(Claude) “The Klotho upregulation data deserve explicit attention because they represent a mechanistic pathway largely absent from every other compound on the top 20 list (a list I had Claude build to specifications). Klotho is a transmembrane protein whose expression declines dramatically with age, and whose genetic variants are associated with longevity across multiple human cohort studies. That telmisartan — a widely prescribed, cheap, generic drug — consistently upregulates Klotho in human studies is an underappreciated finding. Whether this translates to longevity benefit is unknown, but the mechanistic coherence is high and the observation is biologically non-trivial.”
There is a relevant set of experimental data [https://www.semanticscholar.org/paper/Effect-and-mechanisms-of-Telmisartan-on-the-of-in-Mi/bfae3ec8f766009edde0d873289a0f6057951440]. However, this drug doesn’t seem to be common among longevity compounds , it’s actually the first time I’ve heard of it as well.
Losartan and Valsartan are more common, and there is a wealth of human clinical trial data regarding their relationship with Klotho. Telmisartan, however, seems to currently only have mouse data and is rarely discussed among anti-aging compounds [Frontiers | Pathobiology of the Klotho Antiaging Protein and Therapeutic Considerations]. Personally, I am more optimistic about Valsartan.
FWIW…
I take one 20mg{telmisartan] tablet under the tongue once a day. Not for BP.
This is another reason to take
This is obviously not making your bp too low, but I’m curious…
What is your bp normally, and what is it after telmisartan?
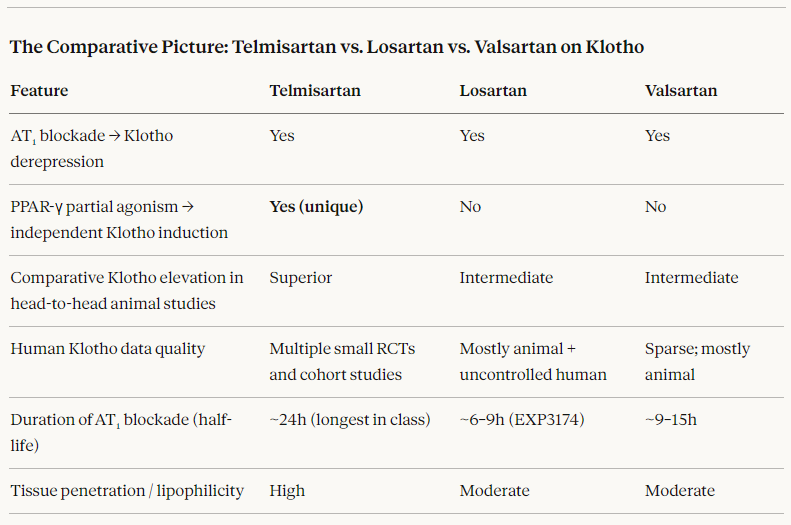
Losartan and Valsartan also upregulate Klotho but the evidence seems to suggest the effect is smaller in magnitude and mechanistically less robust than telmisartan’s effect., at least it is empirically superior in head-to-head animal studies. Then there is telmisartan’s unique PPAR-γ partial agonism leading to independent Klotho induction. This combination may be why it is deemed mechanistically superior, although not well demonstrated in humans.
Here is one comparison table:
Nice data set. This is exactly why I switched to telmisartan 20mg. PITA I have to split the tabs atm, but that’s ok.
I feel that my BP increased a little bit since I switched from 25mg losartan, but really I think it might be the SS-31 I am poking, as that’s when I started seeing the increase.
BP ~110/70 ish? My heart rate kinda sucks though @70 bpm.
Here’s some more information:
Telmisartan demonstrates significant potential in mitigating aortic dilation through multiple mechanisms, independent of blood pressure reduction. In Marfan syndrome (MFS) models, telmisartan fully prevents aortic root aneurysm development via an endothelial nitric oxide synthase (eNOS)-dependent pathway, highlighting its role in enhancing endothelial function rather than solely lowering blood pressure. This effect is more potent than other angiotensin II receptor blockers (ARBs) like losartan and valsartan, even at low, non-hypotensive doses.
In athletes with thoracic outlet syndrome (TOS), telmisartan may counteract ascending aortic dilatation (AAD) by targeting a neuroinflammatory cascade involving sympathetic overactivation, perivascular hypoxia, and the RAGE–CCL2–STAT3 axis. It attenuates this feed-forward loop by reducing sympathetic tone, improving vasa vasorum perfusion, suppressing inflammation, and protecting against extracellular matrix degradation.
Preclinical studies confirm telmisartan’s ability to reduce aortic root widening, inhibit vascular fibrosis, and protect elastin integrity in MFS mice. Additionally, in patients with small abdominal aortic aneurysms (AAA), telmisartan showed a trend toward slowing aneurysm growth, although the difference versus placebo was not statistically significant in one trial.
Overall, telmisartan’s dual action as an AT1 receptor blocker and PPAR-γ agonist makes it a promising disease-modifying agent for aortic dilation in both genetic (e.g., MFS) and acquired (e.g., athlete-related, TOS-associated) conditions.
In addition to all this, it also lowers LDL-cholesterol.
Losartan indeed significantly increases Klotho protein levels in human clinical trials [https://journals.sagepub.com/doi/10.1177/1470320313475905]. However, considering that Telmisartan is more effective at improving metabolic syndrome, insulin resistance, blood glucose, and lipid profiles, Telmisartan is indeed better overall. Furthermore, it can be theoretically inferred that Telmisartan should be even more effective than Losartan at raising Klotho levels. I will definitely give it serious consideration
The directionality may end up being real but you can drive a freight train and a small battleship through the holes in that study in relation to its claims. I see five problems but there are probably more.
Back from a meeting, I can summarize a few reasons the Sage journal article likely should not have been published as framed.
The foundational problem is that this a post-hoc analysis which, especially with this weak sub-design, is not powered to illuminate the klotho issue. The original crossover trial (Lim et al., 2007) was designed to compare losartan versus quinapril for albuminuria reduction in Asian T2DM. Klotho was measured retrospectively in stored samples, or added as a secondary endpoint without prospective power calculation. This means that no a priori sample size calculation for the Klotho endpoint exists and the risk of Type I error inflation from multiple post-hoc comparisons is uncontrolled. One can stop here. At best, this is an hypothesis-generating study.
Irrespective of design issues, the study is significantly underpowered. Thirty-three subjects in a crossover design means 33 observations per arm. For a biomarker with known high inter-individual and intra-individual variability (as I suspect all in this area are) detecting a meaningful between-treatment difference requires substantially larger samples. (In one search, I estimated that klotho has a coefficient of variation in healthy populations of more than 20%.)
It is obvious but noteworthy that there no placebo arm. The study compares losartan to quinapril absent an untreated control, a weakness in the original study compounded here. This means the study can tell you, at best, that losartan raises Klotho relative to quinapril (or that quinapril suppresses Klotho relative to losartan) or that both raise Klotho from baseline but at different rates. You cannot determine from this design whether losartan raises Klotho above untreated baseline. IMO, this is not a minor issue.
I’m sure everyone reading this paper wondered why both drugs were administered at 50% of their maximal approved doses. This is a non-standard approach. Low dose and short time lines are what I often see in a study that wants to make a preconceived point. Although I don’t think that is the case here, it might be the case that the timeline was “shopped” for significance.
I don’t know much about klotho but four weeks seems short to claim a steady state. I have seen ARB studies spanning 24 weeks. Perhaps an expert can add to that.
Another flaw that would benefit from the opinion of an expert is that T2DM patients with pre-existing albuminuria have structurally compromised kidneys, which is the primary organ of Klotho production. It would seem to me, then, that this population likely behaves differently from those in healthy hypertensive individuals. The generalization back to the population seems like an unsubstantiated leap.
Finally, my initial irritation, the title claim: “associated with reduction of albuminuria.” The title asserts an association between losartan-induced Klotho elevation and albuminuria reduction. This is a correlation between two treatment-responsive variables in a four-week trial. It cannot cannot establish that Klotho elevation mediates the anti-albuminuric effect. This problem could have been remedied with a more thoughtful title.
Telmisartan has a half life of 24 hours. Losartan and valsartan have half lives of 6 to 9 hours.
I also take 20mg daily.
Bill Faloon has been enthusiastic about the longevity benefits of Telmisartan for a long time. 10-15 years probably.
Telmisartan doesn’t lower BP immediately after ingestion. It can take weeks for the full effect to be measured. A small dose like 20mg (from memory) only shaves 5-6mm Hg off BP. Barely measurable.
@DrT this is exciting! I never knew I could potentially safely take a low dose. I always have FOMO because my BP is good.
Do many people with good bp take a low dose? Maybe EOD or?
I’ll start reading more about it.
The link below has been posted in other threads on this forum.
Review
Hi Beth, my experience on that is: I started telmisartan 20mg at night 2 months ago ( i already take beta blockers in the morning), my BP move down a little bit like was 11 x 6,5 for 10,5 x 6,0. Some times a little less. Iam confortable w my decision!
Telmisartan is probably the most discussed and most used antihypertensive for its potential anti-aging properties:
- Why would doctors prescribe an inferior drug?
- Rilmenidine vs Telmisartan or other BP meds for Longevity
- Telmisartan -- 40 or 80 mg
- Telmisartan Ameliorates Blood-Brain Barrier Disruption in a High-Salt Diet Mouse Model
That’s why it is being tested in the ITP: https://www.nia.nih.gov/research/dab/interventions-testing-program-itp/supported-interventions
Regarding telmisartan increasing klotho, that effect is actually a general property of drugs and supplements that inhibit angiotensin converting enzyme (ACE) or block the receptors. Other interventions that decrease ACE do the same. This is one of many reasons I’ve tried to remain very lean and have been drinking a bit of pomegranate juice for decades. Being lean and having low blood pressure naturally without drugs will help decrease ACE and could similarly help increase klotho. Pomegranate juice is also a mild ACE inhibitor. Of course the drugs are stronger, but for people with already low blood pressure they can cause more harm than good especially if the diastolic pressure is very low.
I have 40mg on the way for my husband. I’ll try breaking then in 1/4s and see what happens… if that goes well, I’ll take a half
What is the lowest bp that is safe to have? We have a cuff and I’ll closely monitor to make sure I’m not doing any harm.
Thank you!
There may be reasons for not lowering BP too much past the age of 60-70 or so. The literature seems to suggest that slightly higher BP might help with blood perfusion to the brain, particularly if there is some vascular stenosis and also preventing orthostatic hypotension related falls etc. There’s mixed signals in dementia and BP in the elderly, where very low BP is associated with higher risk of dementia, while mildly elevated is not. In general systolic should not go much below 90 and diastolic much below 60.