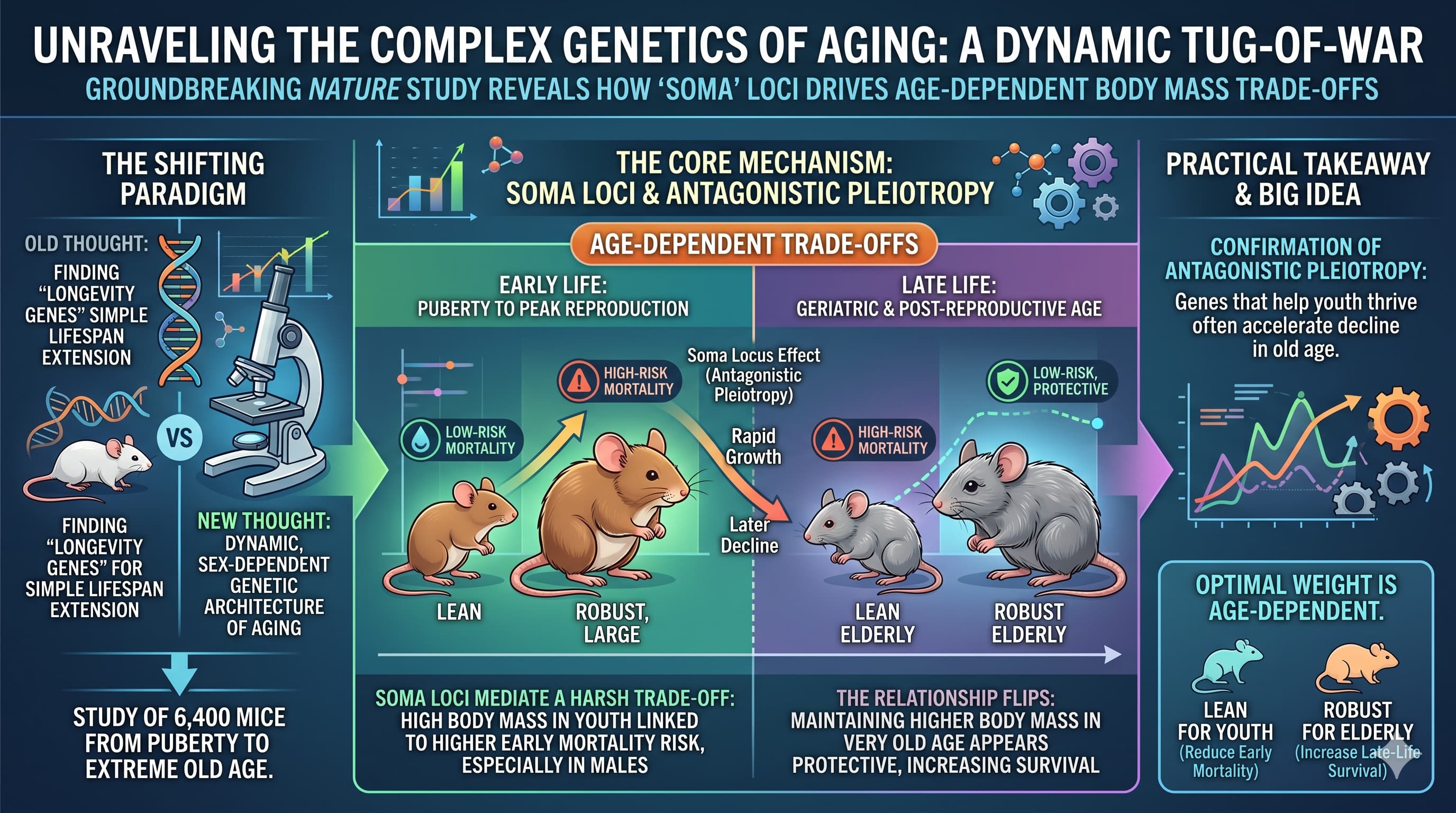
For decades, the search for “longevity genes” has focused on finding DNA variants that simply make animals live longer. A groundbreaking study published in Nature has shifted this paradigm, revealing that the genetic architecture of aging is not a static blueprint but a dynamic, sex-dependent tug-of-war. By tracking the mortality of over 6,400 mice from puberty to extreme old age, researchers identified 59 distinct genetic regions—termed Vita and Soma loci—that control mortality risks at specific stages of life.
The “Big Idea” here is the confirmation of Antagonistic Pleiotropy : the evolutionary theory that genes helping us thrive and grow in our youth are often the same ones that accelerate our decline in old age. The researchers discovered “Soma” loci that mediate a harsh trade-off between body mass and life expectancy. In young mice, being larger and more robust—traits often associated with fitness—is linked to a higher risk of early death. However, as the population reaches the equivalent of human “geriatric” ages, this relationship flips: larger individuals are more likely to survive longer.
Crucially, the study reveals that the genetic networks governing lifespan are almost entirely different for males and females. While 29 Vita loci modulate the risk of death, their effects often invert as an individual ages. For example, a genetic variant that protects a male in his “thirties” might become a liability in his “eighties”. Furthermore, the way these genes interact with each other (epistasis) is strictly divided by sex, suggesting that “one-size-fits-all” longevity interventions are scientifically fundamentally flawed.
Beyond mapping, the team pinpointed high-priority candidate genes like Atp6v1h (linked to lysosomal health) and Acad11 (fatty acid metabolism), validating their effects across species from worms to humans. This research provides the first comprehensive “actuarial map” of aging, offering a precise guide for when and for whom specific longevity interventions—such as dietary changes or drugs—might actually work.
Actionable Insights
-
Body Mass Strategy Inversion: The most practical takeaway is the shifting role of body mass across the lifespan. High body mass (obesity) in early adulthood (puberty to peak reproduction) is genetically linked to higher mortality, particularly in males. However, maintaining higher body mass (e.g. strength) in very old age (post-reproductive phase) appears to be protective. This suggests that “optimal” weight is age-dependent: lean for the youth, robust for the elderly.
-
Sex-Specific Biohacking: Because epistatic (gene-gene) networks are strictly segregated by sex, males and females should not expect identical outcomes from the same longevity supplements or interventions. Genetic “diplomacy” between the sexes means that pathways involving mTOR, autophagy, and lysosomal stress may require sex-tailored dosing or timing.
-
Targeting Late-Life Pathways: The identification of the APEH and Acad11 pathways provides new targets for longevity. Compounds that support lysosomal acidification and mitochondrial beta-oxidation are high-priority candidates for extending healthy lifespan, particularly as they show cross-species relevance in humans.
-
Timing of Interventions: Proactive interventions to blunt the negative coupling of body mass and lifespan must occur early in life to be effective. In contrast, targeting geriatric populations requires a focus on diverse drivers of escalating age-dependent diseases rather than the core aging rate.
Source:
- Source: Dynamics of genetic and somatic trade-offs in ageing and mortality
- Institutions: Northumbria University (UK), University of Tennessee Health Science Center (USA), The Jackson Laboratory (USA), ETH Zurich (Switzerland), and others.
- Journal: Nature, * Published: 22 April 2026
- Impact Evaluation: The impact score of this journal is 50.5 (2024 JIF), evaluated against a typical high-end range of 0–60+ for top general science; therefore, this is an Elite impact journal.