Is there an overview where we can see the results of all the ITP tests done (their website sucks so much, I can’t even find the substances they tested without a direct link given to me and why did they not include an overview or a link to the results in that page is beyond me )
This document covers the best-performing drugs tested by the ITP:
The ITP uses genetically diverse mice that mimic a natural population.
HET3 (also called UM-HET3) mice are an established genetic diversity model for aging intervention studies, currently used by the NIA’s Intervention Testing Program (ITP). HET3 mice are a genetically heterogeneous stock, produced by breeding CByB6F1/J and C3D2F1/J hybrids together.
HET3 (also called UM-HET3) mice are a heterogenous stock produced by a four-way cross by breeding CByB6F1/J hybrid females and C3D2F1/J hybrid males together. HET3 mice are the first filial generation offspring of this cross. As such, HET3 mice have segregating alleles from the genetic backgrounds C57BL/6J, BALB/cByJ, C3H/HeJ, and DBA/2J. Each animal in the test population is genetically unique, but each shares half of its non-mitochondrial genome with every other test mouse. From this perspective, all the animals can be considered siblings. Due to this breeding scheme, the population-wide genetics of HET3 mice are reproducible and consistent.
HET3 mice are used as the model organism for the National Institute on Aging’s Interventions Testing Program (ITP). The ITP was established to facilitate research by multiple laboratories on drugs that may modulate aspects of aging and increase healthy lifespan in mice.
Seems heterogeneous enough for me.
They use the doses recommended by the experts in the field. Time periods are starting early in life (9 months) and mid-life (20 months) as they are the two reasonable starting points in humans. Most people don’t think that starting earlier (i.e. childhood) is a reasonable approach in humans.
I can’t see how their approach could be improved on by much.
Doesn’t matter, most mammals age similarly. See this post: Most mouse research doesn't translate to humans - why do we think rapamycin is different?
Most drugs and supplements are sold as a single substance. Most chemistry/science/medicine is done testing a single substance so that you can identify the biological method of action, binding affinity of the specific drug or compound, etc. If you disagree with the fundamental premise of modern science and medicine, well thats a bigger issue.
Systems biology has great value and is being worked on, but I think its only now becoming possible with the extreme level of computational power required; like protein folding.
Always good to have an alternative view to challenge our thoughts and hypotheses.
Were the mice exercised? On time restricted feeding? Or was the supplement the only intervention?
If not, we will not know if the supplements add to the life extension provided by exercise or TRF, or both.
Extensions, how measured? Life expectancy at birth? Or life expectancy at the time the intervention was initiated? Take a human with life expectancy of 80. If the 10 percent improvement is of the life expectancy at birth, then that is 80 plus eight years. If the intervention were started at 60, his life expectancy is another 20. So the 10 percent improvement is ten percent of 20 years, or two years; a piddling, improvement that can be achieved by exercise and/or diet restriction.
If the intervention were started at 60, his life expectancy is another 20. So the 10 percent improvement is ten percent of 20 years, or two years; a piddling, improvement that can be achieved by exercise and/or diet restriction.
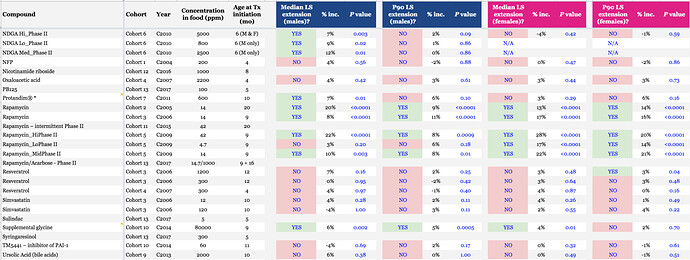
This is an incorrect interpretation of the effect of late life interventions on lifespan. In this study, The ITP found that rapamycin was able to extend 90th percentile lifespan (the best proxy we have for “maximal” lifespan) in mice when introduced at 600 days of age (around ~60 y/o human years) by 9% and 14% in males and females respectively.
The ITP does not measure lifespan increase in terms of change in expected life left, as you suggest; rather, they compare intervention and control by median lifespan increase (e.g. comparing the median lifespan of rapa mice vs control mice) or 90th percentile lifespan increase (i.e. comparing how many more of the longest lived mice are rapa vs control)
Exactly.
Here is a short summary from Peter Attia’s podcast with Richard Miller, leader of the ITP program:
Difference betwee median vs maximum survival [50:00]
- Median survival is the age at which half of the subjects have died and half of them haven’t died yet
- Maximum survival is a “folk notion”
- Basically, how long has the oldest mouse or a person who’s ever lived
- And that’s a value with very limited statistical appeal because the oldest person in a group of 100 people is not going to live as long as the oldest person in a group of 1,000,000 people or 100 million, and so on
A better statistic/best substitute for the oldest subject that lived is: what is the age at which 90% of the subjects have lived?
- This 90% threshold, it what’s crudely referred to that as “maximum lifespan”
- It’s an important concept in terms of understanding how to interpret survival statistics and survival curves
Let’s say we have a group of animals that are dying in the 30s, 40s, and 50s (in human equivalent years)…
- Then an intervention extends their median lifespan up to 60
- That becomes a great public health benefit (think immunizations that we all get, why people don’t smoke, etc.)
- But those types of median life changing interventions haven’t had any real effect on how much longer you’re going to live when you’re 70
- So drugs that only extend the median—and don’t affect the age at death that the longest lived 5% or 10%—might be interesting in some ways, but they are considered less plausible as candidates for anti-aging drugs
Interventions that affect “maximum lifespan”
- But if you had a drug, or a diet, or some intervention that authentically slowed the aging process, then it would extend expectancy of additional healthy years of life, even for those who are already quite old
- And THAT will modify the age of death at the 90th percentile.
- In other words, if you have a drug that’s authentically slowing aging, one of the things you most want to see is that the very oldest animals in this drug treated group are living longer than the very oldest animals in the untreated control group
…
Explaining the total lifespan increase
- The 9% or 14% more life basically translates to 25% more life once you’ve reached mid-life in terms of the number of additional years or months of life after you give the drug
- However, Rich says that statistic is not ”terribly informative”
- Thought experiment: Someone on a ventilator that is on death’s doorstep. Without the ventilator, they’re going to die today. And you leave them on the ventilator for one more day and then you give up and they die. So you’ve doubled their lifespan. — “not a very useful statistic to calculate ”
- But in terms of the biology and pathobiology, the change in the overall median is important.
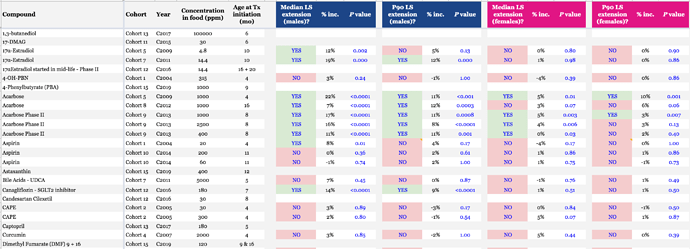
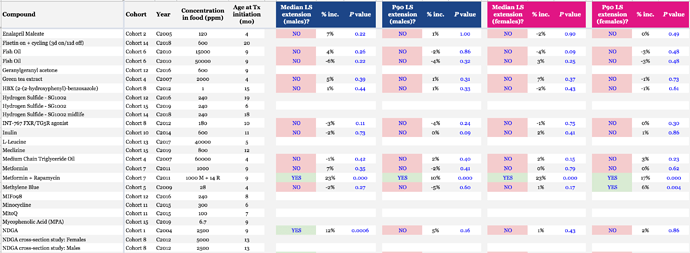
ITP Drug Testing Results:
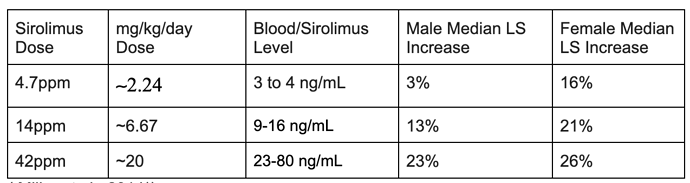
Do we know if Richard Miller takes rapa and what dose?
Thats actually a funny question WRT Richard Miller.
Richard Miller has not officially, explicitly stated that he is taking rapamycin. But, in his Feb, 2021 Podcast with Peter Attia, Peter asked him "Has your conviction about any of the molecules tested in the ITP studies led you to taking any?
Richard Miller responds
"I prefer not to answer that question, because I’m not a doctor and I’d never, ever wish to recommend drugs to someone else " (link to exact question and response here).
Given that rapamycin is by far the best performing drug in all the ITP studies, I’ll take that has a “YES” to him taking rapamycin. Your interpretation may differ.
@Krister_Kauppi contacted Richard recently to try to get him to tell us about his rapamycin use (Krister wanted to add him to his “Longevity experts using rapamycin” list) but Richard would not go on the record in anyway on this. I suspect biohacking is probably not something the NIA / NIH really wants to promote, so I can understand his rationale for staying quiet on this. He’s in a very visible position and I’m sure would get a lot of blowback / criticism from the higher ups in the NIA / FDA if he started talking about what he was using.
Mike, this is how science works. Tweak a single variable in a system to see what happens. Otherwise you can’t be sure what is causing the change.
If your food is hundreds of compounds, and you would like to test the changing interactions of hundreds of compounds to determine the ideal synergy of that many variables, you’ll be looking at $Billion studies, perhaps even Bajillion dollar studies.
Of course you’re right that our body’s many systems work in concert. But as Richard Miller, head of ITP says, people thought aging was due to such complex interactions across so many systems and types of cells that it would not be possible to affect aging meaningfully. Then they realized that it was possible, because a common process was underlying aging in the many systems.
This is why Rapamycin works. It affects a single variable that leads to cascades of outcomes.
So I appreciate your philosophy but I disagree with your proposed methodology in general. Sometimes “cocktails” are the answer, but until AI has human physiology mastered we’re going to be testing single compounds for a long time coming.
Okay. So they (tested substances) increase the part of the population that reaches the 90th percentile. By 9% or 14%.
According to Rapadmin,
Explaining the total lifespan increase
- The 9% or 14% more life basically translates to 25% more life once you’ve reached mid-life in terms of the number of additional years or months of life after you give the drug.
So 25% of the remainder of a human who is 60 years old is five years [20 times 25% = five years, or 85 years of age]. I use 20 as base, because most First World people live to be 80 (Italy 82, Germany 81, Spain 82), give or take a few years. 85 years is still modest compared to:
Loma Linda Adventists - Males 89, females 91
That is human data, versus mouse data.
I’m not saying that is not a benefit. But it is not any better than the benefit of diet and exercise. Now if the mice were also given optimal diets, or were exercised, or both, will there be greater increases? That is what would be interesting to me. For the moment, the improvement is underwhelming, compared to Loma Linda, Yuzurihara, Japan, and the so-called Blue Zones.
Pass me the goat cheese please. Make that Queso Cabrales from Asturias, Spain.
That was only the first ITP study (low dose). Here is an overview including the higher doses:
In the NIA ITP rapamycin lifespan studies, initiated with 9 month old mice orally via chow, the results were:
I don’t think I’ve seen any suggestions that the mTOR pathway is at all related to longevity pathways that might be stimulated by exercise, so I would think that optimal nutrition (if we can ever actually determine that in mice or humans) would be an additive effect for both species, over and and above any benefits from rapamycin, or rapamycin+ other longevity drugs like acarbose, etc.
Optimal for want of a better term. I should have said Loma Linda mimicking diet, or Sardinia-type, or Yuzurihara type diet.
With regard to mTOR and exercise, see below:
Conclusions
Exercise is a potent treatment for many diseases that reduce lifespan and quality of life. Many of the effects of exercise on quality of life likely reflect the ability of exercise to alter mTORC1 activity within diverse tissues.
Full article requires payment.
Here is mouse data versus the ITP mouse data.
Caloric Restriction's Effect on Lifespan Extension in Mice Depends on Timing.
The findings, obtained from hundreds of mice over four years, found that a reduced-calorie diet alone extended the animals’ lives by 10%. But feeding mice the diet only at nighttime, when mice are most active, extended life by 35%. That combination—a reduced-calorie diet plus a nighttime eating schedule—tacked on an extra nine months to the animals’ typical two-year median lifespan.