I am not a nephrologist, but I work with several and I am good friends with one. So here are some things I I’ve learned.
In an elderly person with low muscle mass, they have a nice low creatinine, resulting in a high eGFR (especially when adjusted for their body weight), and that masks their poor kidney function. There also used to be a “racial” adjustment factor, based on the idea that people of African origin carried more muscle mass, which I think was removed from guidelines. And that was basically masking poor kidney function in those people. However, there are actually some differences between ethnicities. Japan applies a multiplier of 0.8, for example, in recognition of their population generally being lower BMI, less muscle, and that the normal equation again masks declining kidney function.
I also learned that cystatin-C isn’t perfect either. It’s affected by inflammation, thyroid activity and obesity.
Basically, if kidney function really matters, then you need to directly measure it with clearance tests where they give you a contrast agent and you collect urine over time to measure actual GFR. (I’m sure @LaraPo would have done before!) But it’s rarely done outside of research labs or very serious conditions (transplants, dosing chemotherapy etc)
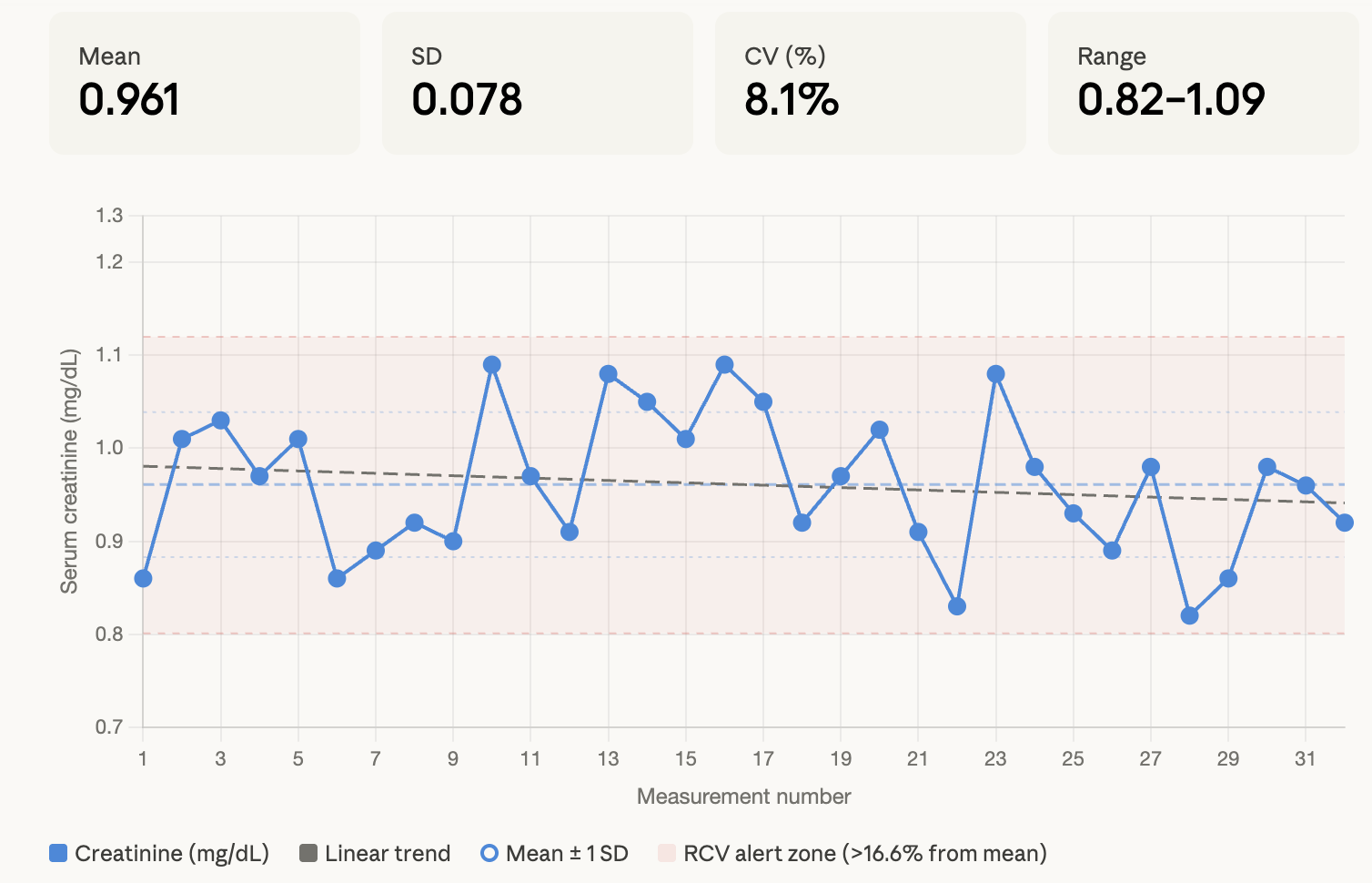
I consider these blood tests as something for monitoring, since you need pretty dramatic changes to cause alarm. For example, here are my readings over the last 10 years:
So, biological CV% is around 5%, analytical CV% of the assay is around 3% at a good lab. So combining these together equates to expected variations of ~7% when measuring creatinine.
Add onto that the “reference change value” which is the way to detect what is measurement noise versus a real change. For my numbers, that would be around 16.6%. For me, that would mean a result outside of 0.83 or 1.15, which is a pretty huge range.
For acute kidney injury, an increase of >0.3mg/dl within 48 hours, or 1.5X your baseline over 7 days would be an indicator that something bad has happened.
Aside from that, I don’t think these values are super useful for anything except long-term monitoring. I wouldn’t take great faith in them being “normal”. You can also see that my own creatinine is slightly falling on average over 10 years, which is the opposite of what we’d expect with ageing. But all of it is still totally within normal parameters, so I don’t get excited either way.