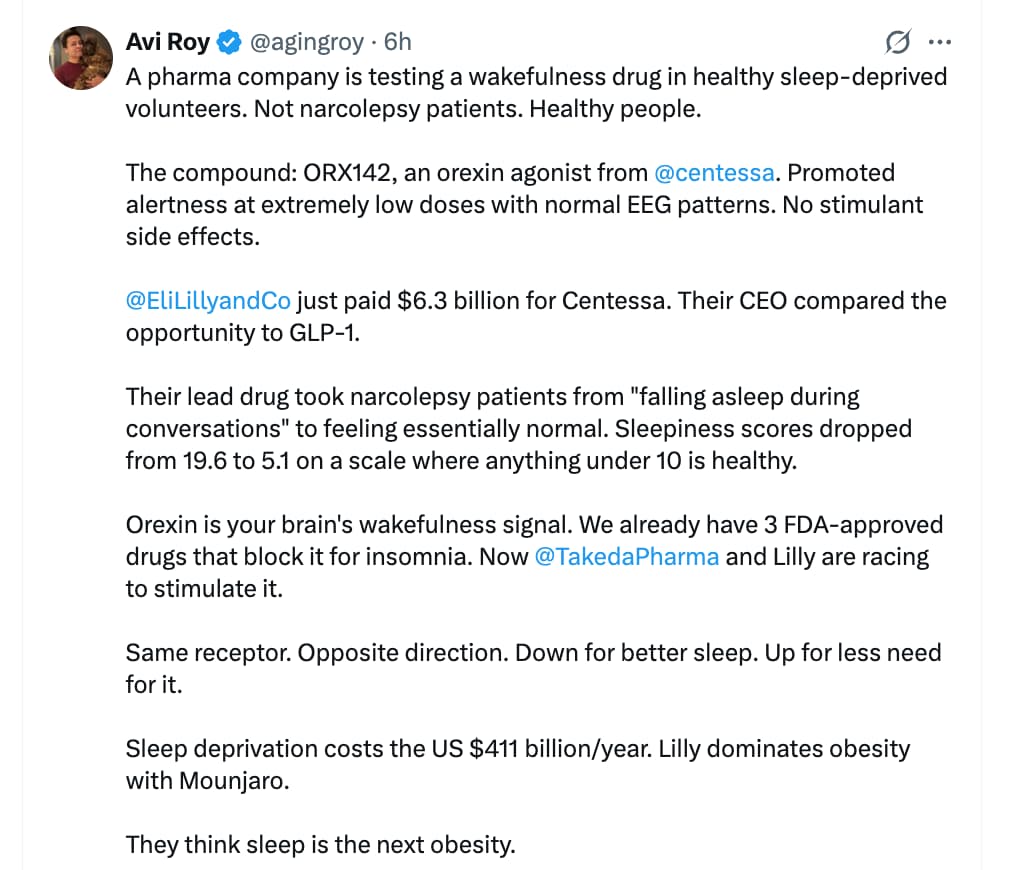
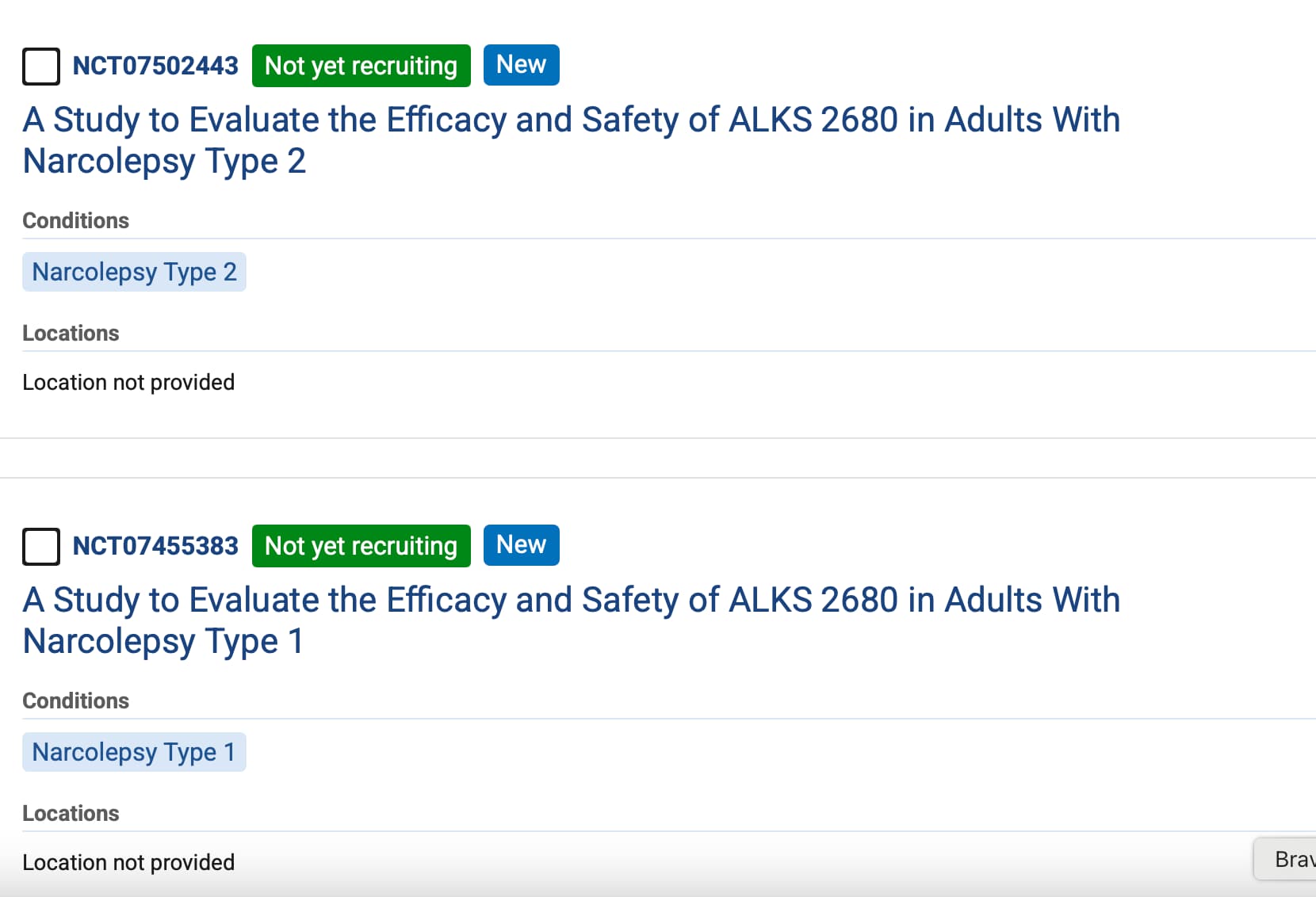
That depends on the dosage range. Do we know what it is?
I have purchased products from Limitless Biotech in the past with no ill effects.
Perplexity Pro (Deep Research) says:
Limitless Biotech (also known and branded as Limitless Life Nootropics, at limitlesslifenootropics.com) is a U.S.‑based online retailer and chemical‑supply company focused on research‑grade peptides, nootropics, and longevity‑adjacent compounds, marketed primarily for “research use only” rather than as FDA‑approved drugs. The company is headquartered in or near Pensacola, Florida, and operates under the trade name Limitless Life Nootropics, with products made and tested in the U.S. and third‑party‑verified for purity.
Founders and leadership
The company is widely described as being founded and led by Christopher (Chris) Mercer, who is shown on LinkedIn as the CEO of “www.limitlesslifenootropics.com” under the entity Live Limitless LLC in the Pensacola area. Public write‑ups and discussions about the brand also refer to him as the owner/CEO who has been involved in interviews (including a 2016 episode of Showtime’s Dark Net) describing how he sourced, repackaged, and sold nootropics from a home‑kitchen‑style setup before the company grew into its current “Limitless Biotech”‑style operation.
History and rebranding
The business started out as Limitless Life Nootropics, selling nootropics and cognitive‑enhancement‑style supplements, often via direct‑to‑consumer and affiliate channels.
Over time it expanded into research‑grade peptides and anti‑aging‑adjacent compounds, rebranding or dual‑branding itself as Limitless Biotech to emphasize its “research‑grade, USA‑made peptides with third‑party testing” angle.
It has positioned itself as a “leading” peptide supplier in the biohacking/anti‑aging community, often promoted by influencers such as Jay Campbell and Ben Greenfield, while also attracting criticism from some Reddit and forum users over quality claims, customer‑service issues, and prior “home‑kitchen” practices.
Limitless Biotech sits in a legally gray, high‑risk niche (research peptides and synthetic nootropics), and its business practices and history create enough red flags that regulators, platforms, and customers continue to scrutinize it despite third‑party testing claims and positive press/“awards.”
Why awards and testing coexist with scrutiny
Limitless has been promoted in niche fitness/peptide media as a “top peptides company,” with review sites and at least one industry press release naming it a leading or “best” peptide vendor, often citing its broad catalog and availability of CoAs. These endorsements come from commercial reviewers and partners, not regulators, so they can coexist with unresolved concerns about sourcing, payment methods, incomplete testing transparency, and uneven customer experience that continue to attract skepticism and complaints."