On this same topic:
https://www.nationalgeographic.com/magazine/article/how-old-face-disease
How old are you, really? The answer is written on your face.
By Connie Chang
National Geographic
January 31, 2023
A picture is worth a thousand words. But when that picture is a high-precision 3D image of the face, it might be worth a thousand blood tests as well. That’s because those full cheeks and under-eye bags are not just unsightly harbingers of age, but also a reflection of our health.
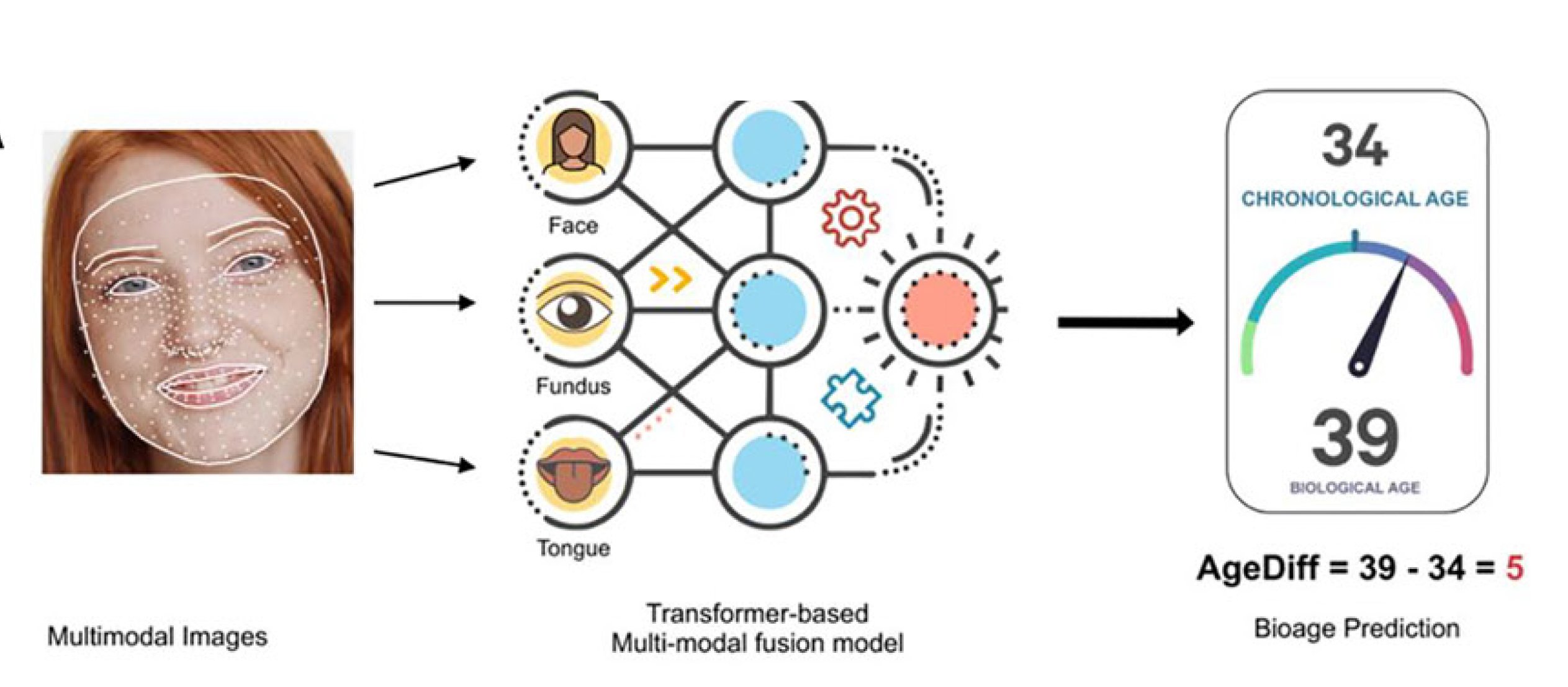
Scientists have known for decades that chronological age—a number that can be gleaned from our driver’s license-–doesn’t tell the whole story. Our so-called biological age, influenced by everything from our environment to diet and exercise habits, reflects the health of our cells and organs and can differ from chronological age by years. But unlike tracking the time we’ve been alive, biological age is more slippery to pinpoint. Now Jing-Dong (Jackie) Han and colleagues have developed an AI-driven process, dubbed a facial aging clock, that takes a 3D image of a person’s face and calculates their biological age.
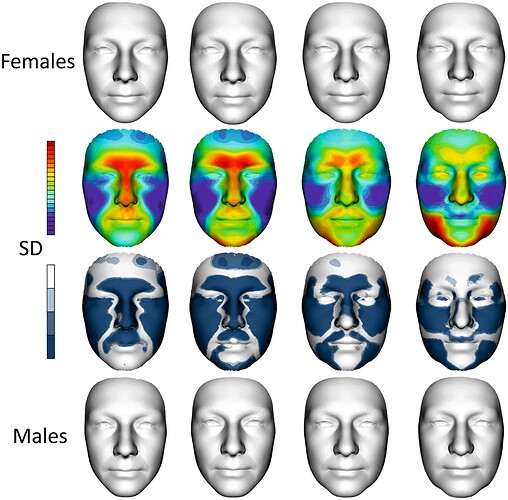
Inspired by a centuries-old Chinese practice, in which practitioners divine a person’s health by “reading” their face, Han, a computational biologist at Peking University, and her team constructed their clock by analyzing 3D facial images of approximately 5,000 residents of Jidong, China. The researchers created two AI-derived clocks—one that predicts chronological age and another that predicts biological age. These facial aging clocks track the changes our visages undergo with time: The corners of the eyes droop, the nose widens, the jowls sag, and the distance between the nose and mouth increases. And certain facial characteristics are known to align with certain ailments. Systemic inflammation, for example, shows up in sagging skin.
According to Andre Esteva, the founder and CEO of a medical AI start-up in Los Altos, California, Han’s work has the potential to upend preventative medicine: “If you could take a photo and get back your biological age, that could really influence your lifestyle.” With this tool, physicians could also track and manage the care of patients undergoing onerous treatments known to prematurely age people, such as chemotherapy. And it has potential to aid research into aging too.
https://www.andreesteva.com/
“We’ve been getting so many requests from companies that want [our tool] to assess their supplements or anti-aging drugs’ efficacy,” Han says.
AI models require examples where the right answer, or “ground truth,” is already known to learn how to see it in new data—a face paired with the subject’s age, for instance. So, a potential sticking point arose: There is no gold standard for biological age. “The concept of biological age is more of an umbrella term for all the multisystemic things that occur with age,” explains Christopher Bell, who studies the relationship between age and chronic diseases at the University of London. From the shortening of our telomeres—the caps that keep our chromosomes from degrading—to the winding down of our mitochondria to the weakening of our immune system, it’s a challenge to choose just one aging marker.
The first clocks to measure aging were based on changes in patterns of methyl groups—chemical tags added to DNA that switch genes on and off. This DNA methylation machinery that regulates gene activity deteriorates over time. The pattern of this deterioration—which areas in our genome are affected—can tell us how fast our cells and tissues are aging. Other clocks assessing age are based on protein distribution in the blood or the number of times stem cells have divided.
Facial aging clock
Han decided in 2016 that she wanted to use another measure—perceived age, or how old you appear to others. She was inspired by a 2009 study in which volunteers assessed the age and health of twins in a photograph, followed by a medical evaluation of the subjects seven years later. Those researchers found that the older-looking of a twin pair was more frail, cognitively impaired, and likely to have died compared to their younger-looking sibling. Clearly, Han thought, biological age, as proxied by appearance, is strongly correlated with health. Her group had already incorporated 3D images of faces in research published in 2015, in which they found that facial features could predict chronological age in a statistical model developed using 300 subjects from Beijing.
At the time, she had been investigating markers, or signatures, in the blood and ascertaining their relationship to age when she noticed that the Chinese Academy of Sciences-Max Planck Society Partner Institute for Computational Biology, where she then worked, had a 3D facial imager. She thought, since they had this imager, they should take it with them when they collect blood samples and use it to compare what the face reveals about aging versus what the blood markers reveal.
By 2016, she had access to a larger cohort, approximately 5,000 people from the city of Jidong, and more powerful AI techniques. With the data-crunching prowess of AI, Han could replicate human perception of how biological age manifests in someone’s face. To minimize fluctuations from any one observation, in Han’s new study, each subject’s biological age was assessed independently by five volunteers and served as the “ground truth” for training the AI.
The AI engine, which learned from and improved on these human observations, was startingly accurate. On average, its predictions deviated from actual ages by about three years for both chronological and perceived age models. In Han’s study, people who appeared more than three years older than the date on their birth certificate she dubbed fast-agers; while slow-agers seemed more than three years younger.
This difference between their predicted and actual ages can be correlated to various health parameters, Han says. And according to Han, these outliers teach us the most about how we age.
To explore the links between appearance, age, and the underlying factors that influence health—Han collected blood samples and surveyed the habits of the research subjects. For example, smoking, snoring, and high levels of total cholesterol in the blood were often characteristics of fast-agers whereas yogurt consumption, regular mealtimes, and greater bone mineral density were a common trait of slow-agers.
Middle age, when individuals are in their 40s and early 50s, seems to be a period when the differences between fast- and slow-agers become more noticeable. Some people might look quite old even though they’re just 40 while others are 55 but still look very young. According to Han, this variability means that interventions to adopt healthier habits could make a significant difference in this window of opportunity.
Han and her collaborators also constructed additional models to examine the underlying molecular mechanisms behind aging and match them to facial features of fast-agers through the AI-derived clocks. For example, they developed clocks which identified active genes in blood from an older-seeming individual, and matched them to the facial features of the same individual.
For those predictions that overlap, researchers can ascertain which genes (and therefore molecular pathways) are turned on for older faces compared with younger faces. For all clocks, “accelerated aging is highly associated with infections and inflammation,” which manifests in the face as a narrowing of the forehead as the skin sags, says Han. On the other hand, high LDL and total cholesterol levels in the blood translates to full cheeks and undereye pouches.
Applications of the technology
Aging research has become mainstream, but one of the field’s problems has always been: We don’t know exactly what biological age is, nor is there an objective way to quantitatively define it. But according to Ruibao Ren—a physician-scientist at the International Center for Aging and Cancer at Hainan Medical University in China—Han’s research, as well as ongoing work in other labs, has made great strides to narrow this information gap.
“Just being able to measure aging is already a big leap forward and Dr. Han’s technology—already in use at the hospital he works at in Hainan—will play an important role in aging studies,” Ren says. Pharmaceutical companies can employ the tool to assess drugs that promote longevity or slow down aging, for example. And as an oncologist, Ren hopes that with the facial clock, he can diagnose cancer patients while the disease is in its early stages. Since cancer is strongly correlated with age, if someone shows signs of accelerated aging, doctors can be more proactive with screening. “Aging is at the root of probably 80 percent of diseases,” Ren says.
Doctors could also incorporate biological age as one of the panel of tests included in your annual physical like cholesterol tests or blood pressure measurements. “It could be a wake-up call that asks: How are you doing with managing your health?” Ren says.
And unlike other clocks which require blood or tissue samples and expensive analysis to implement, the facial clock is comparatively economical and non-invasive, requiring the patient to sit for just one minute in front of a 3D imaging camera.
Future directions
Bell too sees great potential in the further study of aging clocks, especially as datasets become larger. The earliest epigenetic clocks, for example, looked at 1,500 potential methylation sites on our DNA, while the most recent clocks consider more than 900,000. But aging clocks’ real power, Bell says, is their potential to uncover novel insights into the aging process.
He highlights, for instance, how abnormal DNA methylation is a signature of cancer as well as aging. By understanding both scenarios, “we might identify novel therapeutic avenues to treat these diseases,” Bell says.
But he counsels caution in interpreting the results from various aging clocks. “At a population level, these clocks are capturing some aspects of aging.” On an individual level, however, there’s more work to be done. “If we measure someone’s particular epigenetic clock age, we can’t be as confident of how well that tracks in that individual or that changes over time represent reality.”
As for Han, her team continues to explore other clocks such as a transcriptome clock, which reflects DNA damage in the blood, and a single cell clock, based on information from one cell. The aim is to aggregate disparate measures—each clock showcases a different aspect of aging—into a composite clock. They also continue to refine their facial aging clock—a version applicable to all ethnic groups will be published soon.
What is Han’s ultimate goal? “Well, I’m telling my colleagues that I will try to make them live at least five years longer—and look at least five years younger,” she laughs.