Another potent hair growth stimulating drug:
The immunosuppressive immunophilin ligand, cyclosporine A (CsA), is an 11-amino-acid cyclic peptide produced by Cylindrocarpon lucidum [1]. Since the discovery of its efficacy for the sup- pression of renal allograft rejection [2], CsA has become a staple of immunosuppressive therapy in clinical medicine for a wide range of immune-mediated diseases. …
CsA Is a Potent Hair Growth Stimulator
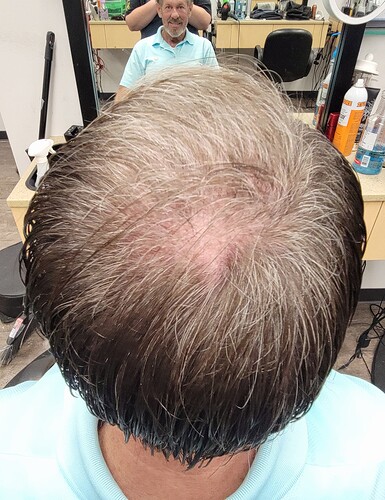
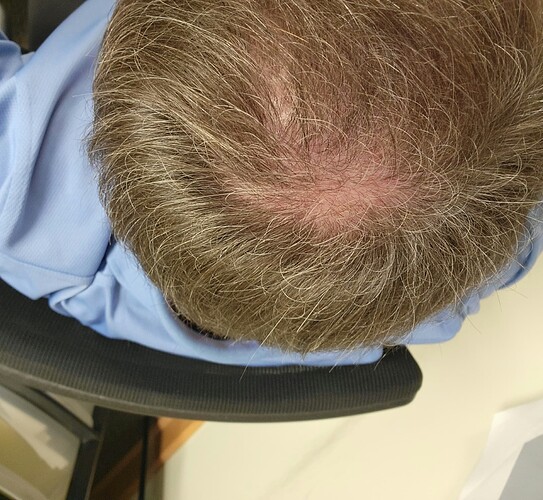
Perhaps the most intriguing and most common of the many adverse effects of CsA (Table 1) is the curiously strong hair growth stimulation this fungal product exerts in many patients receiving CsA, >88% of whom develop a reversible, drug-induced hypertrichosis [10]. Hypertrichosis describes the transformation of tiny vellus hair follicles (HFs), which produce a fine, short, and almost invisible hair shaft, into large terminal HFs that generate thick, long, pigmented hair [11]
In fact, no other drug used in clinical medicine more reliably stimulates human hair growth than CsA [12–14], albeit typically in cosmetically undesired locations. CsA directly targets the HF, promotes its hair shaft formation and prolongs the growth stage of the hair cycle (anagen) in organ-cultured human scalp HFs; that is, in the absence of systemic immunity [15,16]. Major hair growth stimulation is also seen in mice, where CsA activates both resting (telogen) HFs (Figure 1A) to enter into anagen [7] and suppresses the involution of anagen HFs into the apoptosis-driven regression stage of the hair cycle (catagen) [17,18] (Figure 1B).
These hair-growth-stimulatory effects cannot be toxicity induced, since cytotoxic agents almost invariably promote hair loss and catagen [19,20]. Immunosuppression also provides no persua- sive explanation, since even (already severely immunocompromised) nude mice (Foxn1-/-) shed their nudity and develop striking hair growth after CsA treatment [21], while other potent immuno- suppressants like glucocorticosteroids strongly induce catagen and freeze HFs in telogen [17,22]. Moreover, anagen induction by CsA in mice is accompanied by significant increases in the number of HF-associated mast cells, macrophages, T cells, and Langerhans cells [23]. We have therefore long argued that dissecting the unknown molecular mechanisms that drive CsA-induced hair growth stimulation promises important new insights into the as-yet-unclear molecular controls of human hair growth and into novel pharmacological strategies for targeting these controls [7,17].
As hair aficionados among dermatologists know well, the vellus-to-terminal HF transformation that underlies hypertrichosis represents a very complex and rather dramatic transformation pro- cess of a human (mini-)organ [27] that by no means can be explained only by NFAT-dependent apoptosis suppression in a regressing anagen HF [9] or by bulge stem cell activation in telogen HFs [26] (Figure 1A,B). Rather, this radical organ transformation process is primarily driven by the HF mesenchyme, namely by the uniquely inductive fibroblasts of its dermal papilla (DP). The number, volume, and papilla morphogen secretion of this mesenchymal command center of the HF dictate HF size and thus the thickness of the hair shaft and the duration of anagen, and therefore hair shaft length [19,28,29].
Full Paper:
Beyond the NFAT Horizon: From Cyclosporine A-Induced Adverse Skin Effects to
Novel Therapeutics
We exemplify this through our recent discovery that CsA suppresses the potent Wnt inhibitor, secreted frizzled related protein (SFRP)1, in human hair follicles, thereby promoting hair growth and causing hypertrichosis.
http://sci-hub.wf/10.1016/j.tips.2021.02.001