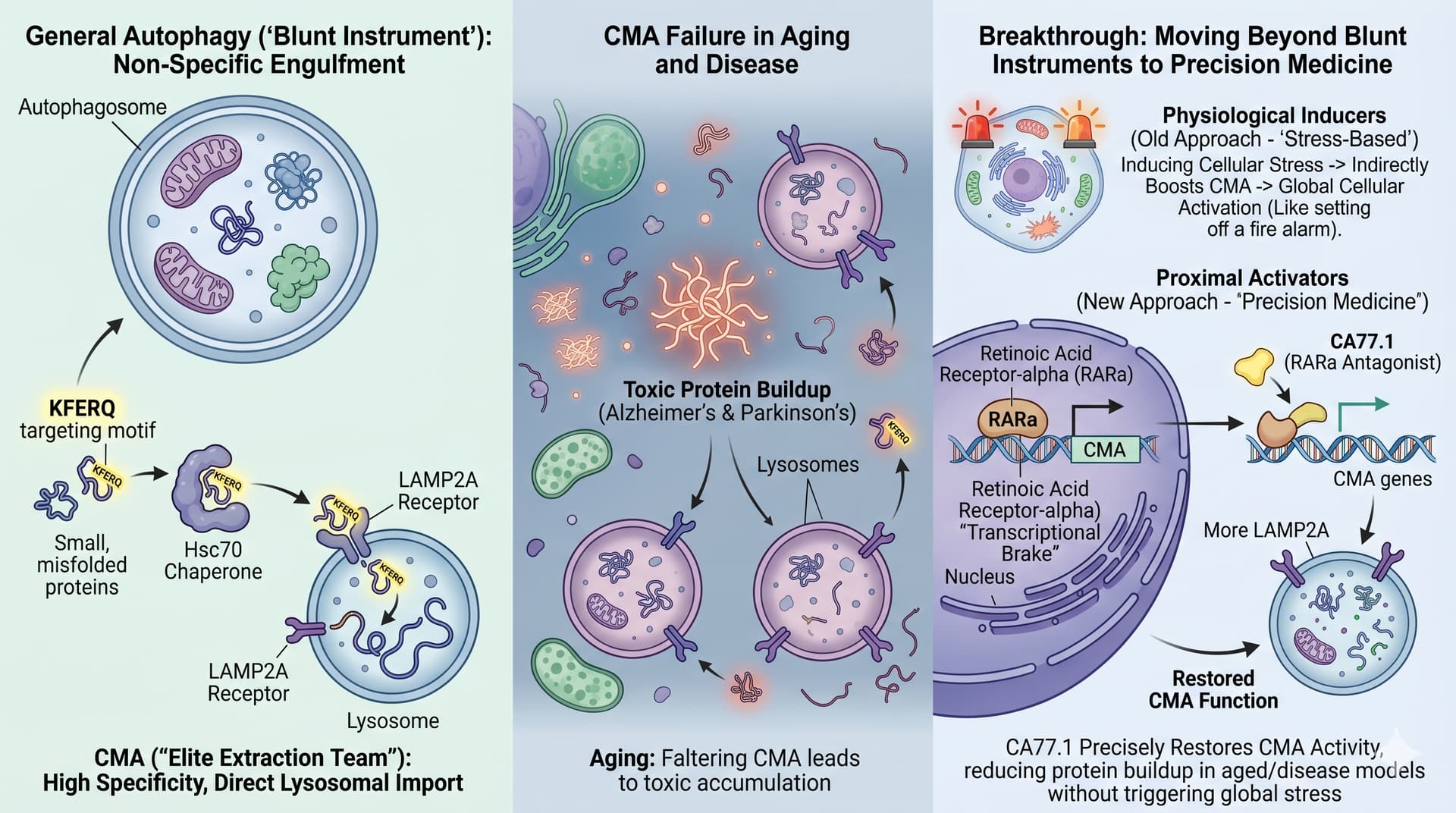
The biological “garbage disposal” of the cell just received a major upgrade in pharmacological theory. For decades, Chaperone-Mediated Autophagy (CMA) was the overlooked sibling of macroautophagy—the bulk recycling process popularized by intermittent fasting. However, a new review published in Trends in Pharmacological Sciences argues that CMA is not just a secondary stress response, but a surgical tool for maintaining proteostasis that is finally becoming “pharmacologically tractable”.
Unlike other forms of autophagy that swallow large chunks of cellular material, CMA is an elite, protein-specific extraction team. It identifies individual misfolded or damaged proteins using a “KFERQ” targeting motif and shuffles them directly into the lysosome for destruction. As we age, this system falters, leading to the toxic protein buildup characteristic of Alzheimer’s, Parkinson’s, and metabolic collapse.
The breakthrough lies in moving beyond “blunt instruments”. Early attempts to boost CMA relied on inducing cellular stress—essentially trying to clean a house by setting off the fire alarm. This review introduces a three-tier classification system to guide drug discovery: physiological inducers (stress-based), permissive potentiators (signaling-based), and proximal activators (transcriptional-based).
The most promising frontier is the “proximal activator” class, specifically Retinoic Acid Receptor-alpha (RARa) antagonists. Researchers have identified that RARa acts as a “transcriptional brake” on the CMA machinery. By neutralizing this brake with synthetic compounds like CA77.1, scientists have successfully restored CMA activity in aged tissues and disease models without triggering global stress or interfering with other cellular pathways. This marks a transition from empirical “biohacking” toward high-precision medicine aimed at extending the human healthspan.
Actionable Insights
Current evidence suggests that CMA can be modulated through three primary avenues:
-
Calorie Restriction (CR) and Mimetics : CR remains the most robust physiological inducer of CMA in vivo. Compounds like Spermidine and Hydroxycitrate act as CR-mimetics by altering the acetylation of chaperone networks (HSC70), effectively lowering the threshold for protein recognition and degradation.
-
Repurposed Compounds : Common longevity drugs like Metformin and Trametinib have been identified as permissive potentiators. Metformin facilitates CMA via the TAK1/IKK pathway, while Trametinib (a MEK inhibitor) stabilizes the lysosomal receptor LAMP-2A, though both have significant pleiotropic effects beyond CMA.
-
The RARa Frontier : While not yet available for general use, RARa antagonists (e.g., QX77, CA77.1) represent the most “pathway-selective” approach to increasing cellular cleaning capacity. These compounds specifically increase the expression of LAMP-2A , the rate-limiting receptor for CMA, providing a potential future pharmaceutical route to combat age-related proteotoxicity.
Context
- Paper: Pharmacological strategies targeting chaperone-mediated autophagy
- Institutions : Karolinska Institutet (Sweden), University of New Mexico Health Sciences Center (USA).
- Journal : Trends in Pharmacological Sciences (TIPS).
- Impact Evaluation : The CiteScore for this journal is 24.5 (2024), evaluated against a typical high-end range of 0–60+ for top general science; therefore, this is a High impact journal in the field of pharmacology