More good news on Irisin: Exercise-induced irisin has shown potential in maintaining muscle homeostasis, but critical research gaps persist
Protective effects of irisin in sarcopenia: a promising treatment
https://www.sciencedirect.com/science/article/pii/S2214031X26000458
Related Reading: Long-Acting Irisin With 30-Fold Longer Half-Life has Potent Anti-Inflammatory Effects
EXERCISE IN A BOTTLE: IRISIN EMERGES AS A MULTI-TARGETED BIOLOGIC TO HALT AGE-RELATED MUSCLE DECAY
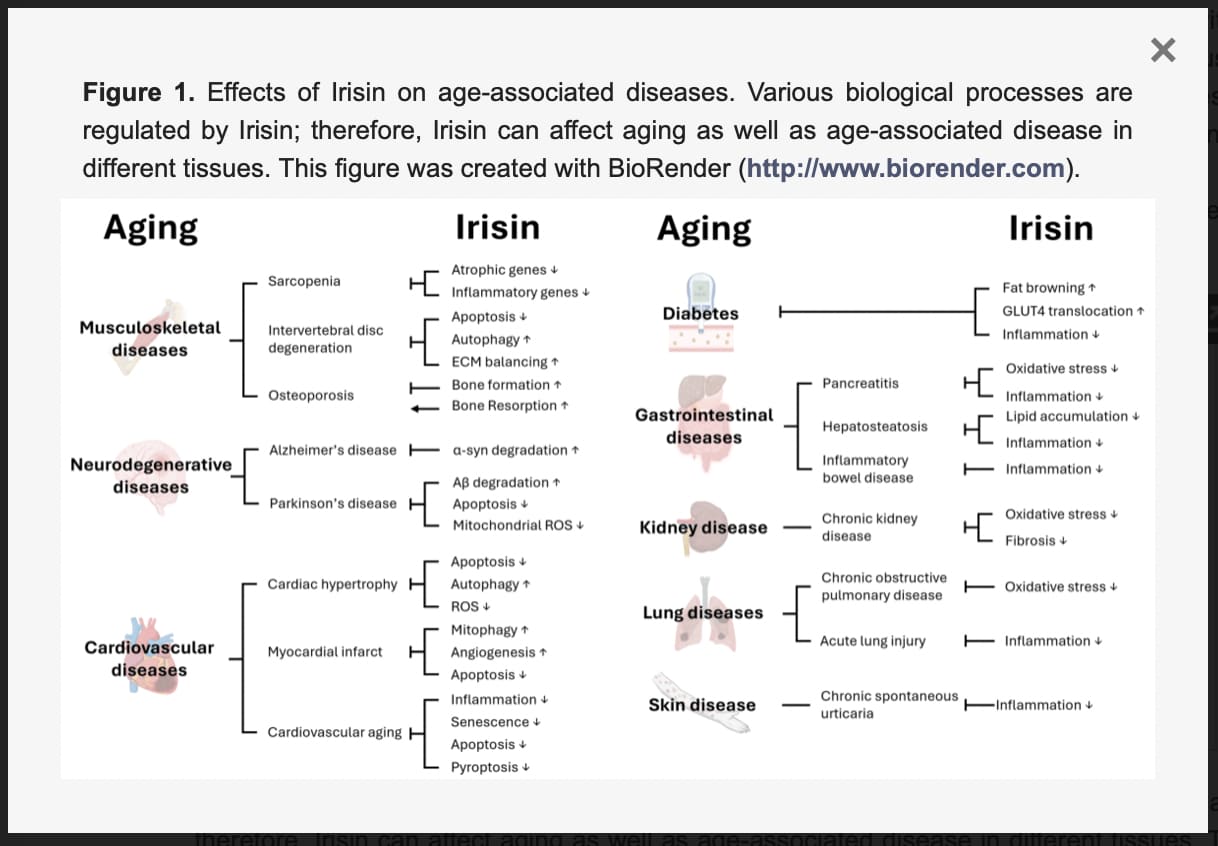
Sarcopenia—the relentless, age-dependent loss of skeletal muscle mass and strength—remains one of the most significant barriers to human longevity and healthspan. To date, no FDA-approved pharmacological interventions exist to halt this musculoskeletal decline. However, a comprehensive analysis originating from Beijing Jishuitan Hospital, Capital Medical University in China, published in the Journal of Orthopaedic Translation, positions the exercise-induced myokine irisin as a highly promising, multi-targeted therapeutic candidate for reversing sarcopenic muscle wasting.
The impact score of this journal is 7.8, evaluated against a typical high-end range of 0–60+ for top general science, therefore this is a Medium impact journal.
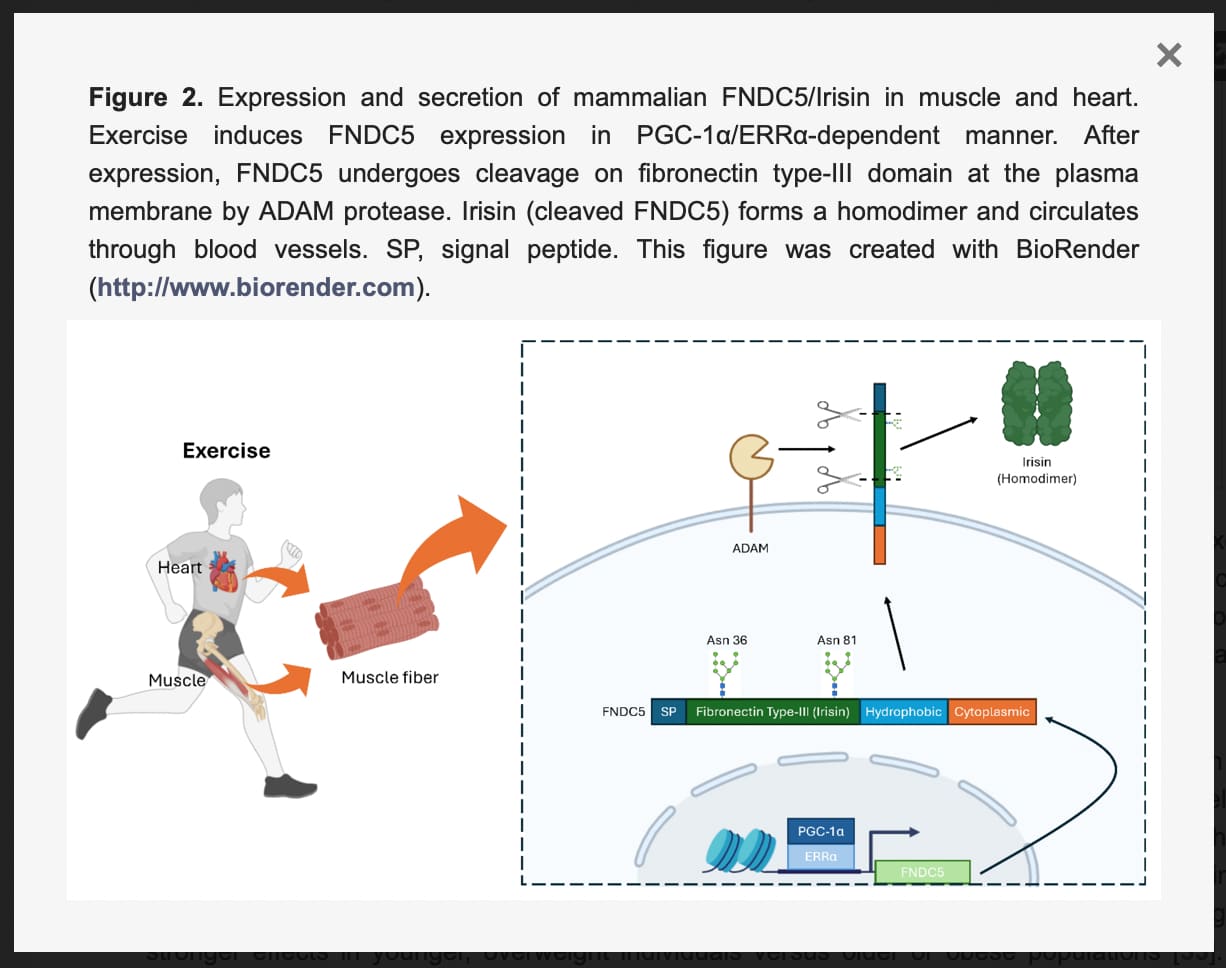
Originally identified in 2012 as a hormone responsible for browning white adipose tissue, irisin is a polypeptide cleaved from the FNDC5 precursor protein during muscle contraction and cold exposure. Circulating irisin levels plummet with age, correlating directly with diminished muscle mass, impaired grip strength, and an increased risk of osteosarcopenia (the deadly combination of muscle and bone loss). This review synthesizes mounting preclinical evidence demonstrating that restoring irisin directly rehabilitates the metabolic equilibrium of skeletal muscle. Rather than acting via a single pathway, irisin operates as a master regulator of protein turnover. Under normal aging, the ubiquitin-proteasome system (UPS) becomes hyperactive, aggressively shredding muscle proteins. Irisin suppresses this catabolic destruction by blocking FOXO transcription factors, while simultaneously boosting anabolism through the IGF-1/PI3K/AKT cascade. Furthermore, irisin actively rehabilitates the cellular energy grid. By upregulating PGC-1α, it drives mitochondrial biogenesis and induces selective mitophagy to clear out defective, ROS-leaking mitochondria.
Crucially, the researchers highlight irisin’s unique ability to inhibit distinct pathways of cellular execution. Beyond classical apoptosis, irisin shows significant potential in suppressing ferroptosis (iron-dependent lipid peroxidation) and pyroptosis (inflammation-induced cellular lysis)—two distinct mechanisms recently implicated in sarcopenic degeneration. It accomplishes this by fortifying the NRF2/GPX4 antioxidant axis and silencing the NLRP3 inflammasome.
Despite its immense physiological potential, translating irisin into a clinical pharmacological intervention faces severe pharmacokinetic hurdles. The most pressing limitation is its abysmal plasma half-life of roughly one hour, rendering standard injections practically useless for sustained therapy. While bioengineers are currently exploring albumin-fused long-acting variants and biodegradable silk hydrogel delivery systems to achieve sustained local release, irisin therapy remains confined to the preclinical laboratory. For now, intense mechanical loading and cold exposure remain the primary validated methods to endogenously pulse this longevity-promoting myokine.
Technical Biohacker Analysis
Study Design Specifications
-
Type: Comprehensive Literature Review and Conceptual Analysis.
Mechanistic Deep Dive
-
mTOR & IGF-1/PI3K/AKT Anabolic Axis: Irisin amplifies the IGF-1/PI3K/AKT signaling cascade. The activation of AKT stimulates mTORC1 (promoting protein synthesis via 4E-BP1 and S6 kinase) and inhibits glycogen synthase kinase 3 (GSK3), improving glucose influx and glycogen synthesis via GLUT4 translocation. [Confidence: High].
-
FOXO & The Ubiquitin-Proteasome System (UPS): By activating AKT, irisin phosphorylates FOXO transcription factors, sequestering them in the cytoplasm. This prevents FOXO nuclear translocation, effectively silencing the transcription of muscle-wasting E3 ubiquitin ligases (MuRF1 and MAFbx/atrogin-1). [Confidence: High].
-
Autophagy & Mitochondrial Dynamics: Irisin upregulates PGC-1α, acting as a direct driver of mitochondrial biogenesis. It also enhances autophagic flux—demonstrated by an elevated LC3-II ratio and accelerated p62 degradation—ensuring the clearance of damaged organelles without pushing the cell into excessive, destructive proteolysis. [Confidence: Medium].
-
Ferroptosis & Pyroptosis: The review postulates that irisin limits iron-dependent cell death by acting through the SIRT1/NRF2/GPX4 axis, and suppresses inflammation-mediated pyroptosis by inhibiting the NLRP3 inflammasome, preventing gasdermin D (GSDMD) cleavage. [Confidence: Low] (Note: These mechanisms are highly verified in renal, vascular, and cardiac models, but their direct role in sarcopenic skeletal muscle remains largely extrapolated).
-
Organ-Specific Aging Priorities: Targeted preservation of skeletal muscle myofibers and the mitochondrial network. Additionally, irisin drives satellite cell (Pax7+/MyoD+) activation and differentiation via the upregulation of the Mst1/2 and YAP pathways.
Novelty
This paper successfully consolidates irisin’s well-established metabolic roles with emerging, highly specific programmed cell death pathways (ferroptosis and pyroptosis). It shifts the biological narrative of irisin from a mere “fat-browning” metabolic agent to a master regulator of skeletal muscle proteostasis, offering a unified molecular theory for how exercise protects against muscle wasting.