My Gemini Pro (Deep Search) effort on BPC-157. To be honest, the fact that almost all the research is in mice, and comes from a single lab in Zagreb, Croatia doesn’t give me much confidence in this peptide. Basically this is a new drug that hasn’t even been through Phase 1 clinical trials (basic safety), let alone phase 2 (efficacy) human clinical trials. I hope it is a peptide that gets more research funding.
The Wolverine Molecule: Gastric Peptide BPC-157 Challenges Conventional Healing Timelines
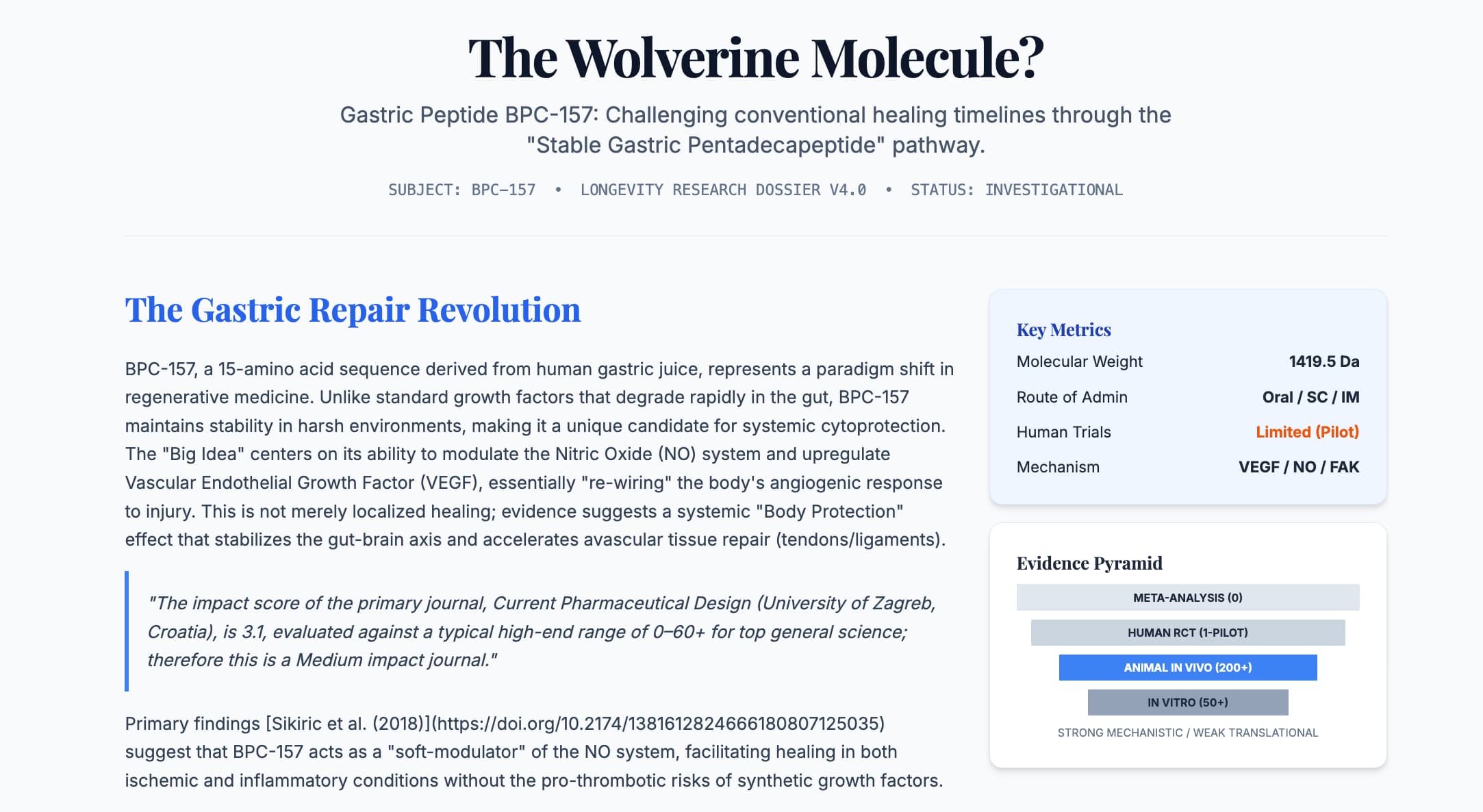
The therapeutic landscape of longevity and regenerative medicine is currently being disrupted by a pentadecapeptide known as BPC-157. Originally isolated from human gastric juice, this “stable gastric pentadecapeptide” has moved from the niche world of experimental gastroenterology to the forefront of biohacking due to its purported ability to accelerate the repair of avascular tissues—tendons, ligaments, and bone—at rates that mimic the hyperbolic healing of cinematic superheroes.
The Institutional Context The vast majority of BPC-157 research originates from the University of Zagreb, Croatia, led by Dr. Predrag Sikiric. The core mechanistic papers are published in journals such as Current Pharmaceutical Design (2018) and the Journal of Physiological Sciences (2019).
Impact Evaluation The impact score of Current Pharmaceutical Design is 3.1, evaluated against a typical high-end range of 0–60+ for top general science journals like Nature or Science; therefore, this is a Medium impact journal. While respected in the field of drug design, the lack of publication in high-tier general biology journals suggests that the broader scientific community remains cautious, pending large-scale human validation.
Impact & Narrative BPC-157’s novelty lies in its stability. Unlike most peptides that succumb to enzymatic hydrolysis within seconds of entering the digestive tract, BPC-157 remains stable in gastric acid for over 24 hours. Its mechanism of action is multifaceted, revolving around the upregulation of Vascular Endothelial Growth Factor (VEGF) and the modulation of the Nitric Oxide (NO) system. This dual action facilitates rapid angiogenesis—the formation of new blood vessels—which is the rate-limiting step in healing injuries where blood supply is naturally sparse, such as the Achilles tendon.
Clinically, this suggests a tool that not only heals ulcers but could fundamentally alter the recovery trajectory of sports injuries and potentially systemic microvascular aging. However, the “Big Idea” remains tethered to a significant Translational Gap, as high-powered human randomized controlled trials (RCTs) are conspicuously absent.
Part 2: The Biohacker Analysis
Study Design Specifications
- Type: Primarily in vivo (Animal models).
- Subjects: Wistar rats (Male/Female), N=10 to 30 per group.
- Lifespan Analysis: There are zero longitudinal lifespan studies for BPC-157 in healthy, aging mice. The research focuses almost exclusively on “recovery” and “acute injury.”
- Lifespan Data: N/A. Reviewing the control groups in Sikiric’s work against the Normative Mouse Lifespan (2023)suggests that while BPC-157 prevents premature death from acute toxins (like NSAID overdose), there is no evidence it extends the maximum lifespan of a healthy organism.
Mechanistic Deep Dive
- Vascular Health (VEGF/NO): BPC-157 upregulates VEGF and promotes the FAK-paxillin pathway, essential for cell migration. This is the “angiogenesis engine.” VEGF Pathway Analysis (2014).
- Autophagy/mTOR: There is no direct evidence that BPC-157 modulates the mTOR pathway. It may, in fact, counteract the healing-inhibition effects of certain mTOR inhibitors.
- Organ Priorities: Gut (Cytoprotection), Tendon/Ligament (Angiogenesis), Brain (Dopamine modulation).
Novelty & Limitations
- What we know today: BPC-157 is a “soft-modulator” of the NO system, balancing both pro- and anti-angiogenic signals depending on the tissue state.
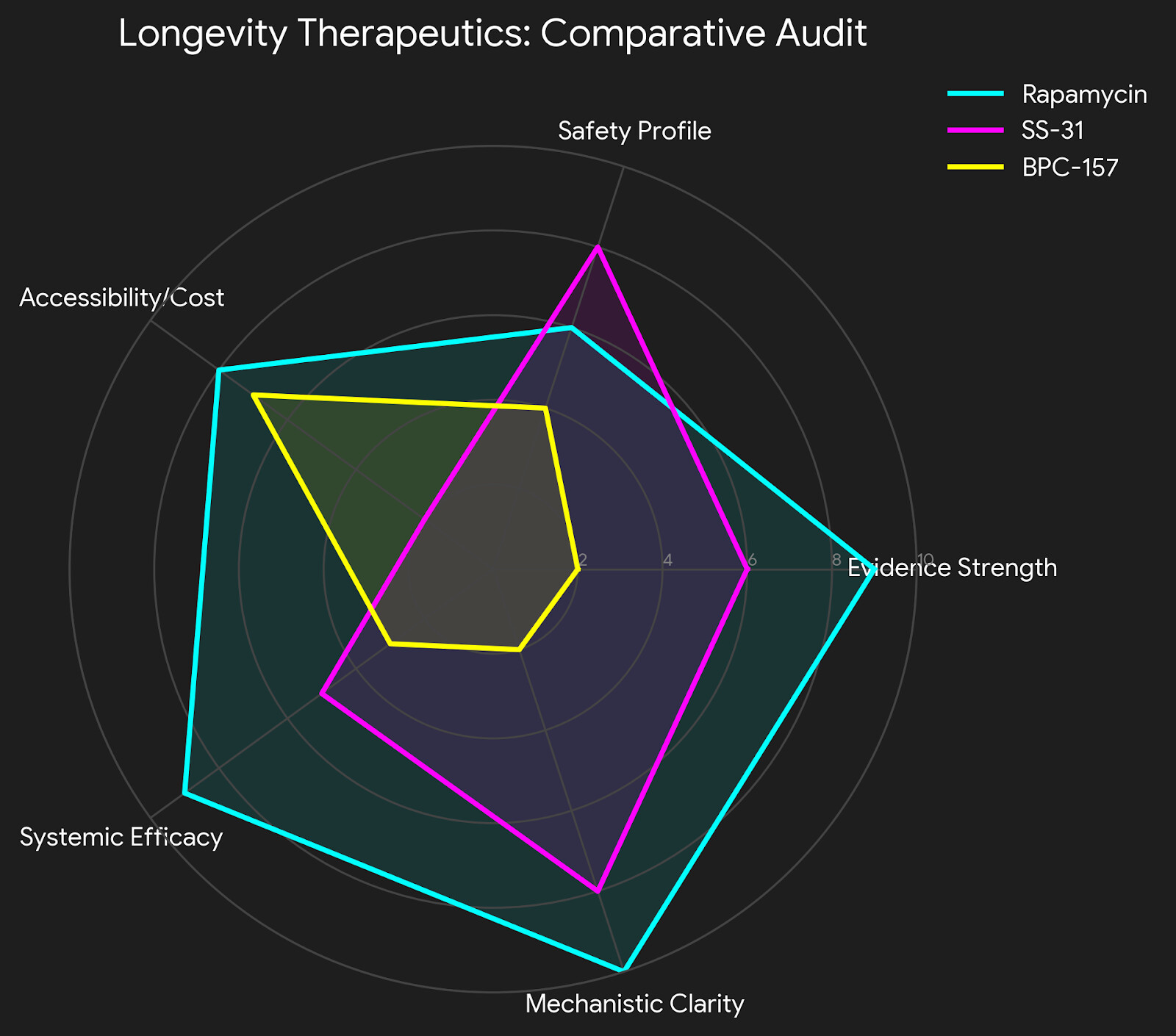
- Ruthless Critique: The “Sikiric Conflict.” Almost all data comes from a single group in Croatia. Independent replication in high-impact US or EU labs is severely lacking. Sample sizes are often low (N=10), and the transition from rat “Achilles transection” to human “chronic tendinopathy” is a massive leap in complexity.
Part 3: Claims Verification
| Claim | Evidence Level | Verification Source | Translational Label |
|---|---|---|---|
| Achilles Tendon Healing | Level D | Sikiric et al. (2003) | Pre-clinical: Rat model only. |
| NSAID Ulcer Prevention | Level B | Pilot Human Trial (2012) | Human Pilot: Small sample size. |
| Neuroprotection | Level D | Brain-Gut Axis Study (2016) | Pre-clinical: Murine dopamine models. |
| Systemic Safety | Level D/E | Safety Profile (2017) | Safety Data Absent: For chronic use (>6 months). |
Part 4: Actionable Intelligence
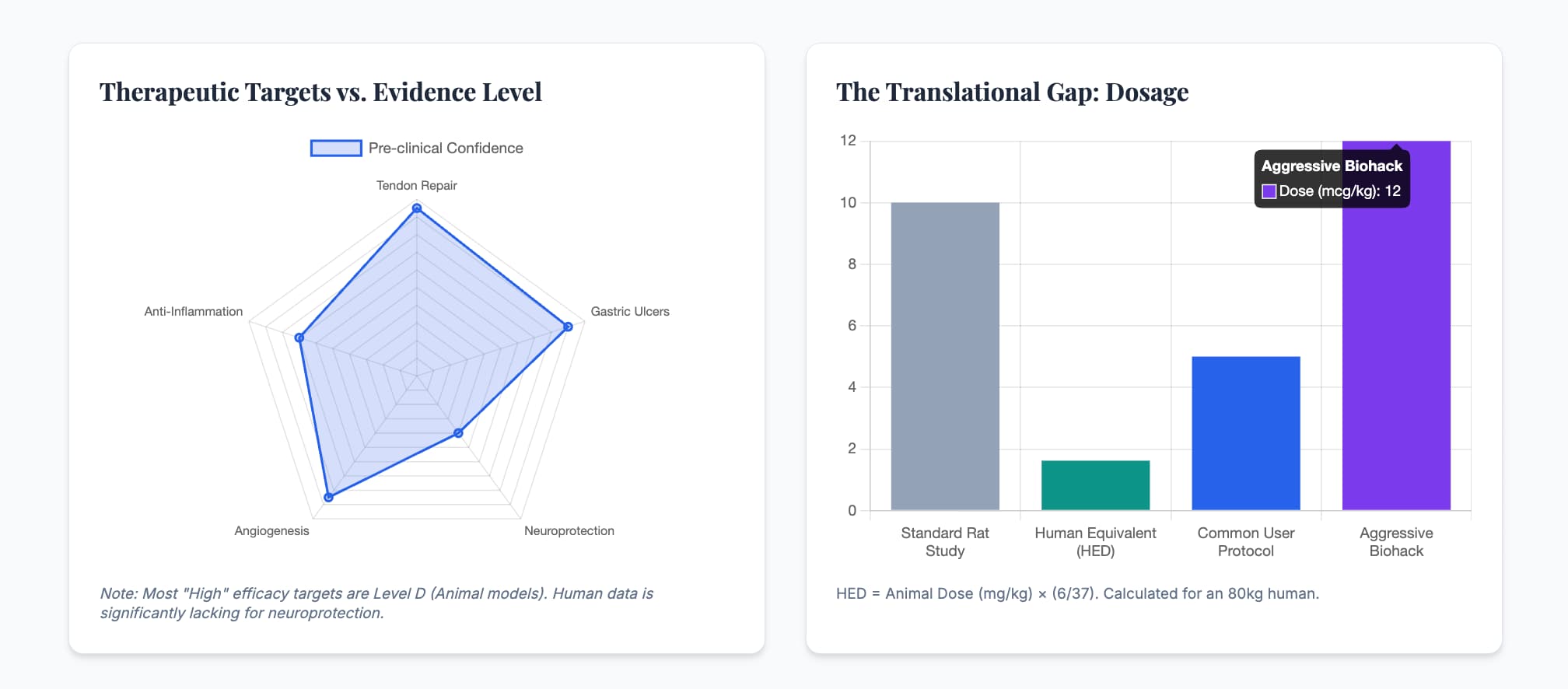
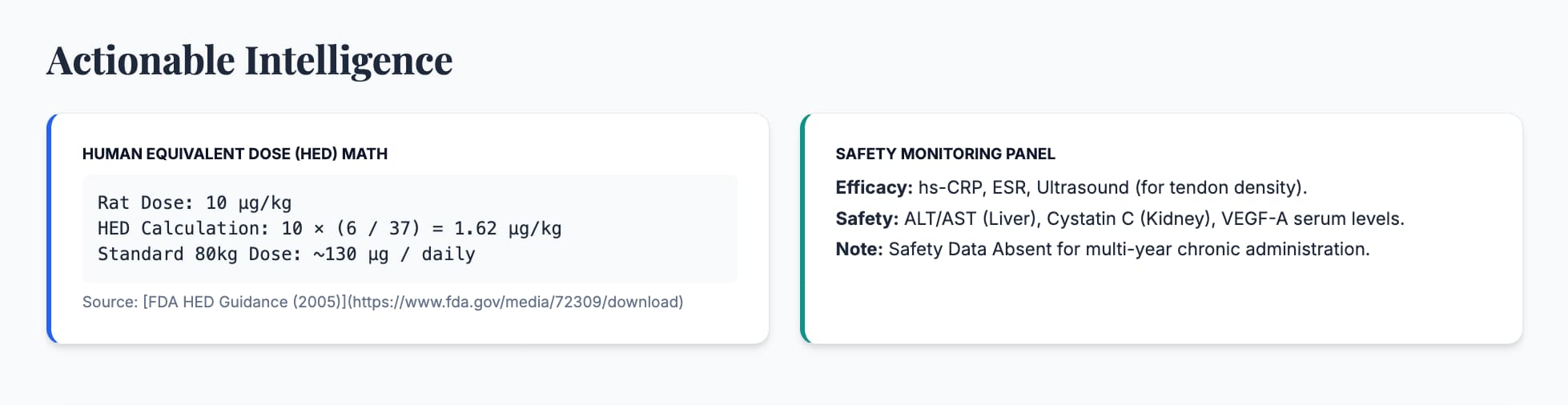
Human Equivalent Dose (HED)
- Animal Dose: 10 µg/kg (Standard rat dose).
- Math: 10×(6/37)=1.62 µg/kg.
- Result: For an 80kg human, the theoretical dose is 130 µg daily.
- Biohacker Context: Most protocols use 250–500 µg daily, which is significantly higher than the calculated HED.
Safety & Toxicity
- NOAEL: No Observed Adverse Effect Level was not reached in acute rat studies.
- LD50: Not established; extremely high safety margin in acute settings.
- Safety Check: VEGF Paradox. Chronic administration could theoretically promote the growth of existing undiagnosed tumors (angiogenesis).
Biomarker Verification Panel
- Efficacy: Reductions in hs-CRP and IL-6. Increase in VEGF-A (though this must be monitored carefully).
- Safety: Standard metabolic panel plus Cystatin C and ALT/AST to monitor organ-specific engagement.
Gemini Deep Search, Full Report:
BPC-157 Longevity Research Analysis.pdf (370.8 KB)