Follow-on Prompt:
A somewhat common therapeutic in the longevity field is to supplement with Growth Hormone injections. Would this potentially provide some of the benefits of raised IGF-1 seen in this research study?
Gemini 3 Pro Response:
Mechanistically, yes. Exogenous human growth hormone (hGH) administration will predictably elevate systemic IGF-1 levels and could theoretically provide the neurovascular coupling (NVC) benefits observed in the parabiosis study. However, deploying this strategy specifically for lifespan extension presents a profound biological contradiction known as the “Longevity Paradox.”
Here is the breakdown of the mechanics, the trade-offs, and the translational viability of this approach.
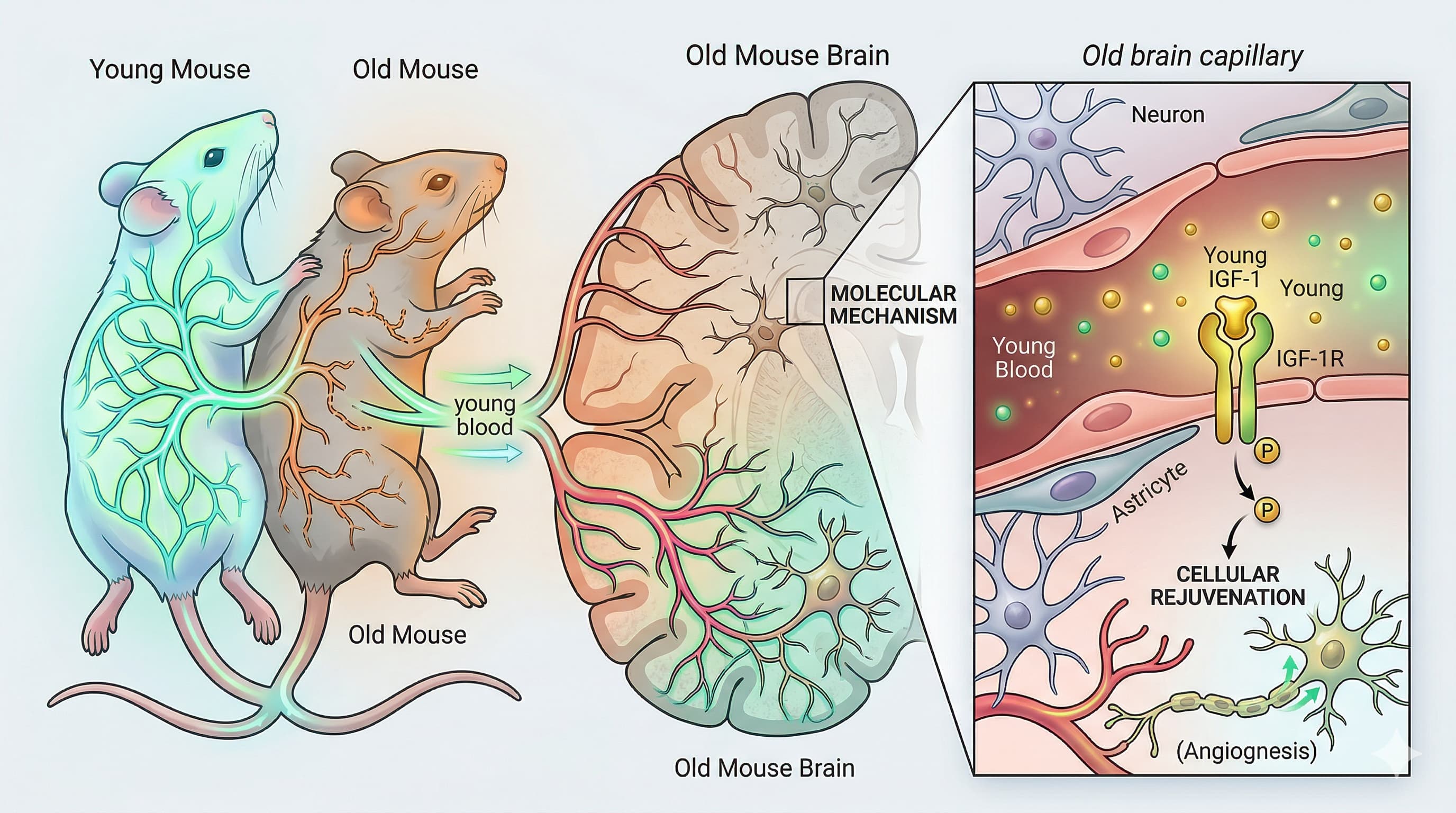
Mechanistic Plausibility: The GH/IGF-1 Relay
When you inject exogenous hGH, it binds to growth hormone receptors (GHR) primarily in the liver, activating the JAK2/STAT5 signaling cascade. This triggers the hepatic transcription and systemic release of IGF-1 into the bloodstream.
Once in circulation, this hepatically derived IGF-1 would act identically to the “young blood” factors described in the study: it binds to the endothelial IGF-1 receptors (IGF-1R) lining the cerebral microvasculature, activating the PI3K/Akt pathway, phosphorylating eNOS, and increasing the nitric oxide bioavailability required for optimal neurovascular coupling.
Therefore, from a strict cerebrovascular hemodynamics perspective, hGH injections would likely mimic the vascular rejuvenation seen in the transgenic models.
The Longevity Paradox: Healthspan vs. Lifespan
While elevated IGF-1 may acutely rescue vascular and muscular phenotypes, systemic elevation of the GH/IGF-1 axis is fundamentally antagonistic to maximum lifespan extension.
-
Lifespan Inverse Correlation: The most robust genetic models for extended mammalian longevity (e.g., Ames dwarf mice, Laron syndrome in humans) are characterized by severe deficiencies in the GH/IGF-1 axis. Downregulating this axis delays cellular senescence, enhances autophagy, and provides extreme resistance to age-related pathologies, particularly cancer.
-
mTOR Activation: IGF-1 is a potent upstream activator of the mechanistic target of rapamycin (mTOR). Systemically driving mTOR promotes cellular proliferation and blocks autophagy. If you are utilizing an mTOR inhibitor like rapamycin to extend lifespan, injecting GH directly counteracts that mechanism.
-
Metabolic Penalty: Chronic hGH administration frequently induces hyperinsulinemia and peripheral insulin resistance. Elevated insulin independently drives oncogenesis and vascular endothelial damage, potentially negating the localized cerebrovascular benefits of the elevated IGF-1.
Actionable Intelligence and Knowledge Gaps
The critical knowledge gap remains whether the localized cognitive and vascular benefits of exogenous hGH outweigh its systemic mortality risks in a healthy, aging human population. Current clinical consensus strongly advises against hGH replacement for anti-aging purposes due to the elevated risk of neoplasms, fluid retention, and glucose intolerance.
If the primary objective is to increase lifespan while maintaining cognitive resilience, the strategic imperative is to decouple the systemic oncogenic risks of IGF-1 from its localized vascular benefits. Practical, non-hormonal interventions to maintain downstream eNOS activation and endothelial nitric oxide production—such as phosphodiesterase type 5 (PDE5) inhibitors, L-citrulline supplementation, or maintaining high insulin sensitivity via SGLT2 inhibitors and acarbose—offer a superior risk-to-reward ratio.
Given the inverse relationship between systemic IGF-1 levels and maximum lifespan, are you currently prioritizing interventions that enhance acute cerebrovascular healthspan or those strictly aimed at extending maximum chronological lifespan?
The Strategic FAQ
1. Why advocate for restoring IGF-1 to rescue vascular aging when genetic reduction of the GH/IGF-1 axis is the most robust method for extending maximum mammalian lifespan? The study highlights a tissue-specific trade-off. While systemic suppression of IGF-1 extends total lifespan by delaying cancer and cellular senescence, the brain’s microvasculature relies on IGF-1 for nitric oxide synthesis and endothelial maintenance. The researchers are identifying a mechanism of decline, not necessarily proposing systemic IGF-1 as a net-positive anti-aging therapy.
2. How do you differentiate the vascular benefits of local endothelial IGF-1 signaling from the systemic oncogenic risks of elevated circulating IGF-1? You cannot effectively separate them with current systemic peptide therapies. The ultimate translational goal resulting from this data is not to administer IGF-1, but to discover downstream small molecules that selectively activate endothelial eNOS or mimic the local vascular effects of IGF-1R activation without stimulating systemic cellular proliferation.
3. Parabiosis shares entire organ systems. How can we be certain the NVC rescue is primarily endocrine (IGF-1) rather than the young liver and kidneys clearing pro-aging SASP factors from the old mouse? We cannot be entirely certain. The study design proves that removing IGF-1 blunts the rescue, meaning IGF-1 is necessary, but it does not prove IGF-1 is sufficient on its own. The partial rescue observed in the knockout models strongly suggests that the clearance of aged factors (like β2-microglobulin) by young organs plays a concurrent role.
4. Did the study assess if the restoration of NVC hemodynamics translated to actual cognitive improvements in the aged mice? No. The paper strictly utilized laser speckle contrast imaging for hemodynamic proxy data. It lacks behavioral assays (e.g., Morris water maze, novel object recognition) to confirm that the increased cerebral blood flow altered the clinical cognitive phenotype.
5. The study exclusively used male mice. Given the profound sexual dimorphism in the GH/IGF-1 axis, how likely is it that these vascular rejuvenation effects apply to females? Translational uncertainty here is high. Female mice frequently exhibit divergent responses to IGF-1 modulation, often showing different lifespan and metabolic outcomes compared to males when the somatotropic axis is altered. Female validation is a mandatory next step.
6. By what specific temporal mechanism does endothelial IGF-1R activation enhance neurovascular coupling? It functions on two timelines: Acutely, IGF-1R activates the PI3K/Akt pathway, leading to rapid phosphorylation of eNOS and immediate vasodilation. Chronically, it stabilizes mitochondrial function, suppresses reactive oxygen species (ROS), and preserves capillary density, preventing structural microvascular rarefaction.
7. Since endothelial IGF-1R deficient parabionts still showed partial NVC improvement, what are the primary redundant factors compensating for the lack of IGF-1? The systemic milieu contains multiple anti-geronic factors. Current candidates for redundant vascular rescue include GDF11, TIMP2, circulating NAD+/eNAMPT enclosed in extracellular vesicles, and oxytocin, all of which have been shown to independently exert rejuvenating effects on aging tissues.
8. How does the pro-inflammatory milieu of an aged brain alter the expression of endothelial IGF receptors, potentially causing IGF-1 resistance? Chronic low-grade inflammation (inflammaging) and SASP factors downregulate the expression of membrane receptors and induce intracellular insulin/IGF-1 resistance via the activation of serine kinases (like JNK and IKK$\beta$), which disrupt the insulin receptor substrate (IRS) cascade. Simply adding more IGF-1 might not overcome severe receptor-level resistance.
9. If a clinician wanted to target this specific vascular pathway safely, are there effectors that can be activated without using systemic IGF-1? Yes. Bypassing the IGF-1 receptor to directly support endothelial nitric oxide production is safer and more feasible. This can be achieved via dietary nitrates, L-citrulline supplementation, or pharmacological PDE5 inhibitors, which sustain the cGMP signaling downstream of NO.