@MAC great find - thanks for posting. From that paper you added, the 2014 (Molecularly targeted therapies for recurrent glioblastoma: current and future targets (UCSF)), I found this paper particularly interesting with regard to the rapalog “Everolimus” - which people here are starting to take instead of (or in addition to) rapamycin. It seems that Everolimus might be particularly good at passing through the Blood/Brain Barrier (BBB).
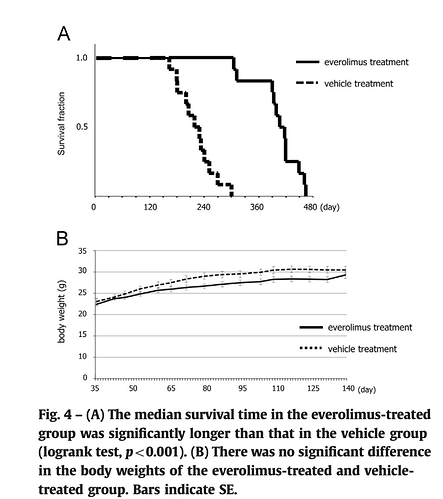
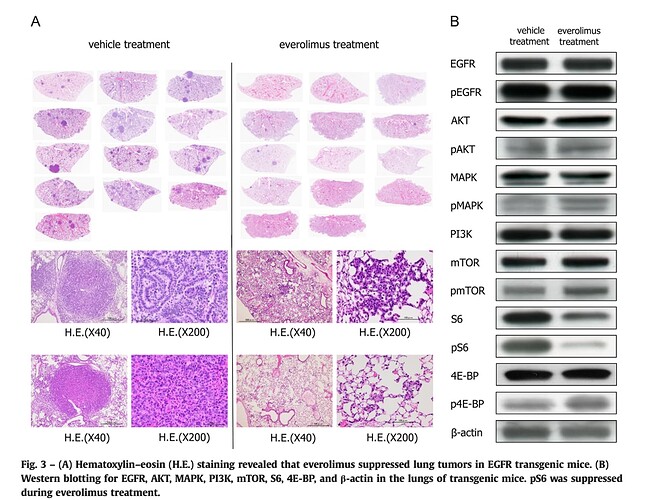
Everolimus — Everolimus is a specific mTOR inhibitor with lipophilic properties that allow it to readily traverse the BBB. The use of everolimus with bevacizumab as a combined modality therapy for newly diagnosed glioblastoma has shown some efficacy; the PFS compared favorably to previous reports of standard radiation and temozolomide therapy.(47).
The referenced paper (47) is this paper:
Phase II study of concurrent radiation therapy, temozolomide, and bevacizumab followed by bevacizumab/everolimus as first-line treatment for patients with glioblastoma.
Full Paper here: https://www.hematologyandoncology.net/files/2013/05/ho0412_hainsworth1.pdf
Note: For people interested in learning more about the rapalog (acts like rapamycin) drug called everolimus - see this discussion thread: Everolimus instead of Sirolimus / Rapamycin? Anyone else trying?
In it, it states:
Conclusions: The use of bevacizumab and everolimus as part of first-line combined modality therapy for glioblastoma was feasible and efficacious.
Additionally, from the paper:
Four weeks after the completion of radiation therapy, patients began oral everolimus 10 mg daily, and continued bevacizumab every 2 weeks; therapy continued until tumor progression or unacceptable toxicity.
The dose of everolimus was modified on the basis of either hematologic or nonhematologic toxicity. Everolimus was interrupted if grade 3 or 4 toxicity occurred, and was held until the toxicity improved to grade 1 or less. At that time, dosing was resumed with a 1-level dose reduction (5 mg orally once daily). Patients were monitored for development of hyperlipidemia or hyperglycemia; if these events occurred, appropriate medical management was initiated.
…
Patients with objective response or stable disease began treatment with concurrent bevacizumab and everolimus. Patients were reevaluated at 8-week intervals with MRI scans until tumor progression was documented.
Interestingly, researchers have been testing Everolimus with many other cancer drugs, where they are finding that not only does Everolimus cross the blood brain barrier well, but it also helps other cancer drugs enter the brain and target tumors better (it seems). Here are some examples:
The research team at the Institute of Cancer Research, London, led by Professor Chris Jones, found that a drug called everolimus could enhance vandetanib’s capacity to pass through the blood-brain barrier in order to treat the cancer.
Combining the two drugs increased the amount of vandetanib in the brains of mice with DIPG by 56 per cent. The researchers also found that the treatment extended survival in mice by 14 per cent compared with those receiving a control treatment.
Source: Scientists identify new drug combination for children with incurable brain cancer
And:
Our results show that the efficacy of dasatinib treatment of PDGFRα-driven HGG was enhanced with everolimus and suggest a promising route for improving targeted therapy for this patient population.
Source: Everolimus improves the efficacy of dasatinib in PDGFRα-driven glioma