Our skin serves as the frontline shield against environmental insults, making it a highly accurate reflection of our biological age. However, the precise mechanisms translating lifestyle factors into epigenetic alterations have remained largely opaque. Now, a collaborative study between Beiersdorf AG and University Medicine Greifswald, published in Clinical Epigenetics, brings new clarity to this process by uncovering 37 specific lifestyle, physiological, and pharmacological factors that significantly alter the epigenetic trajectory of human skin.
Rather than looking at standard biological markers, the research team utilized the skin-specific “Bormann clock” to determine the DNA methylation (DNAm) age of epidermal samples from 851 participants. They cross-referenced these epigenetic ages against 326 distinct life factors. The results were telling: 20 factors were robustly linked to decelerated skin aging, while 17 were associated with accelerated aging.
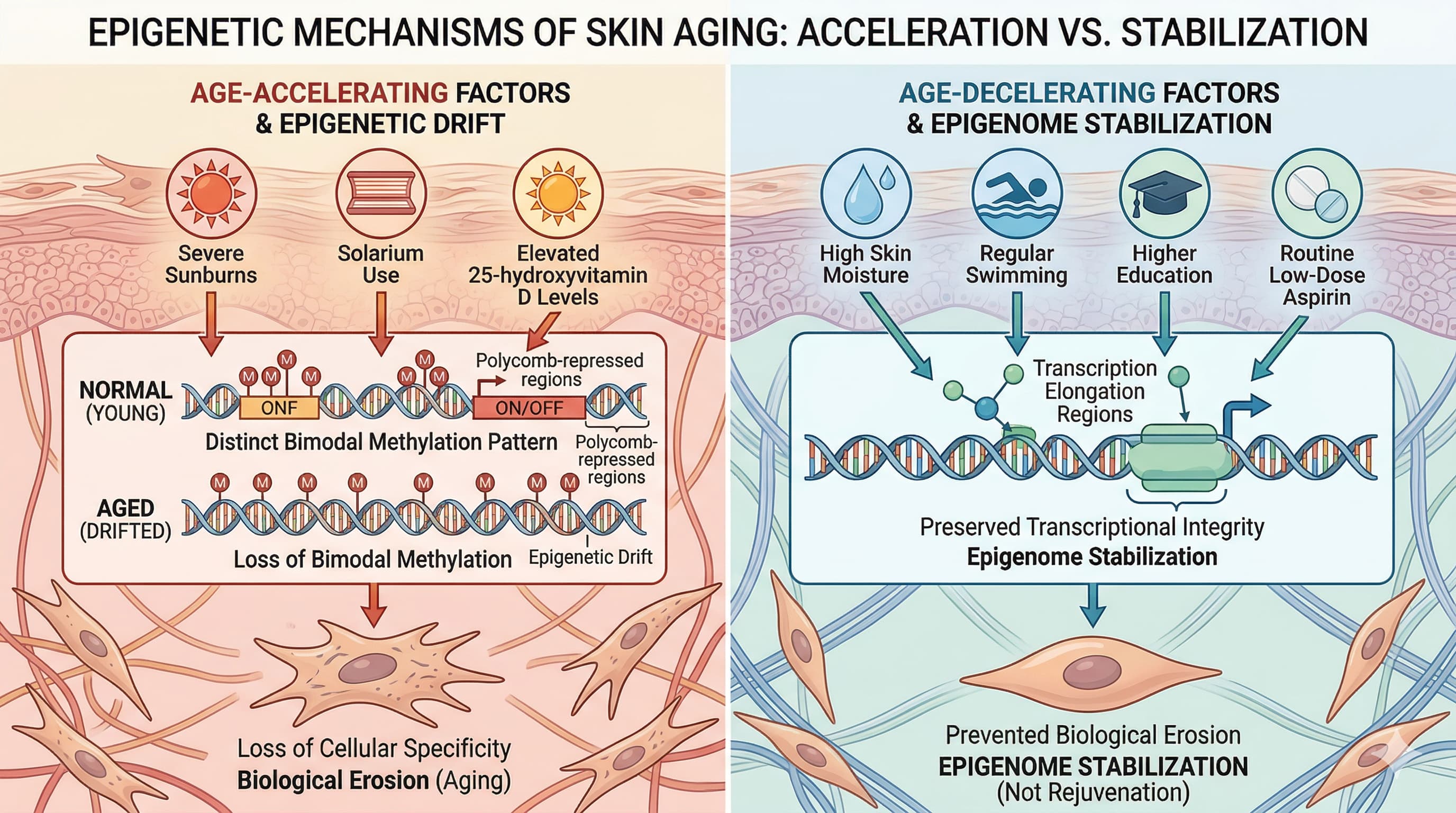
What sets this study apart is its deep dive into how these factors operate. The researchers discovered that age-accelerating factors—such as a history of severe sunburns, solarium / tanning bed use, and elevated 25-hydroxyvitamin D levels—promote “epigenetic drift”. This drift is characterized by a loss of the bimodal DNA methylation patterns necessary for maintaining cellular specificity, particularly in polycomb-repressed regions. Conversely, age-decelerating factors—including high skin moisture, regular swimming, higher education, and the routine use of low-dose aspirin—target transcription elongation regions. Rather than actively rejuvenating the skin, these positive factors seem to stabilize the epigenome, preserving transcriptional integrity and preventing the biological erosion associated with age.
The researchers also matched their findings against known longevity compounds. They demonstrated that patterns induced by topical dihydromyricetin (DHM) and other metabolic modulators share substantial overlap with the CpG sites protected by age-decelerating lifestyle choices. This strongly suggests that the same anti-aging pathways modulated by daily habits can be directly targeted through specific biotech interventions. While the data represents a major step forward, the reliance on correlative population data means we are observing associations rather than definitive causes. Nonetheless, this sweeping epigenetic map provides a vital foundation for next-generation skin longevity interventions.
Context & Impact:
-
Paper: Unraveling the complexity of skin’s biological aging utilizing epigenetic clocks
-
Institution: Beiersdorf AG and University Medicine Greifswald
-
Country: Germany
-
Journal: Clinical Epigenetics, Published: 09 April 2026
-
Impact Evaluation: The impact score of this journal is 4.8, evaluated against a typical high-end range of 0–60+ for top general science, therefore this is a Medium impact journal.
2 Likes
Epigenetic Modulators of Skin Aging: Accelerators and Decelerators
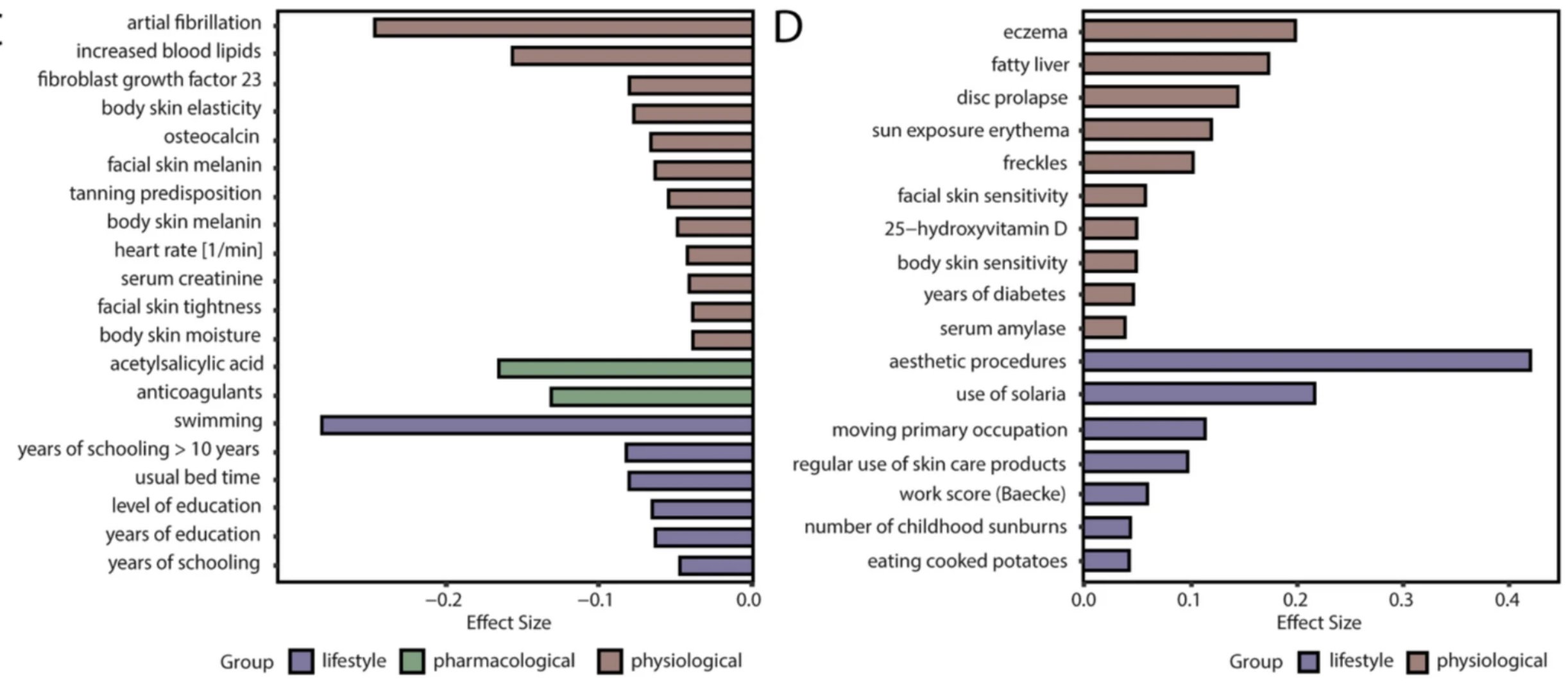
This analysis of the SHIP-TREND-1 cohort data evaluated 851 epidermal samples using the Bormann skin-specific epigenetic clock. By cross-referencing DNA methylation (DNAm) age against 326 variables, the researchers identified 17 factors associated with accelerated epigenetic aging and 20 factors associated with decelerated epigenetic aging.
1. Age-Accelerating Factors
These variables demonstrate a positive correlation with increased DNAm age relative to chronological age.
-
Dermatological Exposures & Phenotypes: Number of childhood sunburns, regular use of solaria, sun exposure erythema (redness upon unprotected exposure), freckles, facial skin sensitivity, body skin sensitivity, and a documented history of eczema .
-
Systemic & Metabolic Pathologies: Years of diabetes mellitus, fatty liver disease, and elevated serum amylase.
-
Biomarkers: High blood levels of 25-hydroxyvitamin D. (Note: This is likely a proxy for high cumulative, unprotected UV exposure rather than a direct toxic effect of the vitamin itself) .
-
Lifestyle & Occupational: Eating cooked potatoes, regular use of skin care products, aesthetic procedures, high occupational physical activity (Baecke work score), and moving primary occupation .
-
Structural: Disc prolapse.
Mechanistic Explanation: The Loss of Cellular Specificity
Accelerating factors drive “epigenetic drift.” At the molecular level, these factors cause a loss of the precise bimodal DNA methylation patterns characteristic of youthful tissue, shifting methylation values toward a uniform mean. The differentially methylated positions (DMPs) associated with these accelerators are heavily enriched in polycomb repressed regions. These regions are critical for silencing developmental genes and maintaining strict cellular identity. The data suggests that accelerating factors compound natural aging by eroding the epigenetic boundaries that keep epidermal cells highly specialized.
2. Age-Decelerating Factors
These variables demonstrate a negative correlation with DNAm age, indicating a slower pace of biological aging in the epidermis.
-
Dermatological Phenotypes: High body skin moisture, high body skin elasticity, facial skin tightness, and high melanin content (facial and body) .
-
Phototypes: High tanning predisposition (the tendency to tan rapidly without burning).
-
Pharmacological Interventions: Regular use of low-dose acetylsalicylic acid (aspirin) and anticoagulants.
-
Systemic Pathologies & Biomarkers (Paradoxical Findings): Atrial fibrillation, increased blood lipids, elevated serum creatinine, high resting heart rate, osteocalcin, and fibroblast growth factor 23.
-
Socioeconomic & Lifestyle: Higher educational attainment (years of schooling > 10 years, level of education), regular swimming, and consistent/usual bedtimes .
Mechanistic Explanation: Stabilization of Transcriptional Integrity
Unlike accelerating factors, decelerating factors do not actively “rejuvenate” the methylome. Instead, their associated DMPs display high variance that resists age-related degradation. These protective DMPs are enriched in transcription elongation and weakly transcribed regions (marked by weak H3K36me3 histone modifications). Furthermore, the genes targeted by these factors are heavily enriched for transcription factors and epigenetic modifiers (such as DNMTs and TETs). This suggests that age-decelerating factors act by stabilizing the epigenetic machinery itself, preserving RNA polymerase II pacing and maintaining transcriptional fidelity over time.
Knowledge Gaps and Methodological Limitations
To effectively utilize this data for longevity optimization, the following translational barriers must be acknowledged:
-
Causality vs. Confounding: This is cross-sectional EWAS data. Several “decelerating” factors (e.g., atrial fibrillation, anticoagulants, high creatinine) are closely linked to cardiovascular disease and the coincident prescription of low-dose aspirin . The model exhibits high collinearity for these variables, making it highly probable that systemic medical management—rather than the pathologies themselves—is driving the epigenetic signal .
-
UV Exposure Proxies: Variables such as elevated 25-hydroxyvitamin D (accelerator) and high tanning predisposition/melanin (decelerators) emphasize the dominant role of UV radiation in skin aging. Additional controlled data is required to disentangle endogenous molecular resilience from simple behavioral photoprotection or phenotypic UV resistance.
-
Translational Equivalency: The study identifies that specific topical compounds, such as dihydromyricetin (DHM), induce methylation shifts that overlap significantly with the CpGs protected by age-decelerating lifestyle factors . Further in vivo interventional trials are necessary to verify if manipulating these transcription elongation pathways pharmacologically translates to a durable extension of tissue healthspan.
Study Design Specifications
-
Type: Cross-sectional epidemiological Epigenome-Wide Association Study (EWAS) with secondary in vitro and in vivo validations.
-
Subjects: * Primary Cohort: 851 human epidermal suction blister samples (SHIP-TREND-1 cohort; mean age 56.8; 54.88% male, 45.12% female).
-
Secondary In Vivo Validation: 30 human females aged 31–65 (topical DHM intervention, vehicle-controlled).
-
In Vitro Validation: Primary human keratinocytes (Bezafibrate/NAD treatments; N=4–5 per treatment vs. N=4–5 controls).
Mechanistic Deep Dive
-
Age-Accelerating Factors: Variables like severe UV damage and duration of diabetes drive “epigenetic drift” by flattening the bimodal methylation landscape—essentially blurring epigenetic contrast. These alterations aggregate in polycomb-repressed chromatin regions and heavily target T-box transcription factors. This pattern points directly to a gradual breakdown in cellular identity and functional specificity.
-
Age-Decelerating Factors: High baseline skin hydration, regular physical activity (swimming), and systemic interventions like low-dose aspirin stabilize DNA methylation in genomic regions associated with transcription elongation (marked by weak H3K36me3 signals). This suggests these factors slow biological aging by supporting RNA polymerase II pacing and maintaining transcriptional fidelity.
-
Core Pathways: Pathway analysis linked the genes associated with both accelerating and decelerating factors directly to recognized Hallmarks of Aging. The most significantly enriched networks were deregulated nutrient sensing (mTOR/IGF-1), altered intercellular communication, and stem cell exhaustion.
Rapamycin’s Potential: mTOR and Geroconversion
In the context of the Beiersdorf study, deregulated nutrient sensing (specifically the mTOR/IGF-1 axis) was identified as a primary driver of epigenetic age acceleration. Hyperactive mTOR signaling drives “geroconversion”—the transition of a cell from reversible cell cycle arrest into irreversible, hyper-secretory cellular senescence. Rapamycin functions as a highly specific, allosteric inhibitor of mTOR Complex 1 (mTORC1). By inhibiting this pathway, it forces epidermal and dermal cells to downregulate anabolic protein synthesis and upregulate autophagy, thereby clearing the accumulated lipofuscin and damaged organelles that characterize aged skin Rapalogs and mTOR inhibitors as anti-aging therapeutics.
Topical Rapamycin (The Localized Strategy)
Topical rapamycin provides targeted mTOR inhibition to the skin without systemic exposure.
-
Clinical Efficacy: A landmark 2019 prospective, randomized, placebo-controlled trial evaluated a low-dose topical rapamycin formulation (10 μM) applied daily for 6–8 months. The treatment significantly reduced cellular senescence, evidenced by a marked decrease in the p16INK4A protein Topical rapamycin reduces markers of senescence and aging in human skin.
-
Structural Impact: The same trial demonstrated a statistically significant increase in Collagen VII, a protein critical for anchoring the basement membrane to the dermis, resulting in clinically observable reductions in photoaging, wrinkling, and sagging.
-
Translational Gap: While the data strongly supports topical efficacy, the optimal carrier vehicle for dermal penetration and the precise concentration curve remain undefined. Off-label compounding typically ranges from 0.001% to 0.1%, but long-term data on local immunosuppression (e.g., susceptibility to cutaneous infections) at higher doses is lacking Commentary: rapamycin for the aging skin.
Oral Rapamycin (The Systemic Strategy)
Oral rapamycin acts systemically. Because skin aging is heavily influenced by systemic metabolic dysfunction, circulating inflammatory cytokines, and advanced glycation end-products, oral administration provides downstream dermatological benefits.
-
Systemic Efficacy: Inhibiting mTOR globally via intermittent oral dosing (frequently 5–10 mg weekly in biohacker/clinical longevity protocols) extends healthspan and lifespan in murine models, maintaining broader stem cell pools and improving proteostasis Unbiased evaluation of rapamycin’s specificity as an mTOR inhibitor.
-
Skin Impact: Systemic downregulation of mTOR theoretically improves skin quality by reducing systemic inflammation and extending the proliferative potential of dermal fibroblasts. However, there are zero randomized controlled trials proving oral rapamycin directly reverses clinical skin aging in humans.
-
Critical Limitations: Using oral rapamycin solely for dermatological anti-aging is an over-leveraged risk. Continuous (daily) or high doses of pulsed systemic mTOR inhibition risks modulating mTORC2, which can trigger immunosuppression, insulin resistance, and lipid dysregulation.
The Verdict
If the primary endpoint is dermal rejuvenation and the prevention of localized photoaging, topical rapamycin is the superior, evidence-backed strategy. It directly targets the mTOR-driven senescence in the epidermis and dermis with a high safety profile. Oral rapamycin remains a systemic longevity play; any improvements to skin architecture from oral use should be considered a secondary phenotypic benefit, not the primary indication.
I had never heard of this, so did some more research…
Topical Dihydromyricetin (DHM), also known as Ampelopsin, is a flavonoid typically extracted from Vine Tea (Ampelopsis grossedentata) or the Japanese raisin tree. While historically utilized as an oral nutraceutical to accelerate alcohol metabolism and support hepatic function, dermatological research has recently repositioned it as a potent active ingredient for epigenetic skin rejuvenation, anti-glycation, and follicular modulation.
Mechanisms of Action in the Skin
Recent literature identifies several distinct pathways through which topical DHM impacts cutaneous healthspan:
-
Epigenetic Age Reversal (DNMT1 Inhibition): A 2024 screening of 1,800 natural substances identified DHM as a robust inhibitor of DNA methyltransferase 1 (DNMT1). By inhibiting this enzyme, DHM reverses age-related DNA hypermethylation, restoring a more youthful epigenetic pattern in primary human keratinocytes and reducing the biological “epigenetic age” of the tissue Frontiers in Aging, 2024.
-
Anti-Glycation (RAGE Inhibition): DHM binds directly to the Receptor for Advanced Glycation End-products (RAGE), effectively functioning as a RAGE inhibitor. This interaction suppresses the AGE-RAGE signaling pathways that typically drive cellular senescence, collagen degradation, and the loss of skin elasticity MDPI Nutrients, 2024.
-
Follicular Modulation: Depending on the formulation matrix and concentration, DHM is utilized to alter the hair follicle cycle. It is frequently applied as an active agent to slow unwanted body hair growth by driving follicles prematurely into the telogen (resting) phase.
Leading Brand Names and Market Segments
| Market Segment |
Leading Brand |
Key Product / Active Name |
Formulation Context |
| Epigenetic Anti-Aging |
Beiersdorf (Eucerin & Nivea) |
Epicelline® (e.g., Eucerin Hyaluron-Filler Epigenetic Serum) |
Marketed as “Age Clock” technology. Beiersdorf patented DHM for its DNMT1-inhibiting properties, formulating it with hyaluronic acid and glycine saponin to visibly reverse structural signs of aging. |
| Hair Growth Inhibition |
The Chemistry Brand (DECIEM) |
Inhibitif Line (Face Serum, Body Hydrator) |
Utilizes DHM’s follicular modulation properties to reduce the apparent thickness and density of unwanted body and facial hair over prolonged use. |
| Body Contouring |
Lipomize (B2B Ingredient) |
Ultramize® |
A nano-encapsulated form of DHM supplied to cosmetic manufacturers, utilizing intracellular signaling to induce localized lipolytic (fat-reducing) and skin-toning effects. |
| Chemical Exfoliation |
Bevel |
Exfoliating 10% Glycolic Acid Toner Pads |
Incorporates DHM as a skin-conditioning and anti-inflammatory agent to soothe post-shave irritation and mitigate follicular inflammation. |
Scholarly Debates and Translational Gaps
While the mechanistic data is compelling, several knowledge gaps remain regarding the efficacy of over-the-counter (OTC) cosmetic applications:
-
Epidermal Penetration: Epigenetic modulation requires the molecule to reach the nuclei of viable cells in the basal epidermis or the dermis. The degree to which raw DHM successfully penetrates the stratum corneum in standard OTC formulations—versus specialized nano-encapsulations—remains actively debated.
-
Therapeutic Thresholds: The effective in vitro doses used in epigenetic and RAGE-inhibition studies are strictly controlled. Commercial cosmetic brands rarely disclose the exact mass fraction of DHM (or Epicelline) in their formulations, making it difficult to verify if they meet the therapeutic threshold required for in vivo DNA demethylation.
-
Independent Validation: Current human data on DHM’s epigenetic reversal capabilities is predominantly funded by the ingredient patent holders. Independent, long-term randomized controlled trials (RCTs) are needed to validate whether the reduction in measurable “epigenetic clocks” directly translates to sustained structural longevity in human subjects.