Good results are being reported for stem cell therapies for skin and frailty:
Skin Trials:
3.2 SC preparations for facial skin aging and photoaging
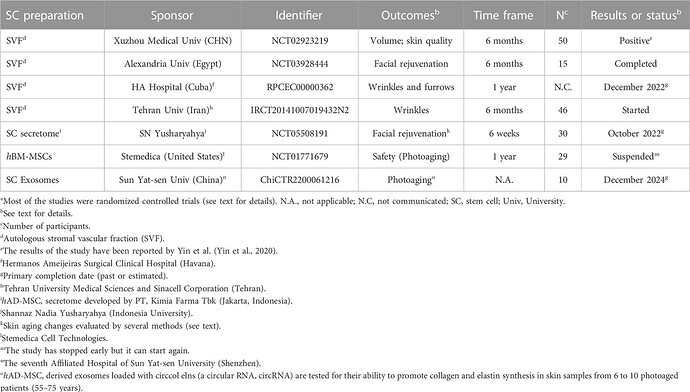
Skin aging is due to natural causes, as well as extrinsic factors (especially Sun exposure: Photoaging) (Zhang and Duan, 2018; Wong and Chew, 2021). Several SC preparations are investigated for facial skin aging and photoaging (Table 2).
Long-term natural aging is a slow process of dermal atrophy due to elastin and elastic fiber degradation, lower collagen production and lower hydration levels, leading to loss of elasticity and wrinkles (Zhang and Duan, 2018; Wong and Chew, 2021). Clinical trials with MSCs preparations for facial skin aging are evaluating their efficacy in regenerating normal, youthful skin (facial rejuvenation) (Figure 2).
Exposure to ultraviolet (UV) radiation facilitates skin aging (photoaging), characterized by the degradation of collagen and elastin, with deposition of collagen breakdown products and abnormal elastin fibers in the dermis (solar elastosis) (Huang and Chien, 2020). Clinical trials with MSCs preparations are evaluating their efficacy in restoring a normal skin.
3.2.1 Stromal vascular fraction (SVF) for facial skin aging
The “stromal vascular fraction” (SVF) is a preparation of autologous hAD-MSCs obtained by liposuction, followed by collagenase digestion, filtration, centrifugation and separation of the SVF (Coleman, 1994; Varghese and Mosahebi, 2017; Alexander, 2019; Khazaei et al., 2021; Surowiecka and Struzyna, 2022). The SVF represents about 10% of the adipose tissue volume, and is composed of hAD-MSCs, adipocyte progenitors, fibroblasts, endothelial cells, vascular smooth muscle cells, lymphocytes, and a variety of immune cells (T-cells and M2 macrophages). The efficacy and tolerability of SVF-enriched autologous fat grafting is currently being investigated in facial skin aging.
3.2.2.1 NCT02923219
NCT02923219 was an RCT comparing the efficacy of SVF-assisted autologous fat grafting (intervention) versus fat transfer alone (control) for facial volume restoration and improvement of skin quality (Table 2) (Yin et al., 2020). Fifty women (mean age: 35.4 years) participated in the study. At 6 months: 1) Whole face volumes (assessed by 3D scanner and Geomagic software) were significantly higher in the intervention group (77.6%) compared to the control group (56.2%, p < 0.001), 2) wrinkles and texture (assessed by VISIA detector) improved significantly more in the intervention group than in the control group, and 3) graft survival rate was significantly higher in the intervention group than in the control group.
3.2.2.2 NCT03928444
NCT03928444 is an RCT comparing intradermal autologous SVF injection on one side of the face versus saline injection on the other side (Table 2). Fifteen female participants with facial aging (35 years or older) are included in the study and will be followed for 6 months. The primary outcome is the degree of aesthetic improvement using the global aesthetic improvement GAIS 5-point scale (Savoia et al., 2014). The trial was completed, but the results were not posted to ClinicalTrials.gov.
3.2.2.3 RPCEC00000362
RPCEC00000362 is an RCT (single-blind) comparing the efficacy of SVF-enriched fat transfer versus conventional fat transfer (Table 2). Participants with facial aging (30–59 years of age) are included in the study and will be followed for 12 months. Outcomes include clinical evaluation and evolution of furrows and wrinkles. Trial completion date was expected for December 2022.
3.2.2.4 IRCT20141007019432N2
IRCT20141007019432N2 is a single-arm clinical trial, designed to investigate the efficacy of autologous SVF transplantation in reducing facial wrinkles (Table 2). Forty-six (46) participants with facial aging (35–65 years of age and with grade 2 to 4 wrinkle type) were included in the study and will be followed for 6 months. The primary outcome is biometric evaluation (with visioface and skin ultrasound) of the amount and extent of facial wrinkles. The trial completion date was not reported.
3.2.2 Soluble paracrine SC factors (secretome) for facial skin aging
3.2.2.1 NCT05508191
NCT05508191 is a single-blind RCT comparing two methods of AD-MSCs “secretome” administration for facial aging (fractional CO₂ laser treatment on one side of the face and microneedle treatment on the other half) (Table 2; Figure 1). The term “secretome” designates the soluble paracrine factors produced by SCs (Xia et al., 2019). Thirty female participants with facial aging (35–59 years) are included in the study and will be followed for 6 weeks. Primary outcomes are: 1) Skin aging changes evaluated by dermoscopy photoaging scale and by Janus-3Ⓡ skin analyzer, 2) skin capacitance evaluated by the CorneometerⓇ and 3) total water content in the stratum corneum of the skin. Primary completion date was expected for October 2022.
3.2.3 Facial photoaging
3.2.3.1 hBM-MSCs
NCT01771679 is a phase I/II safety trial to evaluate the safety and efficacy of a single intravenous injection of allogeneic (non-hematopoietic) hBM-MSCs for the treatment of facial photoaging in men and women 40–70 years of age (Table 2). Recruitment was suspended.
3.2.3.2 Exosomes
SCs secrete exosomes (40–120 nm extracellular vesicles), which contain cytokines, growth factors, messenger RNAs, and different non-coding RNAs, especially micro-RNAs (mi-RNAs) (Hamdan et al., 2021) (Figure 1).
3.2.3.2.1 ChiCTR2200061216. ChiCTR2200061216 investigates if hAD-MSC derived exosomes loaded with circcol elns (a circular RNA, circRNA) can promote collagen and elastin synthesis in skin samples from 6 to 10 photoaged patients (55–75 years). The study compares samples of facial skin tissue (part exposed to light), with skin tissue of the hip or upper arm (part protected from light) (Table 2). The study is open for recruitment. Completion is expected for December 2024.
3.3 Other SC therapies for aging
3.3.1 NT-020
NT-020 (NutraStem®) is a patented nutraceutical formulation containing green tea extract, blueberry extract, carnosine, and vitamin D3 (EurekAlert, 2008). In 2006, Bickford et al. (2006) reported that these agents (as well as catechin) synergistically stimulated the in vitro proliferation of human bone marrow and human CD34+ and CD133+ cells. CD34+ are often used clinically to quantify H-SC numbers in H-SC transplantation (Remberger et al., 2020). CD133 is a well-characterized biomarker of normal and cancer SCs (Barzegar Behrooz et al., 2019).