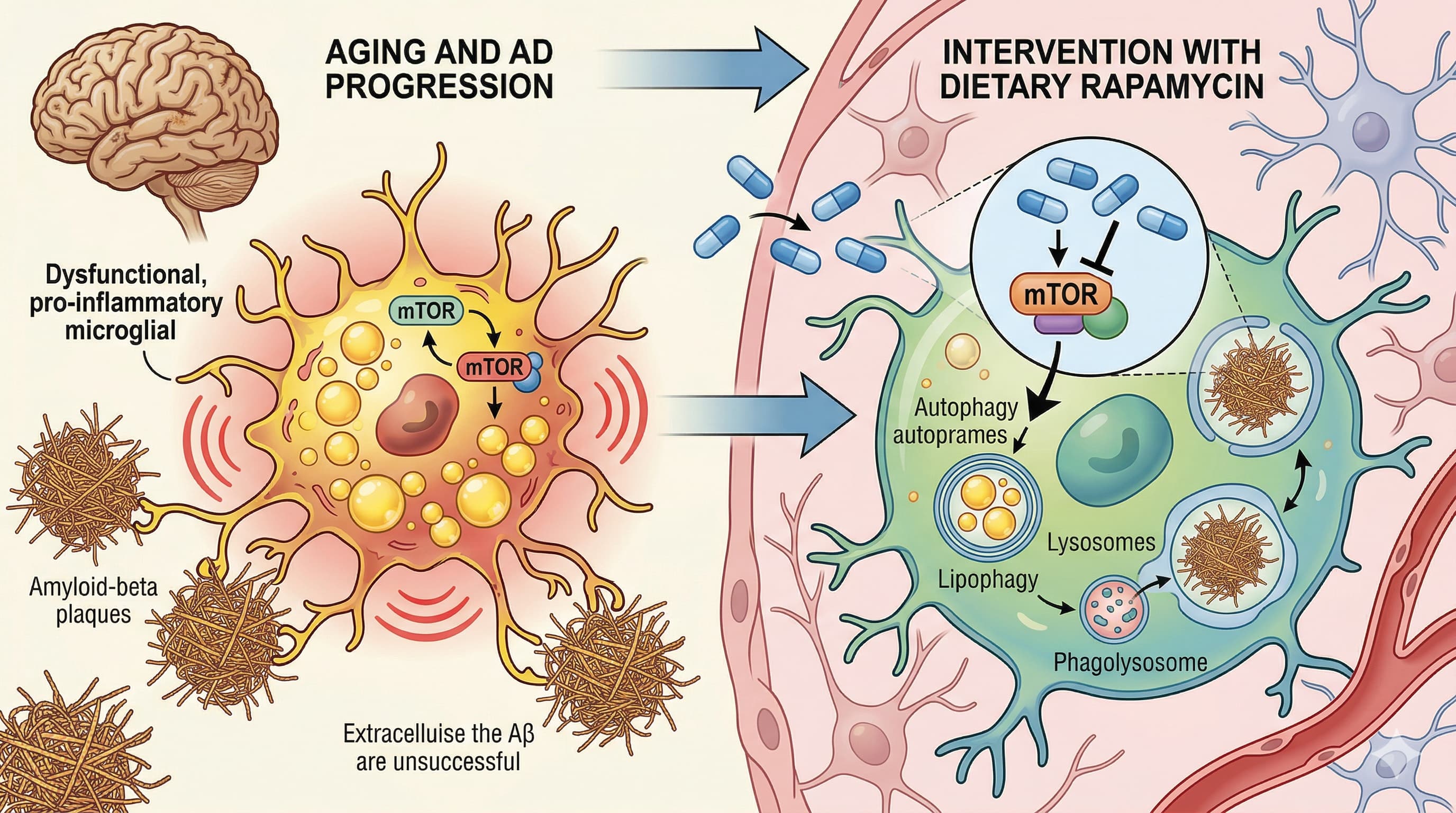
The utility of rapamycin for extending lifespan is a cornerstone of longevity biotechnology, but its efficacy in neurodegenerative conditions like Alzheimer’s Disease (AD) has yielded conflicting data. This paper clarifies a critical mechanism of action: rapamycin acts primarily by rescuing the metabolic dysfunction of microglia, the brain’s resident immune cells.
During aging and AD progression, microglia accumulate intracellular lipid droplets, transitioning into a dysfunctional, pro-inflammatory state that blunts their ability to clear amyloid-beta (Aβ) plaques. This intervention demonstrates that early, continuous dietary rapamycin suppresses mTOR activity, thereby triggering autophagy (specifically lipophagy) to clear these lipid droplets. By metabolically “unclogging” the microglia, rapamycin restores their lysosomal degradative capacity, allowing them to effectively consume and destroy Aβ fibrils.
Crucially, the study reveals a profound sexual dimorphism in response to mTOR inhibition. While rapamycin successfully reduced Aβ plaque burden and improved spatial working memory and fear memory in female 5xFAD and hAPPNL-G-F mice, male mice exhibited only marginal histological improvements and no significant cognitive rescue. Transcriptomic data aligned with this, showing a much stronger modulation of lipid metabolism and phagocytosis pathways in females. This suggests that the baseline pathology, lipid load, or pharmacokinetic response to rapamycin in AD is heavily sex-dependent, necessitating sex-stratified approaches in human clinical translation.
Source:
Related Reading:
1 Like
Study Design Specifications
-
Study Type: Preclinical in vivo intervention and in vitro mechanistic assays.
- In Vivo Subjects:
-
Species/Strain: Heterozygous 5xFAD mice (C57BL/6-SJL hybrid) and hAPPNL-G-F knock-in mice (C57BL/6J).
-
Sex: Both male and female cohorts evaluated.
-
N-Number: n=10 per group for behavioral tests; n=5-6 per group for histopathological and biochemical analyses.
-
Dosing: Dietary rapamycin at 14 ppm active compound (yielding approximately 2.33 mg/kg/day), administered continuously for 90 days starting at 2 months of age.
-
In Vitro Subjects: Primary murine microglia and BV2 microglial cell lines.
Mechanistic Deep Dive
The central thesis bridges mTORC1 inhibition, lipid metabolism, and autophagic flux.
-
mTOR & Autophagy: Rapamycin reduced phosphorylated mTOR without altering total mTOR protein levels. This triggered a downstream reduction in phosphorylated S6 ribosomal protein specifically within plaque-associated microglia.
-
Lipid Clearance: In vitro assays using oleic acid-treated BV2 cells showed that rapamycin significantly reduces lipid droplet size and content. This effect is strictly autophagy-dependent, as the addition of chloroquine (an autophagy inhibitor) completely abolished the lipid-clearing benefits.
-
Phagocytic Degradation: The clearance of lipid droplets functionally rejuvenated the microglia. While rapamycin did not increase the raw rate of Aβ uptake (phagocytosis), it significantly accelerated the lysosomal degradation of Aβ within the cell. RNA-sequencing corroborated this by showing a downregulation of hyperactive immune-phagocytic genes (Itgb3, Tlr2, Fcgr2b) and an upregulation of lipid-processing phospholipases (Pla2g3, Etnppl).
Novelty
This paper successfully isolates microglial lipophagy as a primary downstream mechanism for rapamycin’s neuroprotective effects in amyloidosis, moving the narrative away from generalized neuronal autophagy. Furthermore, it provides stark, quantifiable evidence that female AD models are significantly more responsive to early mTOR inhibition than males, highlighting a critical variable often masked in pooled-sex analyses.
Claims & Verification
Claim 1: Rapamycin treatment reduces amyloid-beta (Aβ) plaque burden and mitigates cognitive deficits in Alzheimer’s disease.
-
Verification: A vast body of pre-clinical literature supports mTOR inhibition for reversing cognitive deficits and clearing toxic protein aggregates in murine models. A recent meta-analysis confirms that rapamycin shortens escape latency in the Morris Water Maze and upregulates insulin-degrading enzymes in transgenic mice. However, early-phase human trials (e.g., the CARPE_DIEM and ERAP trials) have failed to replicate cognitive rescue or definitive Aβ clearance in patients, though pilot studies have noted improvements in cerebral blood flow among APOE4 carriers.
-
Evidence Level: Level D (Pre-clinical) for core disease modification; Level C for emerging human biomarker data. [Flag: Relies heavily on preclinical models].
-
Translational Gap: The leap from treating early-onset transgenic murine amyloidosis to modifying late-onset sporadic human Alzheimer’s disease remains completely unproven.
-
Supporting Evidence:
Claim 2: Rapamycin restores microglial function by inducing lipophagy to clear lipid droplets, thereby accelerating Aβ degradation.
-
Verification: The accumulation of lipid droplets in aging microglia is a verified phenomenon that drives a dysfunctional, pro-inflammatory state. However, the exact role of the mTOR pathway in this mechanism is the subject of fierce scholarly debate. While this analyzed study suggests mTOR inhibition (rapamycin) is required to clear plaques via lipophagy, competing high-impact literature asserts that mTOR activation in microglia is necessary to upregulate Trem2—the receptor responsible for Aβ plaque uptake—and that rapamycin actually worsens Aβ plaque burden by suppressing this pathway.
-
Evidence Level: Level D (In vitro / Animal). [Flag: Highly contentious mechanistic claim].
-
Translational Gap: This mechanism has only been demonstrated in immortalized murine cell lines (BV2) and genetically engineered mice. Whether human microglia respond to rapamycin with lipophagy or harmful Trem2 downregulation requires direct human tissue analysis or advanced human-induced pluripotent stem cell (iPSC) modeling.
-
Supporting Evidence:
Claim 3: The therapeutic efficacy of mTOR inhibition against Alzheimer’s pathology exhibits profound sexual dimorphism, primarily benefiting females.
-
Verification: The sexually dimorphic response to rapamycin is heavily documented in longevity literature. The National Institute on Aging’s Interventions Testing Program (ITP) previously established that rapamycin extends lifespan and healthspan much more robustly in female mice than in males, likely due to pharmacokinetic differences (females maintain higher circulating blood levels of the drug) and baseline metabolic variations.
-
Evidence Level: Level D (Pre-clinical).
-
Translational Gap: It is entirely unknown if this sexual dimorphism translates to human patients. Human clinical trials of rapamycin for aging and neurodegeneration have not yet published adequately powered, sex-stratified data.
-
Supporting Evidence: