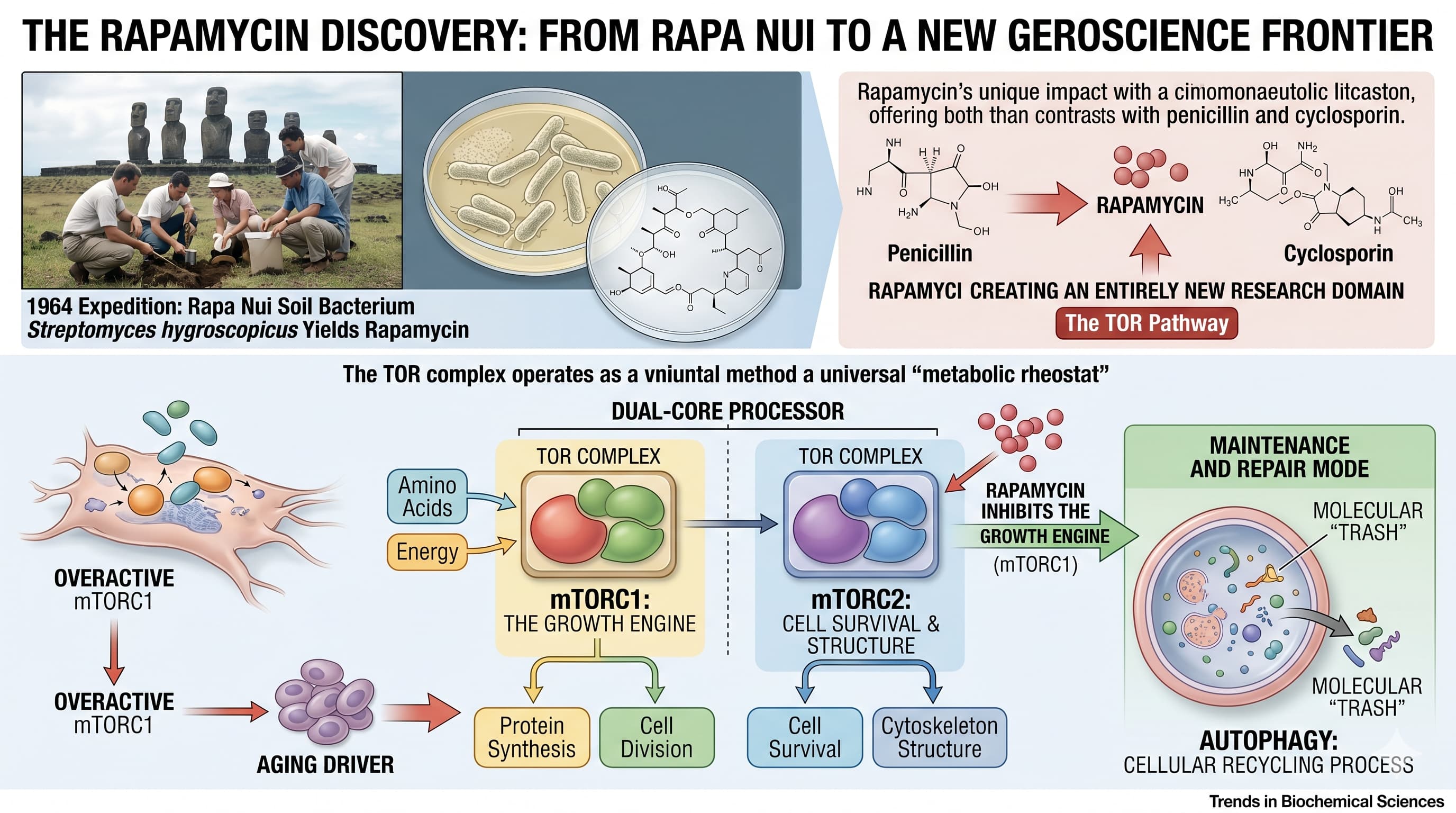
In 1964, a Canadian scientific expedition to one of the most isolated places on Earth, Rapa Nui (Easter Island), set out to study human adaptability in the face of modernization. Instead, it yielded a soil bacterium, Streptomyces hygroscopicus , that produced a compound now recognized as the most potent longevity-extending agent in the pharmacological arsenal: rapamycin.
Published in Trends in Biochemical Sciences , this feature review marks the 50th anniversary of rapamycin’s first reporting. Unlike penicillin or cyclosporin, which merely expanded existing fields, rapamycin is unique because its study actually created an entirely new research domain. It revealed the Target of Rapamycin (TOR) pathway, a universal “metabolic rheostat” that exists in everything from yeast to humans.
The paper details how the TOR complex functions as a dual-core processor. mTORC1 acts as the growth engine, sensing amino acids and energy to drive protein synthesis and cell division. mTORC2 governs cell survival and the structural integrity of the cell (the cytoskeleton). For decades, the “Big Idea” in geroscience has been that overactive mTORC1 is a primary driver of aging. By inhibiting this growth engine, rapamycin forces the cell into a “maintenance and repair” mode, characterized by autophagy —the cellular recycling process that clears out molecular “trash”.
The review traces the evolution from the “first generation” natural product to “third-generation” bisteric inhibitors like RapaLink-1 , which can more precisely target the pathway without triggering the negative feedback loops that hindered earlier cancer treatments. As we move into 2026, the field is shifting from simple inhibition to “nuanced modulation”—tuning the pathway up or down depending on whether the goal is treating a tumor, preventing neurodegeneration, or extending the healthy human lifespan.
Actionable Insights For those looking to apply these findings to longevity, the paper offers several critical take-home messages:
-
Specificity Over Blunderbuss Inhibition: Chronic, high-dose rapamycin can inadvertently inhibit mTORC2 , which may lead to impaired glucose tolerance and suppressed immunity. The “sweet spot” for longevity likely involves selective mTORC1 inhibition.
-
The “Off-Label” Reality: While formal human longevity trials are absent, a survey of 333 adults using rapamycin off-label for anti-aging reported improved cognition and mood with no major adverse events. This suggests a manageable safety profile in healthy cohorts, though controlled trials are still the gold standard.
-
Companion Animal Data: The ongoing TRIAD (Test of Rapamycin In Aging Dogs) trial, involving 580 senior dogs, is currently the most robust proxy for how rapamycin might perform in a complex, “real-world” mammalian environment similar to humans.
-
Nutrient Sensing Synergy: Because mTORC1 is specifically sensitive to leucine and arginine through sensors like SESTRIN2 and CASTOR1 , dietary protein timing remains a potentially powerful non-pharmacological lever to modulate this pathway.
Context
- Open Access Paper: The rapamycin sTORy: 50-year journey from Easter Island to the frontiers of biology and medicine
- Institution: CNRS UMR9018, Université Paris-Saclay, Gustave Roussy.
- Country: France.
- Journal Name: Trends in Biochemical Sciences (TIBS).
- Impact Evaluation: The impact score (CiteScore/JIF) of this journal is approximately 13.8, evaluated against a typical high-end range of 0–60+ for top general science journals; therefore, this is a High impact journal in the field of biochemistry and molecular biology.