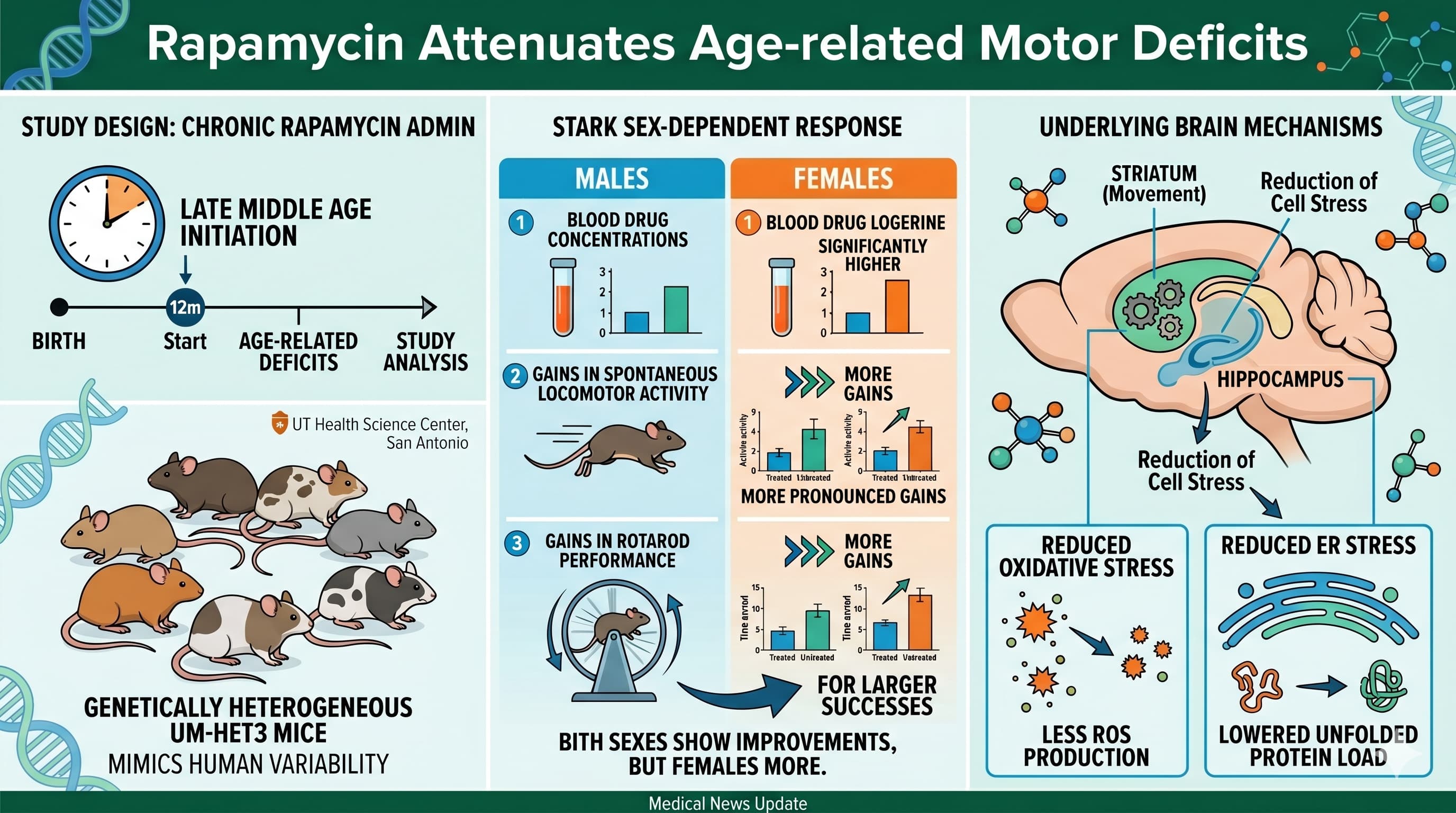
While the Interventions Testing Program (ITP) has solidified rapamycin’s reputation as a robust lifespan-extending pharmacological agent, the question of whether it preserves functional “healthspan”—specifically motor coordination—has remained contested. Researchers at the University of Texas Health Science Center have now demonstrated that chronic rapamycin administration, even when initiated in late middle age (12 months), significantly attenuates age-related motor deficits in genetically heterogeneous UM-HET3 mice. This finding is critical because it utilizes a genetically diverse model that better mimics the variability of human aging compared to standard inbred laboratory strains.
The study revealed a stark sex-dependent response (which is commonly seen in mice dosing rapamycin): while both sexes showed improvement, female mice exhibited significantly higher blood concentrations of the drug and more pronounced gains in spontaneous locomotor activity and rotarod performance. The underlying mechanism appears linked to the reduction of oxidative stress and endoplasmic reticulum (ER) stress within brain regions governing movement, such as the striatum and hippocampus. Specifically, rapamycin lowered levels of protein carbonyls—markers of permanent oxidative damage—in the insoluble protein fractions of these regions.
One paradoxical finding emerged: rapamycin treatment further increased the expression of Glial Fibrillary Acidic Protein (GFAP) in the striatum. While GFAP is typically a marker of neuroinflammation, the researchers hypothesize that in this context, it may represent a defensive astrocytic response that protects neurons from further damage. Ultimately, the study suggests that the “longevity dividend” of rapamycin is not merely a matter of more days, but better days characterized by maintained physical autonomy.
Actionable Insights For those optimizing for longevity and healthspan, this research offers several high-utility takeaways. First, the efficacy of “late-start” rapamycin is confirmed; intervention at an age equivalent to roughly 40–50 human years was sufficient to reverse existing motor decline. This narrows the window of “too late to start” significantly. Second, the study highlights a critical sex-specific dosing reality. Females achieved higher systemic concentrations than males (in mice) on the same dietary dose, which translated to superior functional outcomes but also potentially higher risk profiles.
Practically, the reduction in protein carbonylation suggests that rapamycin’s primary neuroprotective value may lie in its ability to manage “molecular garbage” (unfolded or damaged proteins) that accumulates over decades. However, the observed decrease in lean mass in treated mice warrants caution; while it did not impair grip strength in this study, it underscores the necessity of pairing mTOR inhibitors with resistance training to offset potential sarcopenic effects. The study also hints that rapamycin may improve motivational states (depressive-like behavior) in females, potentially via the same neuro-oxidative pathways.
Source
- Paywalled Paper: Chronic rapamycin treatment attenuates age-related motor deficits in sex-dependent manner in UM-HET3 mice
- Institutions: University of Texas Health Science Center at San Antonio (Barshop Institute for Longevity and Aging Studies); University of Oklahoma Health Sciences.
- Country: USA.
-
Journal: Journal of Gerontology: Biological Sciences.
Impact Evaluation The impact score of this journal is 5.1 (2024 Impact Factor), therefore this is a Medium impact journal. It is, however, considered a top-tier specialty journal within the field of geroscience.