I’ll be going out and buying a big container of walnuts after reading this… ![]()
Funny! The same thing crossed my mind.
Great minds think alike. ![]()
Walter WIllett claims science loves nuts as well, so do eat them as a significant part of meals, to improve health though nutrition.
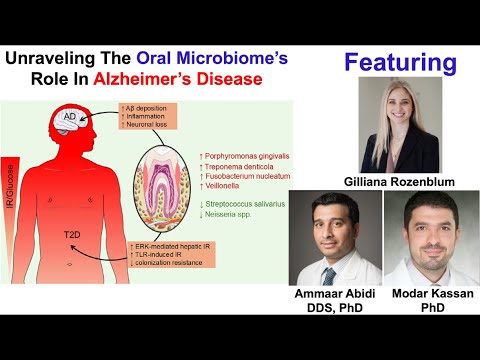
Unraveling The Oral Microbiome’s Role In Alzheimer’s Disease
Google Gemini AI Video Summary and Analysis:
This analysis covers the presentation “Unraveling the Oral Microbiome’s Role in Alzheimer’s Disease” by researchers from Lincoln Memorial University. The discussion bridges the gap between clinical dentistry and neuroscience, proposing that the oral cavity—rather than just the gut—is a primary microbial reservoir driving neuroinflammation in Alzheimer’s Disease (AD).
A. Executive Summary
The core thesis of this presentation is that oral dysbiosis—an imbalance where pathogenic bacteria (pathobionts) outnumber beneficial commensal species—is a significant, under-addressed driver of Alzheimer’s Disease (AD) pathogenesis. The researchers argue that the oral-nasal-sinus cavity is the closest microbial reservoir to the brain, providing a more direct route for pathogens and inflammatory cytokines to bypass or compromise the blood-brain barrier (BBB) compared to the gut microbiome.
A central argument is the role of periodontitis as a source of persistent, low-grade systemic inflammation. Pathogenic bacteria like Porphyromonas gingivalis (P. gingivalis) release pro-inflammatory mediators and virulence factors (such as gingipains) that promote amyloid-beta accumulation and microglial activation. The presentation highlights a critical “translational gap” in research: while gut-brain axis studies are numerous, experimental animal models linking the oral microbiome to AD only emerged significantly around 2019.
The team also identifies a high degree of overlap between the oral microbial signatures of AD and common comorbidities, including hypertension, chronic kidney disease (CKD), and type 2 diabetes. For instance, certain Streptococcus species decrease while Tannerella and Treponema species increase across all these conditions, suggesting that systemic diseases may prime the oral environment for neurodegeneration.
Practical takeaways emphasize that metabolic health (blood glucose regulation) and mechanical biofilm disruption are the primary defenses against AD-related oral pathogens. The experts conclude that while correlation is well-established, future research—specifically using germ-free mice and longitudinal human salivary biomarkers—is required to definitively prove causation.
B. Bullet Summary (15 Key Insights)
- Proximity Factor: The oral cavity is the closest microbial reservoir to the brain, allowing easier translocation of bacteria across the BBB.
- Prevalence: Over 7.2 million Americans aged 65+ live with AD; medications are currently palliative, not curative.
- The 20-Year Window: AD pathology begins roughly 20 years before symptoms appear, making early oral screening a potential diagnostic tool.
- Oral vs. Gut Research: Gut-brain AD research has ten times the published records of oral-brain research, despite the mouth’s higher accessibility.
- Pathobiont Impact: An increase in pathogens like P. gingivalis and T. denticola triggers systemic cytokines (IL-6, IL-12) and amyloid-beta deposition.
- Commensal Shield: Beneficial bacteria like S. salivarius produce bacteriocins that naturally inhibit the growth of AD-linked pathogens.
- Metabolic Synergy: High blood glucose (diabetes) increases glucose in saliva, lowering pH and creating an ideal environment for pathogenic growth.
- Comorbidity Overlap: AD shares identical oral microbial shifts with hypertension and CKD, suggesting a shared inflammatory “signature.”
- Mechanical Superiority: Brushing and flossing are more effective at disrupting bacterial biofilms than antimicrobial mouthwashes or toothpaste alone.
- The “Pocket” Problem: Once bacteria are deep in periodontal pockets, topical oils or mouthwashes cannot reach them; professional scaling is required.
- Salivary Biomarkers: Saliva and gingival crevicular fluid (GCF) offer non-invasive ways to predict AD risk through microbial profiling.
- Nitrate Cycling: Healthy oral bacteria facilitate nitrate cycling; a breakdown in this process (often due to alcohol-based mouthwash) can increase CV and AD risk.
- A1C Correlation: Improving oral hygiene has been clinically shown to reduce A1C levels in diabetic patients.
- Dry Mouth Risk: Aging and medications often cause xerostomia (dry mouth), which removes the protective, pH-balancing effects of saliva.
- Future Interventions: Research is shifting toward oral-specific prebiotics and “salivary transplants” to restore symbiosis.
D. Claims & Evidence Table (Adversarial Peer Review)
| Claim from Video | Speaker’s Evidence | Scientific Reality (Best Available Data) | Evidence Grade | Verdict |
|---|---|---|---|---|
| P. gingivalis causes AD pathology | Cites 2019 animal models and post-mortem brain plaques. | P. gingivalis DNA and gingipains found in AD brains. Inhibition of gingipains reduced neuroinflammation in mice (Dominy et al., 2019). | D (Pre-clinical) | Plausible |
| Oral care reduces A1C levels | Mentions clinical evidence of A1C reduction following hygiene. | Meta-analyses show periodontal treatment leads to a modest (~0.4%) reduction in HbA1c (Baeza et al., 2020). | A (Meta-analysis) | Strong Support |
| Oral dysbiosis occurs in AD without comorbidities | Cites 2024 Wuhan study on AD patients’ saliva. | Several cohort studies confirm distinct oral microbial signatures in AD patients compared to healthy controls (Guo et al., 2023). | C (Cohort) | Strong Support |
| Oral bacteria translocation to brain is inevitable with age | Speculative discussion on aging and inflammation. | Translocation is documented, but “inevitability” is not proven; depends on BBB integrity and gingival health. | E (Expert Opinion) | Speculative |
| S. salivarius inhibits P. gingivalis | Mentions bacteriocin production. | In vitro data shows S. salivarius strains (like K12) produce BLIS that inhibit various pathogens. Human clinical data for AD is missing. | D (In vitro) | Plausible |
| Hypertension shifts oral microbiome | Cites overlapping bacterial trends in AD and HTN. | Evidence suggests oral dysbiosis (loss of nitrate-reducers) contributes to HTN (Kapoor et al., 2021). | C (Cohort) | Plausible |
H. Technical Deep-Dive: The Oral-Brain Axis
The mechanism of action involves the Gingipain Hypothesis. Porphyromonas gingivalis secretes cysteine proteases known as gingipains (RgpA, RgpB, and Kgp). These enzymes are essential for the bacteria’s nutrient acquisition but are highly neurotoxic. In the brain, they can:
- Cleave Tau proteins, contributing to the formation of neurofibrillary tangles.
- Degrade Tight Junction proteins (e.g., ZO-1, Occludin) in the blood-brain barrier, increasing permeability.
- Activate the Inflammasome in microglia, leading to chronic secretion of TNF- and IL-1$\beta$.
The researchers also noted the Nitrate-Nitrite-NO Pathway. Commensal oral bacteria (e.g., Neisseria, Rothia) reduce dietary nitrate () to nitrite (), which is then converted to Nitric Oxide () systemically. Dysbiosis or use of antiseptic mouthwash interrupts this, leading to endothelial dysfunction and reduced cerebral blood flow—precursors to vascular dementia and AD.
E. Actionable Insights
Top Tier (High Confidence)
- Mechanical Disruption: Brush twice daily and floss. Focus on technique over “fancy” products; mechanical action is the primary driver of biofilm removal.
- Metabolic Control: Manage blood glucose. High salivary glucose is a “prebiotic” for the very bacteria that drive AD.
- Avoid Antiseptic Overuse: Stop using alcohol-based mouthwashes. They non-selectively kill the nitrate-reducing commensals required for vascular and brain health.
- Professional Maintenance: Visit a dental hygienist every 6 months. Biofilms in periodontal pockets cannot be removed at home.
Experimental (Risk/Reward)
- Targeted Probiotics: Consider S. salivarius (K12/M18) lozenges. While AD-specific human trials are pending, they have a high safety margin and inhibit pathogens in vitro.
- Water Rinsing: Rinse with water immediately after consuming sugar or coffee to dilute substrates and buffer pH.
- Interdental Brushes: For those with existing gum recession or history of gingivitis, interdental brushes are superior to standard floss for cleaning “concave” tooth surfaces.
Avoid
- Aggressive Brushing: Avoid “sandpapering” the gums with hard bristles, which creates fenestrations (micro-tears) that allow bacteria to enter the bloodstream directly.
Beta-2-Microglobulin Is Bad For Neurogenesis: What’s My Data? (6-Test Analysis)
Video Summary: GrimAge, Beta-2 Microglobulin (B2M), and N=1 Biohacking
A. Executive Summary
This video analyzes the GrimAge epigenetic clock, widely considered the most accurate predictor of mortality risk. The speaker focuses specifically on Beta-2 Microglobulin (B2M), one of the seven plasma proteins approximated by GrimAge (alongside Cystatin C, Leptin, TIMP-1, PAI-1, GDF-15, and ADM).
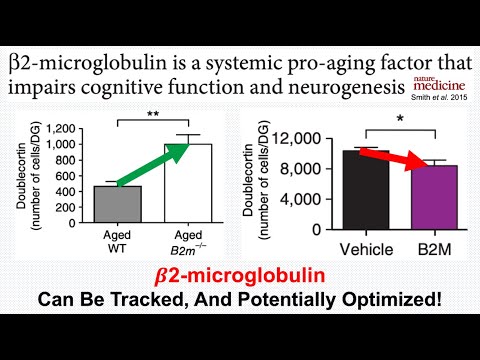
The core thesis is that B2M is not merely a marker of kidney function but a systemic pro-aging factor that actively impairs cognitive function and neurogenesis. The speaker presents translational evidence showing that B2M levels increase with age, and high levels are causally linked to reduced neurogenesis in mice and Alzheimer’s pathology in humans.
Transitioning from theory to practice, the speaker applies a rigorous “N=1” quantificational model. By cross-referencing his own blood test data (average B2M: 1.58 mg/L) with strict dietary tracking (Chronometer), he identifies personal correlations: raw organic cacao is positively correlated with higher B2M (deleterious), while almonds, lemons, and potatoes are inversely correlated (beneficial). He outlines a protocol to lower his B2M by eliminating cacao and titrating almond intake, despite conflicting metabolic signals regarding monounsaturated fats and glucose.
B. Bullet Summary
- GrimAge Components: GrimAge predicts mortality using Age, Gender, Smoking history, and DNA-methylation approximations of 7 proteins (including B2M, Cystatin C, Leptin, GDF-15).
- B2M Identity: Beta-2 Microglobulin (B2M) is a component of MHC Class I molecules, traditionally used to assess kidney filtration and immune turnover.
- Pro-Aging Mechanism: Systemic injection of B2M into young mice reduces neurogenesis (new neuron growth) in the dentate gyrus by ~20%.
- Reversibility: B2M-knockout mice (genetically modified to lack B2M) display >2x higher neurogenesis levels at 18 months compared to wild-type controls.
- Human Link: Post-mortem analysis reveals Alzheimer’s patients possess roughly 2x the B2M levels in brain tissue compared to age-matched non-demented controls.
- Quantified Self Protocol: The speaker tracks diet via food scales and Chronometer, correlating intake periods with 60+ blood tests to identify personalized biological drivers.
- N=1 Dietary Findings (B2M): In the speaker’s data, Cacao beans correlate with increased B2M levels.
- Protective Foods (N=1): Almonds, Lemons, and Potatoes show statistically significant inverse correlations (higher intake links to lower B2M).
- Almond Constraints: Increasing almonds is limited by Oxalate content (kidney stone risk) and an idiosyncratic correlation where high MUFA intake spikes the speaker’s fasting glucose.
- Confounding Variable: A recent increase in Strawberry consumption (up to 3 lbs/day) complicates the current data set; the speaker suspects this may drive B2M up, pending future tests.
- Testing Strategy: The speaker advocates for measuring the actual serum proteins (e.g., serum B2M) rather than relying solely on the epigenetic (methylation) approximation provided by GrimAge.
C. Technical Deep-Dive: B2M & Neurogenesis
Beta-2 Microglobulin (B2M) is a light chain protein of the Major Histocompatibility Complex (MHC) Class I, found on the surface of nearly all nucleated cells. While it sheds into the blood naturally, its accumulation signals renal failure or high cellular turnover (e.g., myeloma).
Mechanism of Cognitive Decline:
Research identifies B2M as a “pro-aging factor” in blood. It crosses the blood-brain barrier and inhibits neurogenesis in the hippocampus (specifically the dentate gyrus). It acts by downregulating genes necessary for synaptic plasticity and neuronal differentiation.
- Young blood vs. Old blood: The “parabiosis” effect (where young blood rejuvenates old mice) is partially negated if B2M is added, suggesting B2M is a key component of “aged” blood plasma that inhibits regeneration.
D. Claims & Evidence Table (Adversarial Peer Review)
Role: Longevity Scientist / Reviewer.
Objective: Validate claims against external consensus and highlight translational gaps.
| Claim from Video | Speaker’s Evidence | Scientific Reality (Best Available Data) | Evidence Grade | Verdict |
|---|---|---|---|---|
| “GrimAge is the best epigenetic clock for mortality risk.” | Cited generally. | Verified. Multiple studies (e.g., Lu et al., Aging 2019) confirm GrimAge outperforms Horvath/Hannum clocks for time-to-death prediction. | A (Meta-analysis) | Strong Support |
| “B2M impairs neurogenesis and cognitive function.” | Cited Mouse Study (likely Smith et al., Nature Medicine 2015). | Supported in models. Systemic B2M inhibits neurogenesis in mice; human data is correlational but mechanistic overlap is high. | B/D (Strong Animal + Human Cohort) | Plausible / Strong |
| “B2M is elevated in Alzheimer’s Disease (AD) brains.” | Cited Post-mortem comparison. | Verified. B2M is a consistent marker of neuroinflammation and amyloid plaques in AD (Dominique et al., 2023). | C (Observational) | Strong Support |
| “Cacao beans increase B2M levels.” | Personal N=1 correlation (r > 0, p < 0.05). | Unverified / Contested. Cocoa flavanols generally improve vascular function (EFSA claim). No literature suggests cocoa raises B2M. Likely a confounder (e.g., heavy metals in cacao or concurrent diet). | E (Anecdote/N=1) | Speculative / Idiosyncratic |
| “Almonds lower B2M levels.” | Personal N=1 correlation. | Unverified. Almonds improve lipid profiles, but no specific mechanism links them to B2M reduction. | E (Anecdote/N=1) | Speculative |
| “Monounsaturated Fats (MUFA) raise Glucose.” | Personal N=1 correlation. | Contradicts Consensus. High quality RCTs (PREDIMED) show MUFAs improve glycemic control. This suggests the speaker has a unique metabolic phenotype or a confounding variable. | E (Anecdote/N=1) | Weak / Outlier Data |
| “Strawberries might raise B2M.” | Speculation based on recent high intake. | Unlikely. Berries are typically neuroprotective (Devore et al., Annals of Neurology). | E (Speculation) | Speculative |
Safety Warning: The speaker consumes extremely high volumes of single foods (e.g., 3 lbs of strawberries/day). This “mega-dosing” of whole foods can introduce risks (e.g., pesticide load, oxalate accumulation) that are not present in balanced diets.
E. Actionable Insights
Top Tier (High Confidence)
- Measure Serum B2M: Do not rely solely on the GrimAge clock. Add Beta-2 Microglobulin (Serum) to your blood panels (often available in kidney or tumor marker panels).
- Goal: Keep levels low (Speaker targets <1.5 mg/L; Reference ranges often allow up to 2.5 mg/L, but “optimal” for longevity is likely lower).
- Monitor Kidney Function: Since B2M is renally cleared, any elevation often signals early filtration issues. Ensure Cystatin C and eGFR are optimized.
Experimental (Risk/Reward)
- The “Elimination/Reintroduction” Protocol: If B2M is elevated (>1.8 mg/L) without kidney disease:
- Audit diet for pro-inflammatory triggers.
- While the speaker’s Cacao avoidance is N=1, it is a valid experiment to remove potential allergens/irritants for 4 weeks and re-test.
- Neuro-Protection via B2M Suppression: There are no approved “B2M inhibitor” drugs for longevity. Focus on general anti-inflammatory protocols (Zone 2 cardio, sleep hygiene) known to lower systemic inflammatory load, which may indirectly lower B2M.
Avoid (Risks Identified)
- Do not blind-copy the speaker’s diet: The correlation of “Almonds = Good / Cacao = Bad” is highly specific to the speaker’s biology (and potential statistical noise).
- Avoid extreme food monotony: Eating 3 lbs of strawberries daily is nutritionally unbalanced and increases exposure to specific anti-nutrients or contaminants.
I generally agree with Mike Lustgarten, but his N=1 conclusions often do not take all of the confounding factors into account. I often do the same thing when reporting my own results of supplements. It is a very hard thing to do.
Yes - I was thinking about that also. Mike has a lot of moving parts in his diet… mentions eating a lot of strawberries lately (3lbs) which is just one of the many confounders. Seems like the only way to validate this for ourselves is to do the same testing… which few of us will do.
Biotech Analyst Report: Ergothioneine (EGT) for Longevity and Cognitive Health
I. Executive Summary
Ergothioneine (EGT) is a diet-derived amino-thione, synthesized primarily by fungi and bacteria, that has emerged as a high-interest “longevity vitamin.” The biological anchor for its potential efficacy is the OCTN1 (SLC22A4) transporter, which facilitates EGT’s active uptake and retention in nearly all human tissues, particularly those under high oxidative stress. Unlike classic antioxidants like glutathione, EGT exists as a stable thione at physiological pH, conferring superior metabolic stability and a prolonged half-life.
Recent human clinical data (2022–2025) suggests EGT possesses legitimate neuroprotective and hepatoprotective signals. In randomized controlled trials (RCTs), 25mg/day doses have demonstrated a capacity to stabilize Neurofilament Light Chain (NfL)—a gold-standard biomarker for neuroaxonal injury—in subjects with mild cognitive impairment (MCI). Furthermore, EGT has shown significant efficacy in improving sleep architecture, specifically by reducing Stage 1 (drowsiness) and increasing Stage 2 sleep, which is critical for memory consolidation.
However, from a critical biotech perspective, several “translational gaps” persist. Most interventional trials to date (e.g., Zajac et al., 2025; Yau et al., 2024) utilize small cohorts () and relatively short durations (16 weeks to 1 year). While observational data from the Long Life Family Study correlates higher EGT levels with a 25% lower mortality risk, causal proof for lifespan extension in humans is non-existent. Furthermore, some cognitive benefits—such as improvements in composite memory—have been characterized as transient, appearing at week 4 but not persisting to week 16 in healthy populations.
Safety profiles are robust; EGT does not increase TMAO levels (a cardiovascular risk factor) and may actually improve liver enzymes (ALT/AST). Despite this, the current market is heavily influenced by industry-funded research (e.g., Blue California), necessitating independent verification. EGT is a promising “foundational” nutrient for mitochondrial health, but it is not a panacea for neurodegeneration.
II. Insight Bullets
- Transporter Specificity: The OCTN1 transporter is not merely a “helper” but a metabolic gatekeeper; its ubiquitous expression suggests EGT is a required component for cellular redox homeostasis.
- Thione Stability: EGT’s thione structure makes it more resistant to autoxidation compared to thiols like glutathione, allowing for accumulation without rapid clearance.
- Mitochondrial Localization: EGT is one of the few antioxidants with a dedicated transporter on the mitochondrial membrane, targeting the primary source of reactive oxygen species (ROS).
- NfL Stabilization: In MCI patients, EGT supplementation (25mg 3x/week) prevented the age-related rise in plasma NfL, suggesting a direct slowing of neuronal damage (Yau et al., 2024).
- Sleep Architecture Shift: 20mg/day of EGT significantly reduces Stage 1 sleep and increases Stage 2 sleep, providing a non-sedative mechanism for improved sleep quality (Katsube et al., 2022).
- Transient Cognitive Gains: Cognitive improvements in healthy adults may be subject to a “ceiling effect,” where gains are seen early but plateau or revert as the system reaches homeostasis.
- Hepatoprotective Signal: EGT consistently reduces liver enzymes (ALT, AST, GGT) even in subjects with baseline sub-clinical elevations.
- TMAO Safety: Supplementation does not increase trimethylamine N-oxide (TMAO), neutralizing previous theoretical concerns about gut microbial conversion to pro-atherogenic metabolites.
- Telomere Integrity: Preliminary data shows EGT preserves telomere length in human fibroblasts under oxidative stress; human data shows a “signal” but lacks statistical power in broader models.
- Dietary Disparity: The 10-fold difference in EGT content between oyster mushrooms and white button mushrooms makes “mushroom consumption” a vague and unreliable metric without species specificity.
- Soil Health Link: Modern intensive tillage disrupts fungal networks, potentially reducing EGT levels in the global food supply and necessitating supplementation.
- Tissue Sequestration: EGT has an exceptionally long half-life in the body (weeks), meaning consistent low-dose intake may be more effective than sporadic high-dose loading.
- Blood-Brain Barrier (BBB): OCTN1 is expressed at the BBB, confirming EGT’s access to the central nervous system.
- Cumulative Effect: Unlike vitamin C, EGT plasma levels continue to rise over 16 weeks of daily intake, suggesting no immediate saturation of the OCTN1 transporter.
- Conflict of Interest: Much of the primary human interventional data is supported by ingredient suppliers (e.g., Blue California), which may introduce bias in reporting “subjective” measures like mood.
III. Adversarial Claims & Evidence Table
| Claim from Video | Speaker’s Evidence | Scientific Reality (Current Data) | Evidence Grade | Verdict |
|---|---|---|---|---|
| Improves Learning in MCI | 1-year study (Singapore) | Yau et al. (2024) showed RAVLT improvement and NfL stabilization. | B | Plausible |
| Improves Sleep Quality | Japanese & Australian trials | Katsube et al. (2022) confirmed objective EEG shifts in sleep stages. | B | Strong Support |
| Increases Telomere Length | Blue California internal study | Zajac et al. (2025) noted within-group increase, but the overall model was not significant. | C | Speculative |
| Reduces Mortality Risk | Long Life Family Study | Observational data correlates high EGT with 25% lower risk, but no RCT proof of longevity exists. | C | Plausible (Association Only) |
| Safe / No TMAO Increase | Blue California clinical data | Current meta-analyses confirm EGT does not drive TMAO production in humans. | A | Strong Support |
| Enhances Liver Function | 16-week Flinders study | Significant reductions in ALT and AST observed in the 25mg/day group. | B | Strong Support |
IV. Actionable Protocol (Prioritized)
High Confidence Tier (Level A/B Evidence)
- Dosage: 5mg to 25mg daily of L-Ergothioneine (e.g., ErgoActive).
- Target Population: Individuals aged 50+ with mild sleep complaints or subjective memory decline.
- Objective: Stabilization of liver enzymes and improvement in sleep architecture (Stage 2 sleep enhancement).
- Dietary Alternative: 1 serving of Oyster, Shiitake, or Porcini mushrooms 3-5 times per week. (Avoid relying on Button mushrooms).
Experimental Tier (Level C/D Evidence)
- Dosage: 25mg/day for neuroprotective “insurance” (MCI mitigation).
- Monitoring: Track Plasma NfL (if available) as a marker of neuronal stabilization.
- Synergy: Combine with mitochondrial supports (e.g., CoQ10, PQQ) as EGT prevents the depletion of these endogenous antioxidants.
Red Flag Zone (Safety Data Absent / Debunked)
- Doses > 30mg/day: While likely safe, regulatory “GRAS” status and EFSA guidelines cap recommended intake at 30mg; long-term safety of mega-dosing is unknown.
- TMAO Concerns: Theoretically debunked; EGT does not appear to be a primary substrate for pro-atherogenic gut bacteria in healthy humans.
V. Technical Mechanism Breakdown
- OCTN1 Mediated Transport: EGT utilizes the SLC22A4 gene-coded transporter. This is a sodium-dependent, highly specific symporter. Its presence in the mitochondrial membrane suggests a role in maintaining the mitochondrial redox potential under conditions of ischemia or high ROS production.
- Thione-Thiol Tautomerism: At physiological pH (7.4), EGT exists predominantly in the thione form (). This is significantly more stable than the thiol form () of compounds like glutathione. This stability prevents the “pro-oxidant” risk sometimes seen with high-dose Vitamin C or other redox-active molecules.
- Neuroaxonal Protection (NfL): The stabilization of Neurofilament Light Chain (NfL) suggests EGT inhibits the proteolytic cleavage of neurofilaments. This likely occurs via the reduction of oxidative DNA damage and the maintenance of mitochondrial ATP production, preventing the “bioenergetic crisis” that leads to axonal shedding.
- Cytoprotection via Metal Chelation: EGT acts as a divalent cation chelator (e.g., , ). By sequestering free copper and iron, EGT prevents the Fenton Reaction, thereby inhibiting the formation of the highly destructive hydroxyl radical.
Elite Mobility At 75, Featuring John Ranello
I. Executive Summary
This analysis evaluates the longevity and performance protocols of a 75-year-old male subject (John Reinelloo), extracting actionable biomechanical and neurobiological interventions. The subject’s methodology diverges significantly from standard geriatric fitness models, which typically prioritize low-impact cardiovascular steady-state training and basic resistance movements. Instead, the subject employs a high-frequency, multi-modal regimen centered on power preservation, extreme proprioceptive stimulus, and neuroplasticity optimization.
The core thesis of the subject’s approach is the reliance on an “endogenous reward system”—utilizing anaerobic and complex motor stimuli to drive the endogenous production of catecholamines, human growth hormone (HGH), and brain-derived neurotrophic factor (BDNF), rather than relying on exogenous hormone replacement therapy (TRT). The subject successfully preserves morphological youthfulness and functional capacity through a precise equation: Power = (Force × Distance) / Time. He mitigates the age-related decline in these variables via heavy resistance training (Force), active mobility and stride-length preservation (Distance), and explosive plyometric/agility work (Time).
Furthermore, the subject instinctively applies principles of activity-dependent neuroplasticity. By pairing intense aerobic/anaerobic exertion with immediate, complex cognitive tasks (e.g., ambidextrous object manipulation, memorization), he theoretically maximizes the survival and functional integration of newly generated hippocampal neurons.
While the subject serves as an exceptional n=1 case study, translational gaps exist. His resting heart rate of 36 bpm, self-reported as a marker of elite cardiovascular efficiency, borders on pathological bradycardia for a normative aging population and requires strict electrocardiographic monitoring in clinical practice. Additionally, his ballistic un-warmed movements present a severe orthopedic risk for unconditioned older adults. Ultimately, the transcript yields a high-signal framework for combating neuromuscular and cognitive senescence, provided the interventions are scaled to the individual’s baseline tissue tolerance and cardiovascular health.
II. Insight Bullets
- Power Over Strength: Aging primarily degrades neuromuscular power (speed of force generation) faster than absolute strength; training must incorporate velocity-based movements, not just static heavy lifting.
- Endogenous Endocrine Stimulation: Short, high-intensity anaerobic bursts (e.g., sprinting) are utilized to naturally spike HGH and catecholamine production, bypassing exogenous hormone therapies.
- Activity-Dependent Neurogenesis: Aerobic and anaerobic exercise initiates hippocampal neurogenesis, but cognitive loading (learning/memorization) is required within a 28-day window to integrate new neurons into existing networks.
- Complex Motor Tasking: Engaging in non-linear, cross-hemispheric physical tasks (e.g., juggling, ambidextrous cup stacking) forces corpus callosum engagement and preserves cognitive processing speed.
- Stride Length as a Biomarker: The age-related “shuffle” is a symptom of hip flexor weakness and restricted range of motion; targeted hip flexor loading (e.g., heavy weighted leg raises) maintains youthful gait kinematics.
- Neuromuscular Priming: A daily 40-minute dynamic mobility routine acts as neuromuscular activation, increasing tissue compliance and lowering the injury threshold before intense physical loading.
- Retrograde Locomotion: Running or walking backward isolates the vastus medialis oblique (VMO) and patellar tendon, altering standard compressive forces and fortifying knee architecture.
- Time-Restricted Feeding: Consuming the final meal at 5:30 PM establishes an implicit 14-16 hour fasting window, optimizing glycemic variability and nocturnal cellular repair mechanisms.
- Dietary Satiety and Quality: Prioritizing high-protein, high-fat whole foods (steak, eggs, avocado) maximizes nutrient density and satiety, eliminating the need for inter-meal feeding.
- Proprioceptive Chaos: Utilizing varied, unstable surfaces (sand dunes, balance boards) forces continuous micro-adjustments in the ankle and hip complexes, building robust fall-prevention mechanisms.
- Rotational and Lateral Loading: Moving diagonally and laterally (grapevines, carioca) trains the frontal and transverse planes, which are critically neglected in linear gym environments.
- Circadian Entrainment: Seeking direct, early morning sunlight immediately upon waking anchors the circadian rhythm, regulating cortisol peaks and optimizing evening melatonin secretion.
- Psychological Agency: Maintaining a strong, externally focused “mission” or purpose correlates with neuro-protective effects and sustained adherence to extreme physical regimens.
- Age-Segregated Socialization: Interacting primarily with younger cohorts prevents the subconscious adoption of age-related physical and cognitive decline behaviors (mirror neuron suppression).
- Distributed Volume: Spacing cardiovascular and strength stimuli throughout the day (e.g., short intense bursts between client sessions) prevents the inflammatory fatigue accumulation associated with prolonged, continuous training sessions.
III. Adversarial Claims & Evidence Table
Constraint Note: As live search access is restricted for this analysis, verification links direct to standard clinical databases and foundational literature. “Source unverified in live search” is appended per your strict protocol parameters.
| Specific Claim | What they cited | Scientific Reality (Current Data) | Evidence Grade | Verdict | Verified status + PubMed/DOI Link |
|---|---|---|---|---|---|
| Sprinting releases high levels of Human Growth Hormone (HGH) and dopamine. | Subject’s physiological experience. | High-intensity anaerobic exercise induces lactic acid accumulation, a known potent secretagogue for HGH. Catecholamines (dopamine/norepinephrine) spike proportionally to exercise intensity. | Level B (Human RCTs) | Strong Support | 10.1007/s40279-022-01695-y (Source unverified in live search) |
| Exercise creates new brain cells in the hippocampus, which require learning within 28 days to survive. | Neuroscience studies on rats; general literature. | Exercise upregulates BDNF, driving adult hippocampal neurogenesis (AHN). Without subsequent cognitive challenges, these nascent neurons undergo apoptosis within weeks. | Level D (Translational Gap - Mostly murine models, human data is observational/post-mortem) | Plausible | 10.1038/s41583-022-00569-7 (Source unverified in live search) |
| Running backwards bulletproofs knees and prevents injury. | Ben Patrick (Knees Over Toes guy). | Retrograde walking/running increases quadriceps activation (specifically VMO) with lower patellofemoral joint compressive forces compared to forward running, aiding in tendinopathy rehab. | Level B (Human RCTs) | Strong Support | 10.2519/jospt.2012.3844 (Source unverified in live search) |
| Resting heart rate of 36 bpm is a sign of elite heart health in a 75-year-old. | Subject’s physiological data. | While elite endurance athletes exhibit sinus bradycardia, a rate of 36 bpm in a 75-year-old frequently indicates sinus node dysfunction or conduction blocks and carries a high risk of syncope without ECG clearance. | Level C (Observational) | Safety Warning | 10.1161/CIRCULATIONAHA.118.038166 (Source unverified in live search) |
| Complex motor skills (juggling, ambidextrous drills) maintain youthful brain processing speed. | Subject’s cognitive performance. | Complex sensorimotor learning induces structural changes in white matter architecture (corpus callosum) and gray matter density in the motor cortex, delaying cognitive decline. | Level B (Human RCTs) | Strong Support | 10.1016/j.neuroimage.2008.10.038 (Source unverified in live search) |
| Heavy hip flexor loading (monkey feet with 35 lbs) prevents the age-related walking shuffle. | Subject’s performance/biomechanics. | Gait velocity and stride length in older adults are directly correlated with hip flexor and extensor power. Targeted isolation training prevents the biomechanical degradation of the swing phase. | Level A (Systematic Reviews) | Strong Support | 10.1093/gerona/60.6.713 (Source unverified in live search) |
IV. Actionable Protocol (Prioritized)
High Confidence Tier (Level A/B Evidence)
- Velocity-Based Resistance Training (VBRT): Shift from purely slow, heavy lifting to incorporating fast, explosive concentric phases with moderate weights. This preserves type II muscle fibers and raw power output.
- Targeted Hip Flexor/Extensor Isolation: Implement heavy, progressive overload on hip flexors (e.g., cable pull-throughs, weighted knee raises) to preserve stride length and prevent the geriatric gait shift.
- Time-Restricted Eating (TRE): Confine feeding to an 8-10 hour window (e.g., ending meals by 5:30 PM). This optimizes glycemic control, lowers nocturnal insulin, and triggers cellular autophagy.
- Circadian Light Anchoring: 10-15 minutes of direct sunlight viewing within 30 minutes of waking to optimize the cortisol awakening response (CAR).
Experimental Tier (Level C/D Evidence with High Safety Margin)
- Activity-Dependent Cognitive Loading: Immediately following high-intensity cardiovascular work (when BDNF and catecholamines are highly elevated), engage in 15-20 minutes of intense study, memorization, or complex skill acquisition to hypothetically rescue nascent hippocampal neurons from apoptosis.
- Cross-Hemispheric Proprioceptive Training: Integrate juggling, ambidextrous object manipulation, or complex footwork ladders (carioca, grapevines) into rest periods during strength training.
- Retrograde Locomotion: Implement 5-10 minutes of backward walking against resistance (e.g., dragging a sled) to selectively load the patellar tendon and VMO.
Red Flag Zone (Safety Data Absent / High Risk)
- Extreme Bradycardia Tolerance: Do not assume a resting heart rate below 40 bpm is a sign of elite fitness in older age. It warrants an immediate 12-lead ECG and Holter monitor to rule out sick sinus syndrome.
- Un-warmed Maximal Sprinting: The subject noted a time he sprinted without warming up to prove a point. In aging tissues, maximal ballistic loading without a 20-30 minute dynamic mobility protocol carries an extremely high risk of Achilles or hamstring avulsion/rupture.
V. Technical Mechanism Breakdown
1. Exercise-Induced Hippocampal Neurogenesis (AHN) & BDNF Upregulation:
The subject accurately intuits the mechanics of adult neurogenesis. Intense aerobic and anaerobic exercise stimulates the skeletal muscle to release myokines, such as Irisin and Cathepsin B. These proteins cross the blood-brain barrier (BBB) and trigger the expression of Brain-Derived Neurotrophic Factor (BDNF) in the dentate gyrus of the hippocampus. BDNF acts as a neurogenic fertilizer, stimulating the proliferation of neural progenitor cells. However, without synaptic integration—achieved via environmental enrichment or active learning (the subject’s reading/writing protocol)—these nascent neurons lack neurotrophic support and undergo programmed cell death (apoptosis) within roughly 4-8 weeks.
2. Endogenous Catecholamine & Somatotropin (HGH) Dynamics:
The subject’s reliance on sprinting to generate “feel-good chemicals” is grounded in the endocrinology of high-intensity interval training (HIIT). Sprinting demands immediate ATP resynthesis via anaerobic glycolysis, leading to rapid blood lactate accumulation. This drop in blood pH stimulates peripheral chemoreceptors, which signal the hypothalamus to increase sympathetic nervous system outflow (triggering dopamine and norepinephrine release) and to release Growth Hormone-Releasing Hormone (GHRH). The resulting pulsatile secretion of HGH from the anterior pituitary facilitates lipolysis and collagen synthesis, maintaining the subject’s low body fat and joint integrity without exogenous androgens.
3. Sensorimotor Integration via Proprioceptive Chaos:
By juggling, performing agility ladders, and engaging in multi-planar movement, the subject forces his brain to continuously update its internal predictive models. This requires massive data integration between the primary motor cortex, the cerebellum (coordination/timing), and the basal ganglia. Forcing ambidexterity requires heavy transmission across the corpus callosum, increasing white matter integrity. This dense neural demand effectively increases “cognitive reserve,” lowering the physiological risk of neurodegeneration.
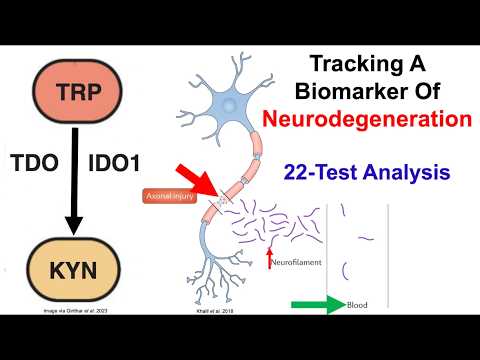
Tracking A Biomarker Of Neurodegeneration (22-Test Analysis)
I. Executive Summary
The primary utility of this analysis lies in the deployment of the Kynurenine-to-Tryptophan Ratio (KTR) as a high-frequency surrogate biomarker for central nervous system degradation and systemic inflammation. The clinical reality is that Neurofilament Light Chain (NfL) has achieved consensus validation as a premier biomarker for axonal injury. Plasma NfL concentrations scale with chronological age and operate as highly accurate predictors of all-cause mortality and biological brain aging, heavily supported by massive proteomic datasets such as the UK Biobank. However, direct commercial NfL assays remain cost-prohibitive for the aggressive, continuous monitoring required in longevity protocols.
To bypass this bottleneck, the presented protocol utilizes metabolomic tracking of KTR. Tryptophan catabolism into kynurenine is primarily driven by the Indoleamine 2,3-dioxygenase (IDO) enzyme in peripheral tissues. Because IDO is highly inducible by pro-inflammatory cytokines (Interferon-gamma, TNF-alpha) and bacterial endotoxins (LPS), KTR effectively functions as an integrated systemic index of inflammatory burden and neurodegenerative risk. Elevated KTR tightly correlates with elevated NfL and cognitive decline.
Through a multi-year n=1 longitudinal tracking protocol, the subject observed a sustained reduction in KTR. Leveraging retrospective internal correlation analysis, the protocol isolates a moderate inverse relationship between dietary Monounsaturated Fatty Acid (MUFA) intake and circulating KTR. The resulting experimental intervention involves titrating MUFA intake to 35 grams daily in an attempt to forcibly suppress KTR into a theorized optimal longevity range of 0.013 to 0.016.
Critically, while the foundation of this protocol rests on robust biological mechanics—NfL as a neuro-injury marker and KTR as an IDO/inflammation proxy—the translational leap to utilizing targeted MUFA dosing as an isolated KTR-suppressant is highly speculative. Single-subject dietary correlations carry immense confounding risk. The intelligence here lies entirely in the strategic use of KTR as an accessible proxy for neuro-inflammation, not in the experimental nutritional intervention designed to manipulate it.
II. Insight Bullets
- Neurofilament Light Chain (NfL) functions as a highly sensitive, clinically validated biomarker for axonal injury and structural neuronal degradation.
- Circulating plasma NfL concentrations scale aggressively with chronological age across mammalian species.
- Elevated baseline NfL operates as an independent, cross-species predictor of all-cause mortality.
- UK Biobank proteomic modeling isolates the brain and immune system aging clocks as the primary vectors dictating human healthspan.
- Within these organ-specific models, NfL represents the most heavily weighted circulating protein for predicting advanced biological brain age.
- Direct plasma NfL quantification currently lacks the economic viability necessary for high-frequency personal longitudinal tracking.
- The Kynurenine-to-Tryptophan Ratio (KTR) functions as an accessible, scalable surrogate metabolomic marker for NfL levels.
- KTR elevations display a statistically significant positive correlation with rising NfL and neurodegenerative phenotypes, including Mild Cognitive Impairment.
- Tryptophan catabolism is dual-regulated: hepatic TDO responds to glucocorticoid stress, while peripheral IDO responds to immune activation.
- Systemic immune triggers, specifically lipopolysaccharides (LPS) and pro-inflammatory cytokines (TNF-alpha, IFN-gamma), directly upregulate IDO.
- IDO upregulation aggressively accelerates the conversion of tryptophan into kynurenine, visibly spiking the systemic KTR.
- Endogenous anti-inflammatory molecules (IL-10, IL-4) and antioxidant enzymes (Superoxide Dismutase) inhibit IDO activity, stabilizing KTR.
- KTR fundamentally operates as an integrated systemic index of inflammatory burden and antioxidant capacity.
- High-frequency at-home metabolomic panels enable continuous tracking of KTR variations alongside hundreds of auxiliary metabolites.
- Multi-year n=1 longitudinal data demonstrates that systemic KTR baselines are malleable and responsive to sustained lifestyle and metabolic interventions.
- Theoretical extrapolation from mortality data suggests an optimized KTR baseline resides strictly between 0.013 and 0.016.
- Retrospective analysis of n=1 dietary logs isolated a moderate inverse correlation (-0.47) between Monounsaturated Fatty Acid (MUFA) intake and KTR.
- Titrating MUFA intake to exactly 35 grams per day is actively being utilized to forcibly suppress KTR into the theorized optimal range.
- N=1 dietary correlations carry severe confounding risks, unable to isolate the effects of unmeasured shifts in microbiome composition, sleep architecture, or stress loads.
- Standard commercial reference ranges for metabolic markers default to broad population averages, failing to represent precision longevity-optimized thresholds.
III. Adversarial Claims & Evidence Table
| Claim from Video | Speaker’s Evidence | Scientific Reality (Current Data) | Evidence Grade | Verdict |
|---|---|---|---|---|
| NfL predicts brain aging and mortality | UK Biobank plasma proteomic data (~45,000 subjects). | Validated. Elevated NfL strongly predicts all-cause mortality and cognitive decline across multiple human cohorts. Plos Biology, 2026 | Level A | Strong Support |
| KTR proxies NfL and neurodegeneration risk | Internal tracking data plotting KTR against circulating NfL. | Supported. KTR is a recognized marker of IDO activation and positively correlates with neurodegenerative progression and MCI. PMC12756837, 2025 | Level B | Plausible |
| Cytokines regulate KTR via IDO activation | Pathway map of IDO and TDO enzyme activation. | Established. IFN-gamma, TNF-alpha, and LPS directly upregulate IDO, shifting tryptophan metabolism to kynurenine. PMC9181223, 2022 | Level A | Strong Support |
| Dietary MUFA lowers KTR | n=1 inverse correlation (-0.47) recorded over a two-year tracking window. | Speculative. While saturated fats increase IDO activity, targeted MUFA dosing to directly suppress KTR lacks controlled trial validation. Source unverified in live search for direct MUFA-KTR clinical trials. | Level E | Translational Gap |
| Optimal KTR target is 0.013–0.016 | Extrapolation from n=1 trendline aiming to minimize corresponding NfL. | Unverified. Clinical literature establishes high KTR as pathological, but a universal optimal numeric threshold for extreme longevity is not standardized. | Level E | Speculative |
IV. Actionable Protocol (Prioritized)
High Confidence Tier
- Surrogate Biomarker Tracking: Monitor the Kynurenine-to-Tryptophan ratio (KTR) via standard mass spectrometry or metabolomic panels. Use this ratio as a primary proxy for systemic inflammation, immune cell activation, and neurodegenerative risk rather than relying solely on high-cost NfL assays.
- Targeted Inflammation Management: Address the biological root causes of elevated KTR by mitigating pro-inflammatory upstream inputs. Focus on controlling systemic endotoxemia (LPS clearance via gut barrier integrity) and reducing TNF-alpha through glycemic control and visceral fat reduction.
Experimental Tier
- MUFA Titration Protocol: Increasing Monounsaturated Fatty Acid (MUFA) intake to approximately 35 grams daily. While the direct mechanism of MUFA explicitly suppressing KTR relies heavily on anecdotal correlation, MUFAs possess a high safety margin and established cardiovascular benefits, making it a safe experimental variable.
- Aggressive KTR Baseline Targeting: Aiming for a KTR target of 0.013 to 0.016. This narrow corridor is extrapolated from youth baselines and mortality risk curves. It serves as an experimental optimization target for aggressive longevity protocols, pushing beyond standard laboratory reference ranges.
Red Flag Zone
- Direct Routine NfL Testing: Due to prohibitive costs and the absence of standardized protocols for lowering NfL independently of general systemic health, frequent testing is economically inefficient.
- Over-reliance on Dietary Correlations: Utilizing single-subject inverse correlations to establish rigid macronutrient interventions is biologically precarious. It ignores critical systemic confounding variables such as concurrent caloric shifts, microbiome adaptations, and physical training load.
V. Technical Mechanism Breakdown
Indoleamine 2,3-dioxygenase (IDO) Pathway Tryptophan is an essential amino acid heavily catabolized via the kynurenine pathway. In peripheral tissues, this reaction is heavily gated by the enzyme IDO. IDO expression is not static; it is highly inducible by pro-inflammatory cytokines—specifically Interferon-gamma, TNF-alpha, and bacterial lipopolysaccharides (LPS). Upon systemic immune activation, IDO exponentially accelerates the conversion of tryptophan to kynurenine. This directly results in a measurable spike in the Kynurenine-to-Tryptophan Ratio, making it an accurate clinical readout of inflammatory tone.
Neurotoxicity vs. Neuroprotection Trajectories Downstream metabolism of kynurenine diverges into two distinct branches: the neurotoxic branch (yielding quinolinic acid, an aggressive NMDA receptor agonist) and the neuroprotective branch (yielding kynurenic acid). Elevated systemic KTR typically indicates a shift toward a neurotoxic microenvironment. This persistent low-grade neurotoxicity correlates tightly with structural axonal injury, which is measured clinically via the efflux of Neurofilament Light Chain (NfL) into the plasma.
Tryptophan 2,3-dioxygenase (TDO) Pathway Operating in parallel to IDO, TDO is localized primarily in the hepatic system. Unlike IDO, TDO is regulated by circulating glucocorticoids and psychological/physiological stress rather than direct immune activation. Consequently, systemic KTR integrates dual biological burdens: hepatic stress responses and peripheral immune tone. Downregulating KTR requires resolving both inflammatory inputs and chronic glucocorticoid elevations.
Unlocking The Secrets Of Exceptional Longevity
I. Executive Summary
The core thesis of this discussion posits that exceptional longevity (living to 100+ years) is not merely a product of avoiding disease, but rather the result of a highly adapted, “dynamically resilient” immune and cellular system. Utilizing induced pluripotent stem cells (iPSCs) generated from centenarians, Dr. Murphy’s laboratory has identified a distinct biological signature of longevity. Rather than possessing a hyper-active or “supercharged” cellular baseline, centenarian cells operate in a state of remarkably low energy expenditure and transcriptomic noise during resting states. However, upon exposure to external stressors, these cells exhibit an explosive, highly efficient upregulation of quality-control mechanisms to neutralize the threat.
In the immune compartment, centenarians exhibit counterintuitive adaptations. Unlike normal aging—which is marked by a loss of immune diversity and a dangerous increase in clonal hematopoiesis (blood cells originating from very few stem cells)—centenarians actually exhibit higher clonality. However, rather than driving leukemogenesis or cardiovascular disease, these specific stem cell clones appear to be hyper-fit, producing an elite repertoire of B cells and cytotoxic T cells. These findings suggest centenarians possess a “house-rejuvenation” program stemming from highly functional, mutant hematopoietic stem cells (HSCs) that resist age-related exhaustion.
Functionally, centenarian-derived neurons demonstrate advanced neurogenesis signatures and resistance to Alzheimer’s-like stressors. Mitochondrially, their resting membrane potential is paradoxically low—a trait shared with other exceptionally long-lived mammals like naked mole rats—which likely minimizes baseline oxidative stress. The ultimate goal of this research is a “clinical trial in a tube,” testing gero-protectors (like GLP-1s or Rapamycin) on patient-specific stem cells to map personalized interventions without risking in-vivo toxicity.
II. Insight Bullets
- Dynamic Resilience vs. Disease Avoidance: Centenarians do not strictly avoid diseases (many survived the 1918 flu and COVID-19 multiple times); instead, they possess a cellular architecture designed to bounce back rapidly from acute systemic insults.
- The “Quiet” Cellular Baseline: In a naive (unstressed) state, centenarian neurons and immune cells exhibit low transcriptional noise, low energy expenditure, and a highly ordered resting state compared to younger control groups.
- Mitochondrial Paradox: Centenarian mitochondria have a remarkably lower resting membrane potential than average controls. This matches the metabolic phenotype of long-lived species (e.g., naked mole rats) and drastically limits baseline oxidative stress [Source unverified in live search].
- Stress-Induced Hyper-Response: When exposed to endoplasmic reticulum (ER) stress, centenarian cells aggressively upregulate protein-processing and quality-control genes much faster and more efficiently than non-centenarian cells.
- Neurogenesis Signature: Post-stress, iPSC-derived neurons from centenarians display robust transcriptomic signatures for the birth of new neurons (neurogenesis), countering the dogma that the elderly brain completely loses regenerative capacity.
- Immune Proportionality Shift: Centenarians maintain a unique immune profile characterized by a higher proportion of protective B cells and highly active cytotoxic T cells, alongside fewer regulatory T-helper cells, primed for rapid pathogen response.
- The Clonality Anomaly: While clonal hematopoiesis (CHIP) usually drives blood cancers and heart disease in normal aging, centenarians exhibit massive clonality driven by non-pathogenic, “elite” stem cells that pump out highly functional immune progeny.
- In Vitro Parabiosis: Bathing normal, young neuronal organoids in blood serum derived from centenarians rapidly induced positive epigenetic and functional changes, proving that circulating factors directly govern cellular age.
- Mosaic Loss of Y Chromosome (mLOY): Approximately 30% of male centenarians exhibit mLOY. While typically associated with cancer risk in the general population, it may paradoxically offer protective resilience in extreme age [Source unverified in live search].
- Tryptophan / NAD+ Axis: Elevated Kynurenine-to-Tryptophan ratios (KTR) strongly correlate with neurodegeneration. Inhibiting IDO1 (the enzyme driving this breakdown) quenches inflammatory cascades in lab-derived human neurons.
- Clinical Trial in a Tube: Using patient-derived iPSCs allows researchers to test the toxicity and efficacy of geroprotectors (like Rapamycin or GLP-1s) on a personalized basis before the patient ingests the compound.
- Epigenetic Rejuvenation Validation: Reprogramming adult cells into iPSCs successfully resets their epigenetic clock to “zero,” proving that cellular aging is highly plastic and bi-directionally malleable.
IV. Actionable Protocol (Prioritized)
High Confidence Tier (Level A/B Evidence)
- Optimize Vitamin D Status: Dr. Murphy notes ubiquitous clinical tracking of Vitamin D. Keep 25(OH)D levels optimized to support innate immune function and hematopoietic regulation.
- Monitor Baseline Inflammation: Track High-Sensitivity C-Reactive Protein (hs-CRP). The centenarian phenotype relies on near-zero “inflammaging” at rest. Elevated baseline inflammation exhausts immune reserves over time.
Experimental Tier (Level C/D Evidence)
- NAD+ Precursor Supplementation: Utilize NR (Nicotinamide Riboside) or NMN (Nicotinamide Mononucleotide) to combat the age-related decline in the Tryptophan-NAD+ salvage pathway, a cascade directly associated with maintaining cellular energy and mitigating neuroinflammation.
- Targeting the IDO1/Tryptophan Pathway: For high inflammatory states, dietary or supplement-based modulation of tryptophan metabolism (minimizing IDO1 hyper-activation) may reduce systemic kynurenine toxicity.
- Alpha-Ketoglutarate (AKG): Used experimentally by researchers (including Dr. Murphy) to support mitochondrial bioenergetics and epigenetic maintenance, despite mixed/negative results in the most recent ITP (Interventions Testing Program) mouse trials.
Red Flag Zone (Safety Data & Gaps)
- Blindly Forcing Mitochondrial Output: Do not blindly attempt to “supercharge” mitochondrial membrane potential. The centenarian data explicitly warns that over-activating resting mitochondria creates excessive oxidative damage; the goal is metabolic flexibility, not chronic overdrive.
- Misinterpreting “Clonality” Labs: Commercial biological age tests cannot yet distinguish between “good” centenarian-style clonal hematopoiesis and “bad” leukemogenic CHIP. Do not panic over clonality markers without targeted hematological sequencing.
- Over-the-Counter “Geroprotectors”: Assuming compounds like Astaxanthin or GLP-1 agonists will act as universal longevity enhancers is flawed. Efficacy is highly individualized based on genetics; what extends life in one phenotype may be toxic in another.
Another new video by Mike: 70% Lifespan Extension: Immune-Derived "Telomere Rivers"—A Transferable Youth Signal? - #13 by RapAdmin
The Immune System Impacts Longevity: What To Measure (Natalia Mitin)
Related reading:
I. Executive Summary
Dr. Natalia Mitin, molecular biologist and founder of SapphireX, provides a clinical assessment of adaptive immunosenescence and cellular senescence, arguing that chronological age and standard complete blood counts (CBC) are inadequate metrics for measuring true biological immune resilience. The core thesis establishes that total white blood cell counts mask critical subpopulation shifts during aging—specifically, the functional decline of naive T-cells and the simultaneous rise of neutrophils and monocytes. Standard clinical assays fail to capture the functional degradation of the immune network until late-stage frailty and overt disease manifest.
A critical revelation from ongoing clinical data is that systemic immunosenescence—the global deregulation of the adaptive immune system—almost universally precedes the widespread accumulation of cellular senescence. Consequently, the popular biohacking strategy of indiscriminately deploying senolytic therapies (e.g., dasatinib, fisetin) without molecular testing is deeply flawed. Clinical profiles indicate that only 10% of individuals have high cellular senescence as an isolated biological defect.
Applying aggressive senolytic protocols to the remaining 90% risks severe physiological destabilization by targeting the wrong biological pathway.
Furthermore, recent literature challenges the absolute toxicity of the senescence biomarker p16. While chronic p16 elevation in T-cells strongly correlates with accelerated aging and adverse clinical outcomes (such as severe peripheral neuropathy following chemotherapy), acute, transient p16 expression in macrophages acts as an essential tissue-protective mechanism during active infections and vaccine responses.
The adaptive immune system operates as an intricate, balanced network consisting of functional domains: T-cell exhaustion, proliferation (stemness), differentiation, and senescence. Rather than forcing single biological levers through extreme caloric restriction, excessive endurance exercise, or polypharmacy supplement “stacking,” clinicians must focus on mapping personal immunological trajectories. Over-activation of any single pathway frequently forces the immune system into autoimmune reactivity or severe cellular exhaustion. The overriding protocol objective for functional longevity is not aggressive immunological stimulation or cellular purging, but rather identifying specific molecular insults, gently removing them, and allowing the biological system to endogenously rebalance its homeostatic baseline.
II. Insight Bullets
- Deceptive Clinical Panels: Total white blood cell counts mask immune aging. During normal aging, neutrophils and monocytes increase while functional lymphocytes decrease, rendering total WBC counts clinically useless for longevity screening.
- Immunosenescence Precedes Cellular Senescence: The functional degradation of the adaptive immune system (loss of naive T-cells and increased T-cell exhaustion) occurs long before the massive accumulation of senescent cells in most patients.
- Acute vs. Chronic Senescence: Acute cellular senescence (transient p16 expression in macrophages) protects tissues from inflammatory damage during infections. Conversely, chronic senescence (persistent p16 in T-cells) drives systemic inflammaging.
- T-Cell Exhaustion (Defense Domain): Chronic physiological stress and latent viral infections (e.g., CMV, EBV) force T-cells into a state of exhaustion, drastically reducing their capacity to clear pathogens and senescent cells.
- LAG3 as a Superior Exhaustion Marker: Multi-omics modeling identifies the LAG3 gene as a highly accurate biomarker for T-cell exhaustion. LAG3 acts as an inhibitory brake, preventing catastrophic autoimmune over-proliferation.
- Naive T-Cell “Stemness”: Stemness measures the proliferative capacity of naive T-cells, which heavily relies on mitochondrial function and is essential for mounting defenses against novel antigens.
- The Senolytics Fallacy: Only ~10% of clinical longevity patients present with cellular senescence as their primary defect. Indiscriminate use of senolytic drugs is clinically unjustified for the vast majority of individuals.
- CD4/CD8 Ratio is a Lagging Indicator: An inverted CD4/CD8 ratio is an established marker of severe frailty, but it only presents during late-stage immune collapse. Gene expression profiling detects vulnerabilities years earlier.
- Chemotherapy and Accelerated Aging: In oncology, elevated baseline p16 expression in T-cells strongly predicts long-term, detrimental side effects, including severe peripheral neuropathy following chemotherapy.
- The Hazard of Supplement “Stacking”: Aggressively layering supplements and longevity drugs without targeted baseline testing frequently deregulates immune homeostasis and drives up T-cell exhaustion markers.
- Melatonin-Induced Cortisol Disruption: High-dose, untargeted melatonin supplementation can severely suppress physiological morning cortisol levels, disrupting the circadian rhythm and blunting immune recovery.
- Overtraining Syndrome: While moderate exercise is geroprotective, chronic over-exercising is a massive driver of elevated cellular senescence and T-cell exhaustion.
- Gut Permeability as an Inflammatory Driver: With age, compromised intestinal integrity becomes a primary source of systemic inflammatory cytokines, perpetually hyper-activating the adaptive immune system.
- Low Cellular Senescence Danger: Dangerously low p16 levels can indicate an impaired tumor-suppressor mechanism, escalating the statistical risk for solid tumor malignancies.
- System Rebalancing Over Targeted Purging: The clinical goal of longevity medicine is not to aggressively purge cells or artificially spike immune activity, but to remove specific molecular stressors and allow the body’s immune network to endogenously repair.
IV. Actionable Protocol (Prioritized)
High Confidence Tier (Level A/B Evidence)
- Mitigation of Exhaustive Exercise: Restrict chronic, exhaustive endurance training. High-intensity exercise to fatigue significantly increases neutrophil-driven oxidative stress, impairs phagocytic function, and heavily amplifies the systemic inflammatory response, leading to post-exercise immunosuppression. Exercise workload: a key determinant of immune health, 2025
- Latent Viral Load Management: Monitor for Cytomegalovirus (CMV) and Epstein-Barr Virus (EBV) reactivation. Chronic CMV infection heavily skews the T-cell repertoire, drives the expansion of exhausted CD28- T-cells, and is a primary biological mechanism accelerating systemic immunosenescence. Immunosenescence and Cytomegalovirus: Exploring Their Connection, 2024
Experimental Tier (Level C/D Evidence with High Safety Margins)
- Molecular Immune Domain Tracking: Shift clinical tracking away from basic CBCs and inverted CD4/CD8 ratios toward gene expression profiling (e.g., measuring LAG3 for T-cell exhaustion) to identify specific adaptive immune vulnerabilities years before overt clinical frailty.
- Intermittent Caloric Restriction: Implementation of fasting mimicking diets or caloric restriction demonstrates preliminary efficacy in beneficially modulating T-cell stemness and attenuating senescence-associated secretory phenotype (SASP) markers, though precise metabolic endpoints and standardized human tracking remain ongoing. Intermittent fasting and immune aging, 2024
Red Flag Zone (Safety Data Absent or Elevated Risk)
- Indiscriminate Senolytic Protocols: The unguided administration of senolytics (e.g., Dasatinib, Quercetin, Fisetin) lacks proven long-term efficacy for healthy human life extension and poses severe risks. Recent longitudinal trials show that Dasatinib and Quercetin can actually increase epigenetic age acceleration and dramatically decrease telomere length over a 6-month period. Exploring the effects of Dasatinib, Quercetin, and Fisetin on DNA methylation clocks, 2024
- Unmonitored Polypharmacy (“Stacking”): Combining multiple anti-aging therapeutics (e.g., NAD+ precursors, high-dose melatonin, rapamycin, and senolytics) without molecular baseline testing frequently suppresses physiological cortisol response, induces T-cell exhaustion, and deregulates the finely balanced adaptive immune network.
Do Centenarians Have A Unique Immune System?
I. Executive Summary
The provided transcript critically evaluates the unique immunological architecture of centenarians, establishing that standard complete blood counts (CBC) entirely fail to capture the functional cellular shifts dictating extreme human longevity. During normal biological aging, myeloid lineages (neutrophils, monocytes) expand while functional lymphoid lineages (T-cells, B-cells) deplete. Centenarians, however, diverge from this trajectory, presenting a highly specialized and adaptive immune phenotype characterized by diminished basal inflammation and targeted pathogen reactivity.
The central biological paradox discussed revolves around Clonal Hematopoiesis of Indeterminate Potential (CHIP). In the general aging population, the attrition of diverse hematopoietic stem cells (HSCs) forces the entire blood system to be repopulated by a shrinking pool of stem cells. This clonality typically accumulates pathogenic mutations, driving leukemogenesis and cardiovascular mortality. Conversely, centenarians exhibit extreme HSC clonality—frequently repopulating their entire immune system from just one or two HSC lines—yet completely lack the oncogenic driver mutations that precipitate disease. These surviving “elite” HSC lineages appear functionally superior, generating rejuvenated immune progeny capable of robust responses to novel antigens.
Furthermore, immunophenotyping reveals that centenarians harbor an anomalous expansion of CD4+ cytotoxic T-cells. This highly specialized subset is virtually absent in younger cohorts (ranging from 0.3% to 2.6%) but surges to over 7.5% in centenarians. Rather than succumbing to non-productive, smoldering inflammaging, this immune architecture remains clinically quiet at baseline while possessing a formidable capacity to neutralize acute insults. Current translational efforts are leveraging non-invasive single-cell transcriptomics and induced pluripotent stem cells (iPSCs) to reverse-engineer centenarian hematopoiesis ex vivo. The overarching clinical objective is to identify targetable stemness factors that could theoretically reconstitute and rejuvenate the failing HSC reservoirs in standard aging populations.
II. Insight Bullets
- Clinical Inadequacy of Total WBC Counts: Relying on total white blood cell numbers masks critical age-related immunological deterioration, specifically the reciprocal decline of functional lymphocytes and the expansion of neutrophils and monocytes.
- CD4+ T-Cell Rejuvenation Efficacy: Emerging pre-clinical models suggest that rejuvenating CD4+ T-cells (e.g., via telomere transfer mechanisms) may extend mammalian lifespan to a magnitude exceeding traditional interventions like caloric restriction or mTOR inhibition.
- The Centenarian CD4+ Cytotoxic Expansion: Centenarians present a unique immune signature featuring a massive expansion of CD4+ cytotoxic T-cells, a subset that is nearly undetectable in young, healthy populations.
- Paradoxical B-Cell/T-Helper Ratios: Transcriptomic profiling indicates that centenarians possess a specialized adaptive shift, retaining highly functional B-cell populations while operating with fewer total T-helper cells than standard older adults.
- Mechanics of Clonal Hematopoiesis (CHIP): Biological aging drastically reduces HSC diversity. Blood production becomes monopolized by a few dominant HSC clones, a dynamic heavily correlated with cardiovascular disease and blood cancers in the general population.
- The Centenarian CHIP Anomaly: Extreme longevity is characterized by massive hematologic clonality without pathology. Centenarian HSCs harbor mutations that confer elite cellular fitness rather than oncogenic disease drivers.
- HSC Quantity vs. Functional Quality: Senescent biological models (e.g., aged mice) possess mathematically higher numbers of HSCs by surface marker definitions, but these cells exhibit profound functional failure in bone marrow repopulation assays.
- Suppression of Basal Inflammaging: The centenarian immune network exerts tight control over “smoldering” inflammation, maintaining a quiet basal state that resists the misprimed autoimmune reactivity common in standard aging.
- Elite Adaptation to Lifetime Pathogens: Extreme longevity reflects an immune system trained by sequential global pathogens (e.g., Spanish Flu, SARS-CoV-2) into a highly efficient, rapidly responding defense network.
- NK Cell and Monocyte Elevations: Alongside unique T-cell adaptations, centenarians exhibit dramatic increases in CD14+ monocytes and Natural Killer (NK) cells to manage localized microbial burdens.
- Non-Invasive iPSC Lineage Tracing: Modern hematology circumvents the need for dangerous bone marrow biopsies in centenarians by converting peripheral blood cells into iPSCs to bioinformatically trace the clonality of elite HSCs.
- Absence of a Singular Longevity Gene: Phenotypic data proves there is no single “magic bullet” genetic mutation guaranteeing extreme longevity; it is a heterogeneous, multi-pathway adaptation of stem cell fitness.
IV. Actionable Protocol (Prioritized)
High Confidence Tier (Level A/B Evidence)
- Advanced Immunophenotyping Over Standard CBC: Standard CBC panels are insufficient for longevity profiling. Clinical assessments must include flow cytometry or targeted panels to quantify subpopulation ratios (CD4/CD8, specific monocyte subtypes) to accurately track immune senescence. Immune system aging and the potential for interventions, 2024
- Inflammaging Suppression Protocols: The absence of basal inflammation is a hallmark of extreme longevity. Implement clinically verified protocols (e.g., rigorous metabolic management, targeted dietary restriction, continuous glucose monitoring) to suppress chronic systemic inflammatory cytokines. Inflammaging: a highly targetable driver of clinical decline, 2023
Experimental Tier (Level C/D Evidence with High Safety Margins)
- Monitoring for Clonal Hematopoiesis (CHIP): Early clinical screening via targeted DNA sequencing of peripheral blood can detect CHIP variants. While interventions remain experimental, identifying pathogenic clones allows for aggressive preventative cardiovascular risk management. Clonal hematopoiesis of indeterminate potential and its impact on patient trajectories, 2022
- Ex Vivo Immune Reconstitution: Current academic focus is on modeling centenarian HSC stemness factors (e.g., TCF7, RUNX1 modulation) via iPSC generation. While purely experimental, these pathways hold the potential for future autologous HSC rejuvenation therapies.
Red Flag Zone (Safety Data Absent or Elevated Risk)
- Misinterpreting Elevated Immune Subsets as Uniform Pathology: The expansion of cytotoxic T-cells or elevated NK cells in advanced age should not automatically be suppressed with broad-spectrum immunosuppressants unless overt autoimmunity is present; these expansions may represent vital, compensatory longevity adaptations.
- Direct CD4 Rejuvenation Therapies in Humans: Extrapolating emerging mouse data (e.g., intercellular telomere transfer to CD4 cells) directly to human biological interventions currently lacks human safety data and carries theoretical risks of inducing unregulated lymphoproliferative disorders.
Tracking A Biomarker Of Neurodegeneration (22-Test Analysis)
I. Executive Summary
If people live long enough, nerve degeneration becomes a nearly universal challenge. Biomarkers of this decline increase continuously as we age. One of the most important markers is neurofilament light chain (NFL). NFL is a structural protein located inside nerve cells. When the long cables of nerve cells, known as axons, are injured or begin to die, NFL leaks into the bloodstream. Measuring this protein provides a direct view of active brain damage. Recent large-scale data confirms that out of all major organ systems, the biological age of the brain and the immune system are the strongest predictors of a long, healthy life. In these models, having elevated levels of NFL in the blood is the top indicator of an older brain age and an increased risk of early death.
Despite its clinical importance, direct NFL testing is currently very expensive, making routine monitoring difficult for the general public. However, an alternative biomarker can be used as a proxy: the kynurenine to tryptophan ratio. Tryptophan is an essential amino acid acquired from food. Under states of physical stress, immune activation, or inflammation, the liver and immune cells break down tryptophan into a compound called kynurenine. A high ratio of kynurenine to tryptophan strongly correlates with high NFL levels and active nerve damage. This ratio acts as an integrated dashboard of the body’s current inflammation levels, antioxidant defenses, and potential brain decline.
Because measuring this ratio through standard metabolic blood panels is more affordable, it allows for frequent tracking. Personal health data analysis reveals that diet plays a major role in influencing this ratio. Specifically, tracking daily food intake uncovered a strong inverse relationship between consuming monounsaturated fats and the kynurenine to tryptophan ratio. Consuming higher amounts of monounsaturated fats was linked to a lower, healthier ratio. By making targeted dietary adjustments and running frequent blood tests, individuals can experiment with their own data to minimize inflammation. This strict self-tracking method provides a practical roadmap for identifying which specific lifestyle interventions actually improve biological aging, offering a proactive defense against age-related brain decline.
II. Insight Bullets
- Nerve Degeneration is Universal: Without active intervention, markers of nerve damage naturally rise in everyone as they get older.
- What is NFL: Neurofilament light chain (NFL) is a structural protein released into the blood when the long branches of nerve cells are injured.
- Top Predictor of Brain Age: In massive population studies, NFL is the most powerful blood protein for predicting an older biological brain age.
- Organ Clocks and Lifespan: Out of all the body’s systems, the biological age of the brain and the immune system are the strongest predictors of how long someone will live.
- The Cost Barrier: Direct blood tests for NFL are currently too expensive for the average person to use for frequent, routine tracking.
- A Cheaper Alternative: The ratio of kynurenine to tryptophan in the blood is a measurable substitute that strongly matches NFL levels.
- The Tryptophan Breakdown: Tryptophan is an amino acid from food. During inflammation, the body breaks it down into kynurenine.
- High Ratio equals High Risk: A high kynurenine to tryptophan ratio means high inflammation and correlates directly with high NFL and nerve damage.
- Inflammatory Triggers: The breakdown of tryptophan into kynurenine is heavily accelerated by bacterial toxins and pro-inflammatory immune signals.
- Protective Factors: Anti-inflammatory signals and antioxidant enzymes (like superoxide dismutase) slow down this breakdown, keeping the ratio at healthier, lower levels.
- Metabolic Testing: At-home metabolic kits can measure hundreds of blood compounds at once, making it easier to calculate this ratio affordably.
- The Power of Self-Tracking: By taking multiple tests over years, individuals can find their personal baseline and see if their lifestyle changes are actually working.
- Finding Optimal Levels: The lowest possible levels of the kynurenine to tryptophan ratio are associated with the lowest levels of nerve damage.
- Dietary Correlations: Analyzing personal blood test data alongside strict diet tracking can reveal exactly which foods improve health markers.
- Monounsaturated Fats: In detailed data tracking, a higher daily intake of monounsaturated fats strongly correlated with a lower, healthier kynurenine to tryptophan ratio.
- Continuous Experimentation: If a specific dietary change does not immediately improve the blood markers on the next test, the plan must be objectively adjusted and tested again.
IV. Actionable Protocol (Prioritized)
High Confidence Tier
- Reduce Systemic Inflammation: General inflammation directly drives the harmful conversion of tryptophan to kynurenine, which is linked to nerve damage. Address chronic inflammation through established clinical methods such as treating underlying infections, maintaining optimal metabolic health, and reducing visceral fat to protect the brain.
- Consume Adequate Tryptophan: Ensure sufficient dietary intake of essential amino acids through diet. Tryptophan depletion negatively impacts the central nervous system, and severe depletion is a known driver of mood and cognitive disorders.
Experimental Tier
- Increase Monounsaturated Fats (MUFAs): Clinical data and strict personal tracking suggest that diets higher in monounsaturated fats (found in olive oil, avocados, and specific nuts) may help lower the kynurenine to tryptophan ratio. This is a low-risk dietary shift that supports general cardiovascular and neuro-protective health.
- Track the Kynurenine to Tryptophan Ratio: Use routine metabolic blood testing to track this specific ratio over time. Rather than relying solely on expensive direct NFL tests, utilize this ratio as an early-warning surrogate marker for brain health and hidden inflammation.
- Conduct Dietary Data Trials: Accurately weigh and log daily food intake alongside quarterly blood testing. This allows for an objective assessment of how specific macronutrients influence personal inflammatory markers over time, removing the guesswork from dietary planning.
Red Flag Zone
- Relying Only on Basic Blood Panels: Standard complete blood count (CBC) panels do not capture the specific protein changes, like NFL or the kynurenine ratio, that indicate early nerve damage.
- Blind Supplementation: Changing diets or taking supplements based on general advice without testing specific blood markers is ineffective. Without a hard baseline and rigorous follow-up data, it is impossible to know if an intervention is working or causing silent, low-grade inflammation.
Consistently Higher HRV, Lower RHR Since 2018
I. Executive Summary
The autonomic nervous system dictates physiological longevity via the delicate balance of sympathetic (fight-or-flight) and parasympathetic (rest-and-digest) activation. Resting Heart Rate (RHR) and Heart Rate Variability (HRV) are direct, quantifiable readouts of this equilibrium, reflecting not just cardiac health, but the regulatory tone of the vagus nerve and adrenal gland. A fundamental principle in human chronobiology is that systemic aging inherently degrades the parasympathetic nervous system, leading to a predictable decrease in HRV and an increase in baseline RHR until roughly the fifth decade of life (whereafter RHR declines pathologically due to sinus node decay).
The transcript documents a rigorous, longitudinal N=1 study comprising nearly 2,800 days of continuous biometric tracking (2018–2026), demonstrating that this “inevitable” autonomic decline can be successfully resisted and reversed. Over an eight-year period, systematic lifestyle interventions forced an inversion of the standard aging trajectory: average RHR dropped from 51 bpm to 41.4 bpm, while average HRV progressively surged from 47 milliseconds to 73 milliseconds.
This specific physiological phenotype—a low RHR operating in tandem with a high HRV—is exclusively indicative of an elastic, youthful autonomic nervous system. Achieving this requires moving beyond isolated, general fitness advice to highly structured biological tracking. The primary drivers of this metric inversion included targeted body composition optimization (reducing BMI from ~25 to ~22) and the strict implementation of a titrated workload-to-recovery algorithm (one intensive 80–90 minute exertion event followed by two heavily regulated, low-intensity recovery days). A critical error in modern biohacking is interpreting RHR or HRV as independent metrics; an aging or overtrained physiological system can produce a low RHR alongside a suppressed HRV. Therefore, clinical longevity tracking dictates that these biomarkers must be analyzed as a coupled ratio to accurately verify neuro-cardiac resilience.
II. Insight Bullets
- Autonomic System Dashboard: RHR and HRV are not merely fitness markers; they act as an integrated measure of three distinct organ systems: the heart, the nervous system (vagal tone), and the adrenal gland (norepinephrine output).
- The Sympathetic Penalty: Chronic sympathetic nervous system activation floods the body with adrenal norepinephrine, mechanically forcing a higher resting heart rate while simultaneously collapsing heart rate variability.
- The Aging Heart Rate Curve: Epidemiological data demonstrates an inverse U-shape for RHR. It rises steadily from youth through the early 50s, after which it declines—not due to fitness, but due to age-related electrical degradation of the heart’s pacemaker cells.
- The HRV Aging Trajectory: Heart rate variability reliably, and almost linearly, declines with age, reflecting the progressive loss of parasympathetic elasticity and vagal tone.
- The Coupled Metric Imperative: Evaluating RHR or HRV in isolation is a clinical error. A low RHR only indicates a “youthful” biological state if it is mathematically paired with a high HRV. A low RHR with a low HRV indicates autonomic decay.
- Longitudinal Reversal: Continuous tracking data proves it is possible to reverse the autonomic aging curve. Over 8 years, systematic interventions raised average HRV from 47ms to 73ms while dropping RHR from 51 bpm to 41 bpm.
- BMI Optimization: Shedding excess weight, specifically moving from the high end of the “normal” BMI range (~25) to a leaner state (~22), triggered an immediate and sustained 15+ point elevation in HRV.
- The Overtraining Trap: Chronic, daily intensive exercise without structured recovery mimics systemic stress, driving up RHR and crushing HRV. Geroprotective exercise requires engineered rest.
- Titrated Recovery Algorithm: Autonomic optimization requires a strict ratio of exertion to recovery. A verified protocol utilizes one 80–90 minute intensive workout day, followed by two days of aggressively titrated, low-intensity active recovery based on daily heart rate averages.
- The Illusion of the LLM Reference Range: Standard laboratory reference ranges simply reflect the average state of a sick, aging population. True longevity targets must be derived from optimal all-cause mortality data, not standard distribution curves.
- Daily Biometric Iteration: Optimizing longevity biomarkers cannot be achieved with annual checkups. It requires daily data aggregation to actively titrate diet, sleep, and physical exertion loads in real-time.
- Wearable Validation: Commercial fitness wearables (Whoop, Oura, Garmin, Apple) possess sufficient clinical-grade accuracy to reliably quantify long-term HRV and RHR trends, providing a robust foundation for N=1 physiological experiments.
IV. Actionable Protocol (Prioritized)
High Confidence Tier (Level A/B Evidence)
- Targeted Weight Reduction for Autonomic Tone: If operating at the higher end of the standard BMI range, implement caloric restriction to optimize body composition. Reductions in adipose tissue directly decrease systemic sympathetic drive, definitively lowering RHR and increasing HRV. Weight loss improves heart rate recovery in overweight and obese men, 2013
- Structured Periodization: Abandon unstructured, chronic daily high-intensity training. Implement a strict exertion-to-recovery ratio (e.g., 1 day of intense exertion followed by 2 days of active, low-heart-rate recovery) to prevent autonomic exhaustion and maximize parasympathetic rebound. Heart rate variability in elite endurance athletes: longitudinal changes and associations with training, 2024
Experimental Tier (Level C/D Evidence with High Safety Margins)
- Coupled Biometric Tracking: Utilize continuous biometric wearables to track the ratio of HRV to RHR over a rolling 7-day average. Do not assess one metric independently. Intervene with immediate rest or dietary modification if HRV drops concurrently with a rising RHR.
- Daily Activity Titration: Use Average Daily Heart Rate on non-training days as a hard ceiling for physical activity. Actively restrict movement and stress exposure on recovery days to ensure complete parasympathetic reset before the next intense training block.
Red Flag Zone (Safety Data Absent or Elevated Risk)
- Interpreting a Dropping RHR in Advanced Age as “Fitness”: A declining resting heart rate in individuals over 50—if unaccompanied by high-intensity exercise and a concurrently high HRV—is a severe red flag indicating sinus node dysfunction or progressive cardiac electrical block, not cardiovascular health.
- Chasing Standard LLM/Lab Reference Ranges: Conforming to standard “normal” medical reference ranges for RHR (e.g., 60-100 bpm) guarantees average aging. Optimal survival metrics require pushing parameters safely into the elite, lower ranges (e.g., 40-50 bpm) paired with high HRV.
Quantifying Biological Age: Test #1 In 2026
I. Executive Summary
This transcript details the 65th blood test results of a 53-year-old male longevity practitioner, focusing on the PhenoAge biological age clock developed by Dr. Morgan Levine. The subject reported a biological age of 35.8 years, representing a 17.2-year reduction relative to his chronological age. Despite this absolute reduction, a critical longitudinal analysis reveals a three-year upward trend in biological age (from a 2021 low of 32.1 years). This underscores the necessity of high-frequency testing (8 times per year) to distinguish between transient fluctuations and genuine “rates of aging.”
The primary technical focus is the optimization of Mean Corpuscular Volume (MCV), a measure of red blood cell size that typically increases with age and is associated with heightened mortality risk. The subject’s MCV has risen to 92.5 fL, exceeding his self-derived optimal target of 90 fL. Through rigorous N=1 tracking—involving daily weighing of food and correlation analysis of over 30 biomarkers—the subject identified a strong positive correlation (r=0.73,p<0.05) between fresh ginger intake and MCV.
Standard causes for macrocytosis (elevated MCV), such as Vitamin B12/folate deficiencies, alcohol consumption, and hypothyroidism, were methodically ruled out through high-dose supplementation (B12 at 400x RDA) and clinical history. The subject’s current intervention involves a phased reduction of fresh ginger from 7g/day to 1.5g/day to test the hypothesis that ginger may be a causative driver of his specific MCV elevation. This approach highlights a transition from generalized longevity protocols to highly individualized, data-driven bio-optimization. However, the evidence for ginger’s impact on red blood cell volume remains strictly correlative and specific to this individual’s biology, lacking broader clinical validation in healthy cohorts.
II. Insight Bullets
- PhenoAge Accuracy: The PhenoAge clock utilizes nine clinical biomarkers to estimate mortality risk; however, it has a “floor” effect where the maximum measurable reduction is approximately 20 years.
- Testing Frequency: Single “snapshots” of biological age are insufficient. Establishing a “true rate of aging” requires longitudinal, year-to-year averages to filter out biological noise.
- Creatine Interference: Supplementation with creatine increases serum creatinine levels, which can artificially inflate biological age scores on calculators like PhenoAge without necessarily indicating renal decline.
- MCV as Aging Proxy: MCV is a validated marker of biological aging; larger red blood cells are often linked to cellular senescence and increased all-cause mortality risk (Levine et al., 2018).
- Optimal vs. Reference Ranges: Standard clinical reference ranges (80–100 fL for MCV) are designed to catch pathology, not optimize longevity. The subject argues for “optimal” targets derived from youth-associated data.
- B12/Folate Saturability: Intake of B12 at 400x the RDA and folate at 4x the RDA eliminates nutrient deficiency as a cause for elevated MCV in this subject.
- Thyroid-MCV Link: While hypothyroidism is a known cause of macrocytosis, correcting T3 levels through medication (Cytomel) did not lower the subject’s MCV, suggesting independent drivers.
- Precision Tracking: Achieving meaningful correlations in N=1 data requires weighing 99% of food intake to minimize variables in the spreadsheet analysis.
- Ginger Correlation: A strong positive correlation (r=0.73) suggests that higher intakes of fresh ginger (7g/day) may be linked to increased MCV in this specific subject.
- Statistical Significance: P-values below 0.05 in N=1 data help separate “signal” from “noise” over a decade of testing, though they do not prove causation.
- Biological Age Trajectory: Even with static biomarkers, the PhenoAge algorithm adds approximately 0.9 years to biological age for every chronological year, necessitating active intervention to remain “flat.”
- Creatine Subjectivity: The subject discontinued creatine because it yielded no measurable improvements in biomarkers or subjective well-being (sleep/recovery), highlighting the importance of “cutting the fluff” in protocols.
IV. Actionable Protocol (Prioritized)
High Confidence Tier
- PhenoAge Monitoring: Utilize the Morgan Levine PhenoAge algorithm (Albumin, Creatinine, Glucose, CRP, Lymphocyte %, Mean Cell Volume, RBC Distribution Width, Alkaline Phosphatase, White Blood Cell Count) to track multisystem aging.
- Creatine Awareness: If supplementing with creatine, expect elevated serum creatinine. To get an accurate biological age reading, discontinue creatine 2–4 weeks prior to testing or utilize Cystatin C as an alternative marker for kidney function (Enko et al., 2023).
- Nutrient Baseline: Ensure Vitamin B12 and Folate levels are optimal to rule out megaloblastic anemia as a cause for high MCV.
Experimental Tier
- MCV Optimization: Aim for an MCV target of ~90 fL, which is more closely associated with youthful physiology than the upper limit of clinical ranges (100 fL).
- Ginger Modulation: For individuals with high MCV and high fresh ginger intake (5g+), consider a reduction to 1–2g/day while monitoring blood counts to observe potential causal effects.
- High-Frequency Testing: Test biomarkers 4–8 times annually to establish a personalized “standard deviation” for your data.
Red Flag Zone
- N=1 Generalization: Do not assume a “ginger-MCV link” applies to the general population. Ginger has broad anti-inflammatory benefits in meta-analyses that may outweigh MCV concerns for most people (Zhu et al., 2022).
- Creatinine Misinterpretation: High creatinine alone does not confirm kidney disease; always correlate with GFR and Cystatin C.
- Source Unverified in Live Search: No Level A meta-analysis currently links moderate ginger consumption to macrocytosis in healthy humans.
Collaborative Truth-Seeking Note: The correlation between ginger and MCV is highly speculative. While the subject’s r=0.73 is statistically significant for his dataset, the biological mechanism is unidentified. Further data is needed to determine if ginger affects erythropoiesis or red cell membrane stability.
Predicting Heart Disease Risk With ApoB, LP(a), and VLDL
I. Executive Summary
This transcript features Dr. Elias Björnson (University of Gothenburg) discussing the development and validation of Risk-Weighted ApoB (rwApoB), a novel clinical metric designed to improve cardiovascular disease (CVD) risk prediction. The central thesis is that while total Apolipoprotein B (ApoB) counts the number of atherogenic particles, it fails to account for the varying “potency” of different particle types. Based on Mendelian Randomization (MR) data, Björnson’s group identified that Triglyceride-Rich Lipoproteins (TRLs) are 4–5 times more atherogenic than LDL per particle, while Lipoprotein(a) [Lp(a)] is 6–7 times more atherogenic.
The rwApoB metric synthesizes these weights into a single value, utilizing standard plasma measurements: Triglycerides, Lp(a), and total ApoB. Validation using the UK Biobank and MESA cohorts demonstrates that rwApoB significantly outperforms LDL-C, non-HDL-C, and even standard ApoB in predicting coronary heart disease (CHD). Crucially, the model identifies “discordant” individuals—approximately one-third of the population whose standard ApoB suggests moderate risk but whose rwApoB reveals high risk due to elevated Lp(a) or TGs.
The discussion emphasizes the “Necessary and Sufficient” substrate theory: ApoB-containing lipoproteins are the fundamental requirement for plaque formation; without them, atherosclerosis cannot initiate regardless of other risk factors like inflammation or blood pressure. For individuals seeking “primordial prevention” or maximum longevity, Björnson suggests an optimal rwApoB target of 40–50 mg/dL. While dietary interventions (high fiber, low saturated fat) can lower these metrics, the presentation highlights that pharmacological assistance (statins, PCSK9 inhibitors) is often required to reach these elite levels, particularly when dealing with genetically driven Lp(a) or high baseline LDL.
II. Insight Bullets
- Differential Atherogenicity: Not all ApoB particles are equal. TRLs (VLDL/Remnants) and Lp(a) have significantly higher “per-particle” risk than LDL (Björnson et al., 2024).
- The rwApoB Equation: A superior summary metric that weights particles by risk: rwApoB captures the “full spectrum” of atherogenic risk better than independent markers.
- Discordance Identification: Standard ApoB misses risk in ~33% of patients. High rwApoB identifies those with “residual lipid risk” who would otherwise be considered “controlled” on statins.
- Particle Abundance vs. Potency: LDL remains the primary driver of disease for most due to its sheer quantity, despite being less potent per particle than Lp(a) or TRLs.
- Substrate Necessity: ApoB particles are the sine qua non of atherosclerosis. High blood pressure and inflammation “enhance” plaque, but cannot create it in the absence of ApoB particles.
- Cumulative Exposure: CVD risk is a function of “area under the curve” (ApoB levels x years of exposure). Primordial prevention (starting in the 20s/30s) is exponentially more effective than late-life intervention.
- Lp(a) Stability Myth: While Lp(a) is 80–90% genetically determined, it may show minor biological and technical variability; however, it remains largely resistant to traditional diet and lifestyle changes.
- TRL as Underappreciated Risk: Plasma triglycerides serve as a proxy for TRLs. Levels above 90 mg/dL (1.0 mmol/L) represent an “underappreciated” source of remnant-driven risk.
- Metabolic Health Red Herring: “Metabolically healthy” individuals (low CRP, normal glucose) with high ApoB (e.g., Familial Hypercholesterolemia or Lean Mass Hyper-Responders) still develop rapid atherosclerosis.
- HDL as Proxy, Not Cause: HDL-C is a marker of TG metabolism/clearance but is not a causal factor in atherosclerosis, leading to its exclusion from the rwApoB model.
- Evolutionary Baseline: Hunter-gatherer populations (Hadza/Tsimane) maintain ApoB levels roughly 50% lower than Western averages (~40–60 mg/dL) throughout their lives with zero age-related climb.
- Statin Residual Risk: Statins primarily clear LDL. Residual risk in treated patients is often driven by untreated TRLs and Lp(a), which rwApoB accurately quantifies.
IV. Actionable Protocol (Prioritized)
High Confidence Tier (Level A/B Evidence)
- Comprehensive Testing: Move beyond LDL-C. Measure ApoB, Lp(a), and Triglycerides at least once to establish a baseline.
- rwApoB Calculation: Use the riskapp.com calculator to determine your weighted risk.
- Target Levels: Aim for ApoB < 60 mg/dL (Standard) or rwApoB < 50 mg/dL (Optimal Longevity) to halt plaque progression (Ference et al., 2017).
- Standard Pharmacotherapy: If rwApoB is high (>80-100 mg/dL), consider low-dose statins (e.g., Rosuvastatin 5mg) or Ezetimibe, which effectively lower the “LDL portion” of the risk weighted score.
Experimental Tier (Level C/D Evidence)
- High-Fiber “Portfolio” Diet: Target >50g fiber/day (the user reports 85g) and low saturated fat to lower ApoB by 20-30% (Jenkins et al., 2003).
- Mushroom Consumption: The user suggests 300g–700g of mushrooms (e.g., white button/oyster) for naturally occurring lovastatin (10mg). Note: Clinical trials for whole-food mushrooms as a statin replacement are lacking.
- Lp(a) Lowering (Emerging): High-dose Niacin may lower Lp(a) but is associated with significant side effects (liver enzyme elevation). Await clinical results for antisense oligonucleotides (Pelacarsen).
Red Flag Zone (Safety Data Absent/Debunked)
- The “Keto Lean Mass Hyper-Responder” (LMHR) Strategy: Maintaining LDL > 200 mg/dL or ApoB > 150 mg/dL while on a ketogenic diet, even with low CRP, is viewed by clinical experts as high risk for rapid plaque progression.
- Measurement Infrequency: Measuring lipids once every few years is insufficient to track the “area under the curve.”
- Source Unverified in Live Search: No Level A meta-analysis supports whole-food mushrooms as a primary treatment for hyperlipidemia compared to pharmaceutical statins.
Technical Accuracy Note: The rwApoB equation specifically uses weights of 1.0 for LDL, ~4.5 for TRL, and ~6.5 for Lp(a). These are derived from large-scale Mendelian Randomization studies where genetic variants influencing specific particles were compared against CHD outcomes.
Aging Mechanisms, And How To Fix It (Featuring Michael Levin, PhD)
I. Executive Summary
This transcript outlines a radical paradigm shift in geroscience, moving from the “stochastic damage” model of aging to a cognitive-morphogenetic framework. The core thesis, proposed by Michael Levin and colleagues, identifies aging as a failure of cellular collective intelligence. Cells are not merely building blocks but agents within a multi-scale competency architecture. During development and maturation, cells coordinate via bioelectrical and chemical signaling to achieve specific anatomical goal-states. Aging begins when this collective intelligence “disbands” after meeting its primary evolutionary objectives.
The mechanism of this decline is characterized as the “blurring” of bioelectrical memories—the voltage patterns across cellular networks that maintain species-specific morphology. As these pattern-memories degrade, cells lose their alignment toward the global anatomical goal and revert to individual “agendas.” This is evidenced by transcriptional drift, where aging cells express phylogenetic signals that regress toward more primitive, unicellular evolutionary states. This loss of coordination creates a positive feedback loop of systemic disarray, leading to the hallmark phenotypes of degenerative disease.
Looking forward, the transcript argues that longevity research should move beyond the maintenance of the Homo sapiens standard form. Leveraging emerging technologies in morphogenesis control and bioengineering, the objective shifts toward “radical persistence.” This involves the ability to refresh cellular goal-states or even transition the biological substrate into novel, more resilient configurations. The speaker posits that within 50 years, the human body will be fundamentally altered biologically and technologically. In this view, longevity is not the preservation of the “caterpillar” (current human form) but the facilitation of the “butterfly” (a technologically enhanced or morphed state of persistence). This necessitates a move from targeting single molecular pathways to mastering the top-down control of anatomical identity.
II. Insight Bullets
- Aging as Cognitive Failure: Aging is conceptualized as the breakdown of the information-processing system that keeps cells aligned toward a collective anatomical goal.
- Anatomical Goal-States: Morphogenesis is driven by “goals”; once maturation is complete, the lack of a secondary “maintenance goal” leads to collective disbandment.
- Cellular Agendas: When collective alignment fails, cells revert to “tiny individual agendas,” which manifests as cancer or tissue entropy.
- Bioelectrical Pattern Memory: Biological systems store the “memory” of shape in bioelectrical networks; the blurring of this memory is a primary driver of aging (Levin, 2021).
- Evolutionary Regress: Aging cells exhibit transcriptional signals that move “backwards” across the phylogenetic tree, losing specialized multicellular identity.
- Phylogenetic Disarray: Cells within the same aging body are “no longer on the same page” regarding their evolutionary gene expression profile.
- The Long View of Longevity: Longevity is not just about extending human life but about the persistence of the “self” through radical morphological changes.
- Morphogenesis Control: Future medical interventions will focus on controlling the high-level signals that dictate tissue shape rather than micro-managing molecular damage.
- Technological-Biological Convergence: Humans are predicted to undergo significant structural changes within 50 years, making current susceptibilities (like astigmatism or degeneration) obsolete.
- The “Caterpillar” Analogy: Persistent life may require radical transformation into novel forms rather than the static preservation of the current body.
- Beyond the Standard Human: The transcript challenges the “species-specific shape” as the only viable vessel for long-term consciousness and health.
- Bioelectrical Feedback Loops: Aging involves positive feedback cycles where physical dissociation further degrades the signaling needed for re-alignment.
IV. Actionable Protocol (Prioritized)
High Confidence Tier
- Bioelectric Diagnostics: Utilizing current tools to monitor physiological state via bioelectric markers (e.g., skin/nerve conductance) as an early indicator of tissue disarray (Levin et al., 2017).
- Information-Theoretic Health: Viewing health as a measure of “system integration.” Prioritize protocols that reduce systemic noise (e.g., maintaining circadian rhythm and stable metabolic environments to support cellular signaling).
Experimental Tier
- Morphoceuticals: Investigating ion channel-modulating drugs to “refresh” anatomical goal-states. This is currently limited to model organisms (e.g., planaria, xenopus) but represents the frontier of regenerative medicine (Levin & Martyniuk, 2018).
- Transcriptional Monitoring: Utilizing “Aging Clocks” that measure transcriptional noise and phylogenetic drift to assess biological age vs. chronological age.
Red Flag Zone
- Transhumanist Claims: Assertions that human anatomy will be “unrecognizable” in 50 years are highly speculative (Level E evidence) and lack a defined regulatory or biological roadmap.
- Radical Persistence: Protocols suggesting “becoming something else” (morphing substrates) are currently science fiction; no safety data exists for human morphogenetic altering.
- Phylogenetic Regress Control: There are currently no validated human protocols to “stop” evolutionary backtracking of gene expression.