For over a century, the medical establishment has operated under William Osler’s 1892 maxim that “a man is as old as his arteries”. While historically viewed as an irreversible decline, a comprehensive new synthesis published in 2026 confirms that cardiovascular aging (CVA) is a modifiable biological process driven by specific, targetable etiologies. The global burden of cardiovascular disease (CVD) has nearly doubled since 1990, and despite advanced lipid-lowering and antihypertensive therapies, “residual risk” remains high because current treatments often ignore the underlying biology of aging itself.
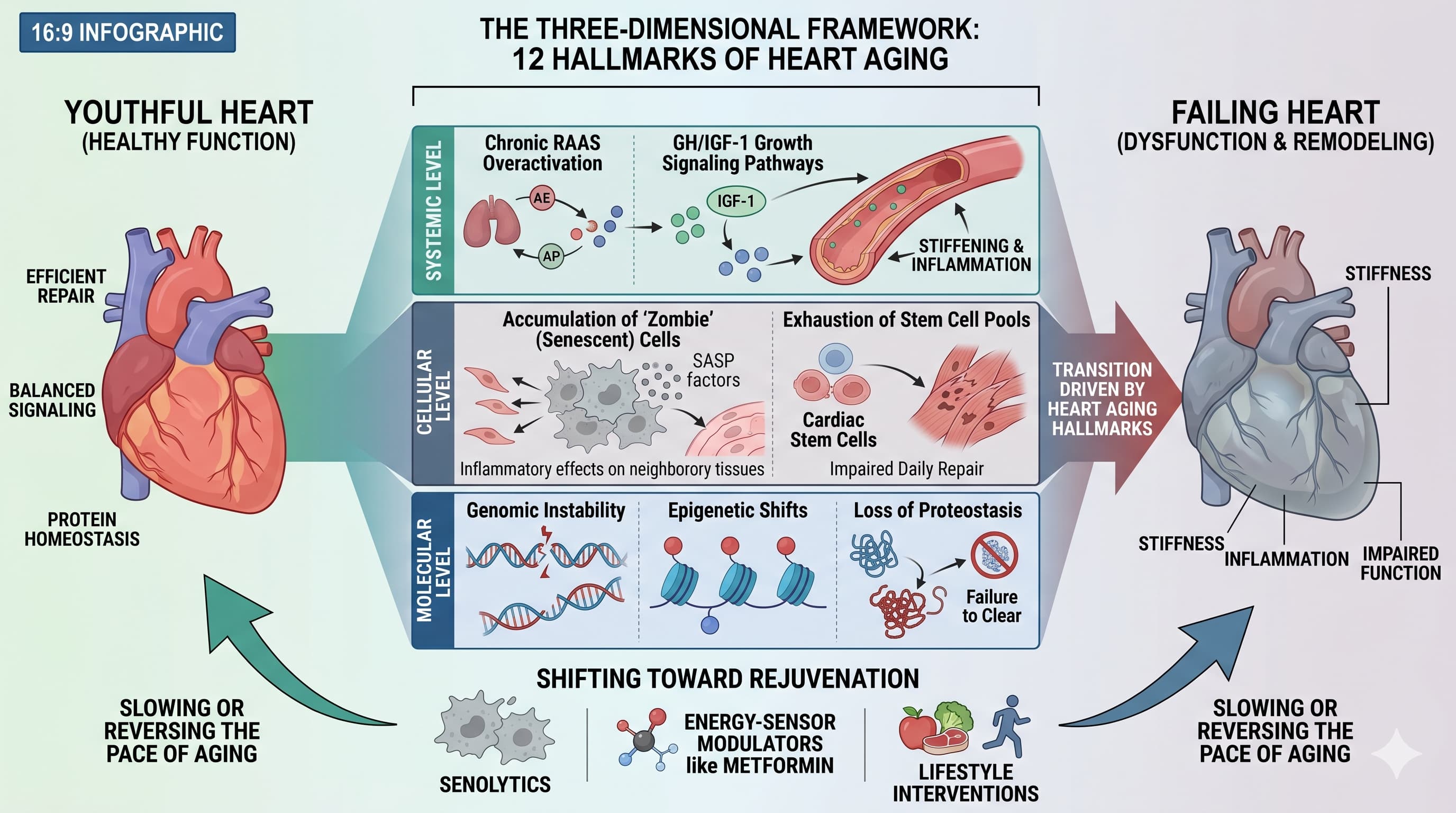
The authors propose an innovative “three-dimensional” framework of 12 hallmarks that drive the transition from a youthful heart to a failing one. At the molecular level , the heart is battered by genomic instability, epigenetic shifts, and the “loss of proteostasis”—the failure to clear misfolded protein aggregates. At the cellular level , the accumulation of “zombie” (senescent) cells and the exhaustion of stem cell pools prevent the heart from repairing daily wear and tear. Finally, at the systemic level , chronic overactivation of the Renin-Angiotensin-Aldosterone System (RAAS) and growth signaling pathways like GH/IGF-1 foster an environment of stiffening and inflammation.
This review highlights a “vicious cycle” where metabolic disorders like diabetes accelerate vascular decay, which in turn worsens metabolic health. However, the narrative is shifting toward rejuvenation. By targeting these hallmarks through senolytics, energy-sensor modulators like Metformin, and lifestyle interventions, it may be possible to slow or even reverse the “pace” of aging.
Actionable Insights
To improve longevity and optimize cardiovascular health, individuals should focus on “poly-therapeutic” lifestyle and compound strategies:
-
Nutritional Modulation : Adhering to Mediterranean or high-quality dietary patterns and considering caloric restriction can optimize vascular architecture and reinforce proteostasis.
-
Targeted Compounds : Molecules such as Spermidine (autophagy enhancer), and NAD+ precursors (like NMN) show promise in reversing arterial stiffness and enhancing cellular repair.
-
Vascular Integrity : Managing postprandial hyperglycemia is critical, as intermittent glucose spikes accelerate endothelial senescence more aggressively than sustained high blood sugar.
-
Neuro-Cardiology : Prioritizing sleep and stress management is essential; sleep deprivation and chronic psychological stress (including social isolation) are independently associated with accelerated vascular aging and plaque vulnerability.
-
Exercise : Regular physical training remains the most effective “medicine” for attenuating arterial stiffening and improving heart rate reserve.
Source:
-
Open Access Paper: Cardiovascular ageing: hallmarks, signaling pathways, diseases and therapeutic targets
-
Institutions: Zhongshan Hospital, Fudan University; Shanghai Institute of Cardiovascular Diseases.
-
Country : China.
-
Journal : Signal Transduction and Targeted Therapy, Published: 21 April 2026
-
Impact Evaluation: The impact score (JIF) of this journal is approximately 40.8, evaluated against a typical high-end range of 0–60+ for top general science; therefore, this is an Elite impact journal.
Study Design Specifications
-
Type : Comprehensive Review Article (Synthesis of in vivo, in vitro, and clinical trial data).
-
Subjects : The review synthesizes data from multiple species including Drosophila (nutrient sensing), Murine models (C57BL/6, ApoE-/-, INK-ATTAC, p21-ATTAC), and Human cohorts (UK Biobank, NHANES, MIVAS).
Mechanistic Deep Dive
The paper identifies a hierarchy of decline:
-
Mitochondrial Dynamics & Oxidative Stress : Mitochondria are the central organelles linking vascular aging to ROS production. Impaired electron transport leads to reduced ATP and increased ROS, which degrades endothelial-derived Nitric Oxide (NO), causing vasoconstriction. [Confidence: High]
-
The cGAS-STING Pathway : This innate immune pathway senses cytosolic DNA (from genomic instability) and drives the SASP (Senescence-Associated Secretory Phenotype), creating a proinflammatory “neighborhood effect” that induces senescence in healthy cells. [Confidence: High]
-
RAAS and Beta-Adrenergic Overdrive : Aging is characterized by chronic overactivation of the RAAS and increased circulating catecholamines. This leads to beta-receptor desensitization , reducing cardiac reserve and promoting fibrotic remodeling. [Confidence: High]
-
The Glymphatic-Vascular Link : A critical “HBA” (Heart-Brain Axis) connection is highlighted: vascular stiffening and PBM (Parenchymal Border Macrophage) dysfunction impair CSF flow and brain waste clearance, directly linking cardiovascular decay to Neurodegenerative Disorders of Aging (NDAs) like Alzheimer’s. [Confidence: Medium]
Novelty
The paper’s primary contribution is the tri-level stratification of 12 hallmarks (Molecular, Cellular, Systemic) and the explicit mapping of these hallmarks to specific diseases like heart failure, aortic dissection, and neurodegeneration. It also provides a cutting-edge consolidation of FDA-approved drugs (Metformin, SGLT2i, GLP-1RAs) being repurposed as “geroprotectors”.
Critical Limitations
-
Translational Uncertainty : Significant reliance on Doxorubicin-induced or genetically modified murine models of senescence, which may not accurately reflect natural human cardiovascular aging.
-
The “Double-Edged Sword” of Senolytics : While clearing senescent cells (SnCs) improves cardiac function, excessive or poorly timed clearance carries risks like impaired wound healing and hepatic fibrosis due to the loss of SnCs required for structural integrity.
-
Methodological Gaps : The review acknowledges that the specific functional roles of different senescent cell types (e.g., cardiac vs. vascular) in driving CVD remain poorly understood.
-
Data Scarcity : Long-term human clinical trial data for “rejuvenation” (not just disease treatment) is largely missing; most “longevity” claims for Metformin or Rapamycin in humans remain speculative or epidemiological.
Based on the research paper, several FDA-approved drugs are being investigated and repurposed for their “geroprotective” potential by targeting specific hallmarks of cardiovascular aging. These interventions aim to address molecular, cellular, and systemic decay rather than just treating symptoms of cardiovascular disease.
The following table summarizes the application information for these repurposed drugs as identified in the study:
Repurposed FDA-Approved Drugs as Geroprotectors
| Drug Category |
Key Mechanisms & Application Information |
| Metformin |
Activates the AMPK pathway and regulates metabolism. It targets multiple aging pathways, including inhibiting mitochondrial respiration, suppressing proinflammatory signals, and modulating the gut microbiota. Epidemiological data suggests it reduces all-cause mortality and age-related comorbidities. |
| Rapamycin |
Functions as an mTOR inhibitor. It is widely used in anti-aging research to improve healthy cardiac phenotypes. At low doses, certain rapalogues have been shown to safely enhance immune function in elderly patients. |
| SGLT2 Inhibitors |
These drugs reduce oxidative stress, inhibit inflammation, and improve overall vascular function. They are noted for providing metabolic “unloading” that can complement other hallmark-targeting therapies. |
| GLP-1 Receptor Agonists |
Primarily used for glucose regulation and satiety, they serve as “lazy strategies” for weight management. Maintaining a healthy weight through these agents is considered crucial for healthy aging and cardiovascular protection. |
| ACEI & ARBs |
ACE inhibitors (ACEIs) increase nitric oxide (NO) production and improve cardiac, metabolic, and endothelial function. Angiotensin II receptor blockers (ARBs) reduce peripheral vascular resistance and aldosterone secretion to lower blood pressure. Both target the RAAS system , which becomes chronically overactivated during aging. |
| β-blockers |
These agents reduce cardiac output, blood pressure, and myocardial oxygen consumption. They prevent excessive cardiac responses to chronic catecholamine stimulation, thereby improving cardiac reserve and prolonging survival in aging populations. |
| Statins |
Beyond lowering cholesterol, statins reduce reactive oxygen species (ROS) levels and increase both NO synthesis and the formation of new blood vessels (neoangiogenesis). |
| PCSK9 Inhibitors |
These lower LDL levels, regulate inflammation and immunity, and improve endothelial function. |
| Acarbose |
This drug is used to stop postprandial blood sugar spikes, which are known to accelerate endothelial senescence. |
| Aspirin |
It decreases the expression of iNOS and Cox-2. However, the paper notes that clinical results in individuals over 70 have been negative, suggesting its use for primary prevention in the elderly is questionable. |
| N-acetylcysteine |
Acts as an antioxidant and anti-inflammatory agent with immunomodulatory properties to mitigate age-related damage. |
| DPP-4 Inhibitors |
Used to mitigate age-related glucose intolerance. |
| CCBs |
Calcium channel blockers are applied to reduce the vascular resistance specifically associated with the aging process. |
| SGC Agonists |
These activate the cGMP pathway to promote vasodilation and counteract vascular stiffness. |
Strategic Considerations for Specialists
-
Multitargeted Approach: The paper emphasizes that because these hallmarks are interconnected, a single-target focus is often ineffective; future strategies should utilize the systemic interactions of these drugs.
-
Translational Challenges: While many of these drugs show promise, specialists must weigh therapeutic benefits against side effects, such as the risk of muscle mass reduction with GLP-1RAs or the potential for impaired tissue repair when clearing senescent cells.
-
Timing of Intervention: Many age-related vascular changes, such as blood-brain barrier breakdown, begin as early as middle age, suggesting that early assessment and prevention are critical.
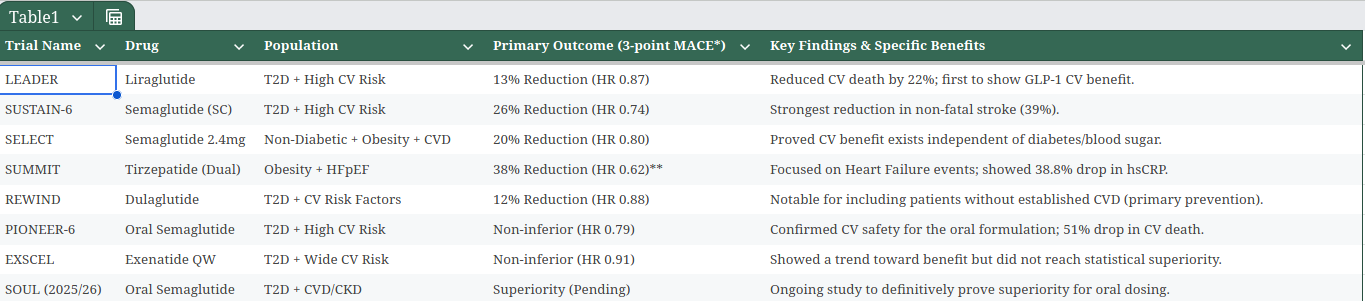
Review does a poor job of summarizing benefits of GLP1s. According to the clinical data, it protects the heart by reducing chronic inflammation and improving vascular elasticity.
CVOT outcomes from trials show 12-38% reduction in 3-point MACE. 3-point MACE typically includes CV death, non-fatal myocardial infarction (heart attack), and non-fatal stroke.
Several post-hoc analyses show that huge reductions in hs-crp, IL-6, and other inflamatory markers are about 50% mediated by weight loss:
To compare: SGLT2 reduce hs-crp by 5-15%, while GLP1 reduce it by 30-50%+ : Frontiers | Inflammatory biomarker response to GLP-1 receptor agonists versus other glucose-lowering medications in patients with type 2 diabetes: a systematic review and meta-analysis
1 Like