One of my new quests is trying to modify my gut biome to optimal.
Looking at gut biome reports from centenarians’ guts, it looks like it may be a key part of the puzzle. I think that there are some more promising probiotics, especially for the elderly gut.
I have tried several shotgun (supplements containing 10 or more) probiotics.
This year am trying a more specialized approach based on the latest research. You have to be careful when ordering and using probiotics because some research is based on very specific organisms. I.e.:
Bifidobacterium longum is not the same as Bifidobacterium longum BB536®, Clostridium butyricum is not the same as Clostridium butyricum MIYAIRI 588, and Lactobacillus reuteri is not the same as Lactobacillus reuteri ATCC PTA 6475. This really, really counts if you are seeking the same results from the studies. Also, sometimes the form counts; in this case of the Akkermasia pasteurized version is preferable to the common versions. Strain specificity matters enormously. “Lactobacillus plantarum” without a strain designator is essentially meaningless.
Excerpts from: Claude Opus 4.7 and Haiku 4.5
“This is a topic where the science has advanced considerably in just the last several years, particularly with the centenarian microbiome work coming out of Japan, Sardinia, and China.”
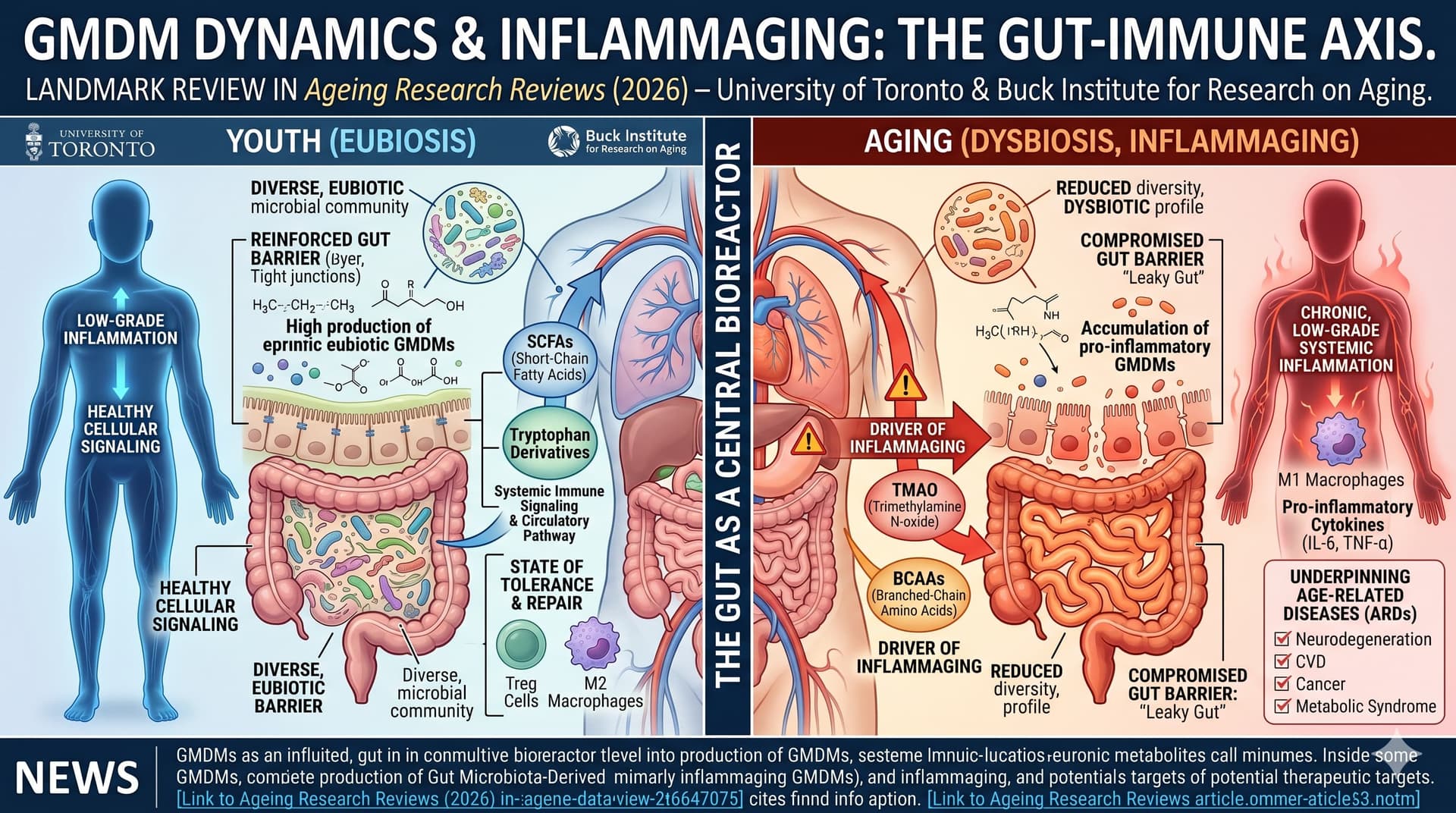
Most centenarian-enriched organisms (Odoribacteraceae, Eubacterium limosum, Christensenella, and Bacteroides fragilis) are not available as commercial probiotics. So when matching probiotics to longevity biology, you’re working with a proxy problem.
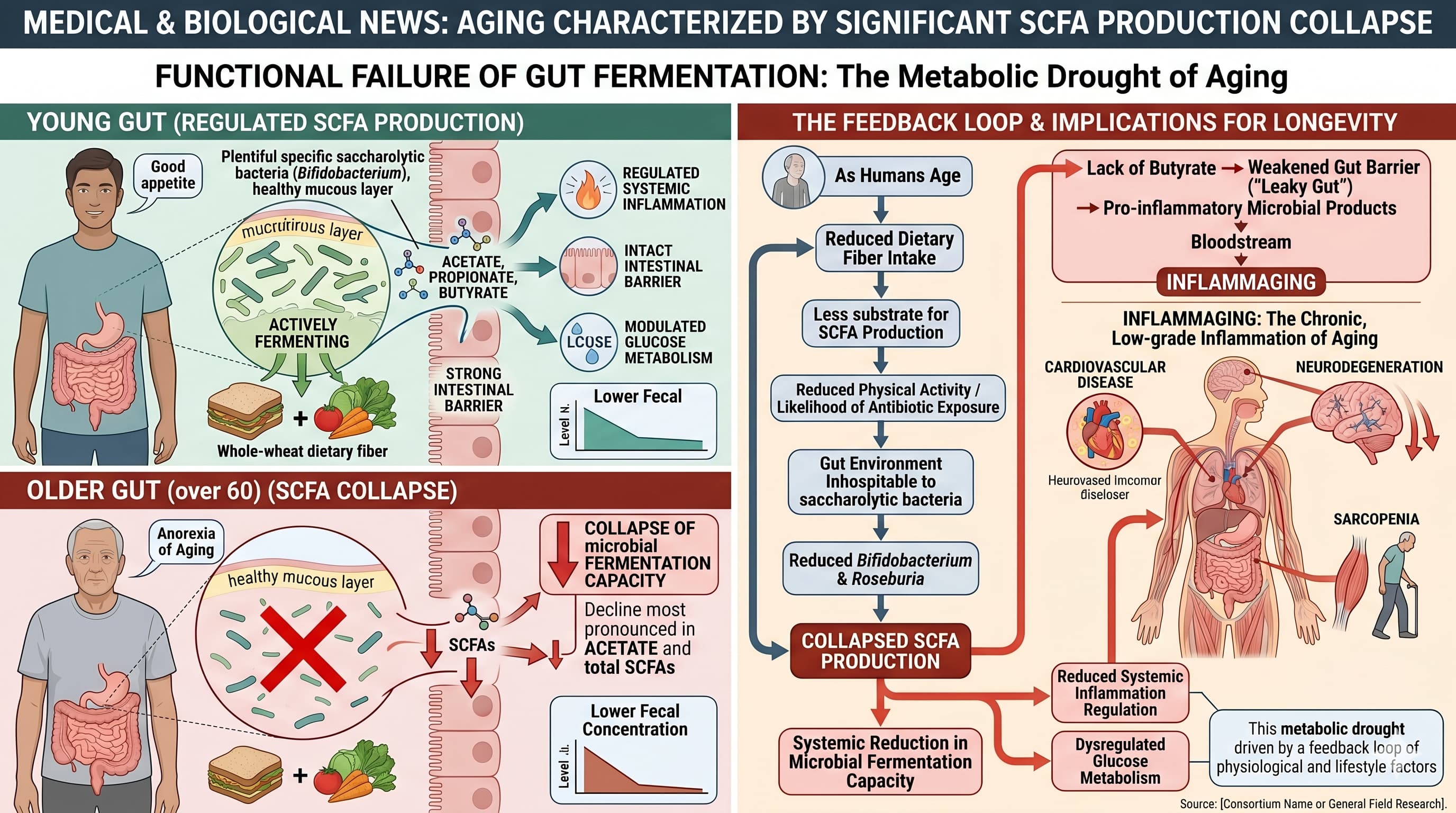
The aging gut has a stereotyped pathophysiology: thinning mucus layer, increased permeability (“leaky gut”), reduced butyrate production, declining bifidobacteria, and a shift toward pathobionts driving inflammaging. Levels of LPS-binding protein and soluble CD14—markers of leaky gut—are significantly higher in older adults and are linked with age-associated phenotypes such as decreased physical activity and increased risk of heart-failure. Strain selection should map to these mechanisms. biorxiv
Akkermansia muciniphila AH39. Of everything on your list, this is arguably the best longevity-aligned probiotic with actual human data
Clostridium butyricum MIYAIRI 588 (Miyarisan). Mechanistically excellent for the aging gut because butyrate is exactly what’s deficient. The 12-week RCT in malnourished elderly (mean age 83) showed C. butyricum increased Akkermansia muciniphila and Alistipes putredinis, decreased IFN-γ, increased the gut barrier tight junction protein occludin, and improved the nutrition biomarker prealbumin—a remarkable finding since it suggests CBM 588 recruits Akkermansia rather than replacing it. The bacterium also increased the abundance of Bifidobacterium, Lactobacillus, and Lactococcus species and enhanced intestinal barrier function, and administration of CBM 588 increased mucin production (measured as MUC2 expression) and significantly decreased epithelial damage. As a spore-former, it survives stomach acid and antibiotics—a meaningful practical advantage in this age group. This is one I’d weight heavily. nih + 2
Bifidobacterium longum BB536. RCT: a randomized double-blind placebo-controlled trial in 80 older adults (mean 77.9 years) with chronic constipation using BB536 at 5×10¹⁰ CFU daily for 4 weeks, with constipation being one of the most common aging-gut complaints. Given Bifidobacterium decline is one of the most consistent features of the aging gut, this is a sensible foundational strain. PubMed + 2
Lactobacillus reuteri ATCC PTA 6475. This is the strain with the cleanest hard-endpoint RCT data in older women. The Nilsson trial: L. reuteri 6475 should be further explored as a novel approach to prevent age-associated bone loss and osteoporosis—after one year of twice-daily dosing. Follow-up mechanism work showed good responders had significantly higher gut microbial gene richness and improved inflammatory state, with detrimental enrichment of E. coli alleviated. PubMednih
Lactobacillus plantarum (TWK10 strain specifically). Unique among lactobacilli for muscle-mass evidence. The Lee et al. RCT in frail older adults found L. plantarum TWK10 supplementation produced increased muscle mass and improved functional performance. Given your interest in sarcopenia, myostatin pathways, and the gut-muscle axis, this strain has the best human data of the lactobacilli for that endpoint. Lactobacillus probiotics serve as an adjunct treatment for sarcopenia by alleviating the inflammatory state, clearing excess reactive oxygen species, improving skeletal muscle metabolism, and regulating the gut microbiota. nihnih
Lactobacillus rhamnosus GG. Good safety data in the elderly (the MGH phase 1 confirmed tolerability at 10¹⁰ CFU twice daily for 28 days), broad evidence for antibiotic-associated diarrhea prevention, and reasonable mucosal immunity effects. Less specific to longevity biology but a reliable workhorse.
Lactobacillus casei / paracasei, L. helveticus. Modest evidence for immune modulation in the elderly; L. helveticus has interesting blood-pressure data via ACE-inhibitor peptides, which may matter for your cardiovascular context, but the trials are smaller and shorter than the BB536 or reuteri 6475 work.
B. lactis (BB-12, HN019), B. breve, B. infantis, B. adolescentis. All reasonable bifidobacteria with general gut-health data. B. adolescentis is interesting because it’s a GABA producer and tends to decline with age. B. infantis is more relevant to infant/young adult guts. B. lactis HN019 has decent transit-time data in older adults.
L. acidophilus, L. bulgaricus, S. thermophilus. Yogurt-tradition strains. Useful for general digestion and lactose handling, modest immune effects, but not where .
L. gasseri. Body composition data exists (LG2055 strain, visceral fat reduction); not a longevity-first pick.
L. crispatus, L. salivarius. More associated with vaginal/oral health respectively than aging gut.
Pediococcus pentosaceus, P. acidilactici, Lactococcus lactis. Generally safe lactic-acid bacteria, sometimes used in formulations for fermentation diversity, but lack standout aging-specific human data.
L. brevis. Some GABA-production interest for sleep/anxiety; modest evidence overall.
What I’d actually prioritize for your context
Given your history, borderline A1c, sarcopenia/myostatin interest, and evidence-tiered approach, the strains with the best risk-adjusted case for someone your age:
Akkermansia muciniphila AH39 — barrier + metabolic + longevity-cohort signature
Clinical trial evidence: AH39 is one of only a few Akkermansia strains that has been evaluated in a randomized, double-blind, placebo-controlled human trial. In a 2025 clinical trial published in Clinical Nutrition ESPEN over 8 weeks, the AH39 group showed significantly reduced waist circumference, waist-to-height ratio, body fat percentage, and AST liver enzyme levels, with no adverse effects recorded. AH39: A Clinically Studied Akkermansia Strain at the Center of Metabolic Research
Clostridium butyricum MIYAIRI 588 — butyrate, barrier, recruits Akkermansia, antibiotic-resilient
Bifidobacterium longum BB536 — best-studied elderly bifido, immune support, constipation
Lactobacillus reuteri ATCC PTA 6475 — bone density, only if osteopenia is on your radar
Lactobacillus plantarum TWK10 — only if specifically targeting sarcopenia/muscle mass.
NOTE: Only this specific form TWK10 has a significant effect on muscle mass.
Three honest limitations worth flagging:
Strain specificity matters enormously. “Lactobacillus plantarum” without a strain designator is essentially meaningless—TWK10 has muscle data, WCFS1 doesn’t show the same effects, and a generic L. plantarum capsule may be neither.
Diet trumps capsule. The Sardinian centenarian data shows significant correlations between bacterial taxa and clinical and lifestyle data
I have taken a course of Akkermansia in the past, but I don’t remember the specific species or source. I don’t recall the subjective results.
My next experiment will be with Lactobacillus plantarum TWK10. Source
and Clostridium butyricum MIYAIRI 588 (CBM 588) Source
“Miyarisan is a well-known Japanese probiotic supplement containing the unique Clostridium butyricum MIYAIRI 588 (CBM 588) strain.”
At my age, anything that helps with muscle mass and bone density is important to me.
For those of you who want to use Akkermansia, the Double Wood brand on Amazon has the preferred strain AH39.