Higher dose biweekly dosing seems to be the more in vogue option. Can anyone make for or against this approach?
We all believe rapamycin will do us good for health and longevity, but we don’t know the dosing. However, up to a certain level, it is safe. So I believe a scattershot approach is as good as any, and perhaps better than any fixed dose schedule. I’d advise that once one is accustomed to a certain dosage, sometimes slightly raise it, sometimes lower it, and occasionally take breaks.
As far as weekly or fortnightly, vary your schedule and do both. The results might not be as good the ideal schedule, but no one knows what is ideal.
Yes, I agree with what you said. I was hoping for a more rational approach that would convince me to do something else, but for now I will try as you have said.
Yes, my approach is akin to a mixed-strategy in game theory, where you can optimize your result (long life) by using more than one strategy.
Somebody, I don’t recall who, suggested that having some time with mTor acting however it will in the absence of rapamycin might be good. Mice have had continuing benefit after only one dose. The Mayo Fisetin and Quercetin/Dasatinib dosages allow a month off, then months and months off. It seems like weekly dosing would never let your body clear out all the rapamycin, making it closer to the daily dosing used for anti-rejection.
I’ve been dosing every fourteen days, but am going to move to nineteen. I got lab tests for peak and trough and know that that’s seven half-lives for me. Which is not to say I won’t hear something that will make me move to weekly. I’m following whatever seems to intuitively make sense.
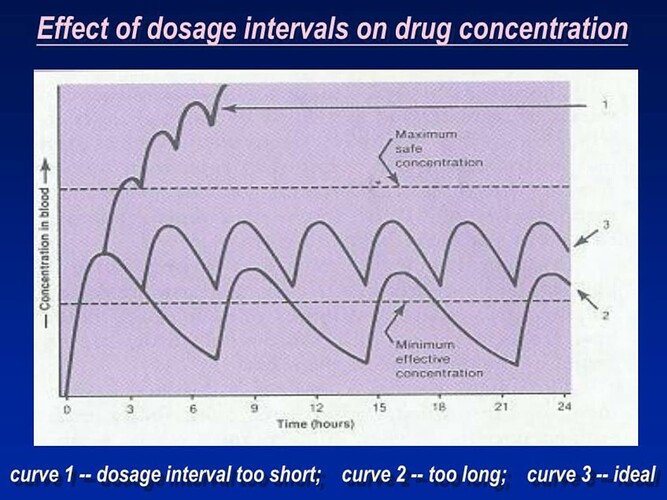
Just my own thoughts after 8 months of taking rapamycin: If you take higher doses, take it biweekly, smaller doses, take it weekly. The rationale for which I have no proof is the amount of rapamycin in the blood after 5 half-lives from a large dose might be the same as 2 1/2 half-lives of a smaller dose. You could do the math and figure out the equivalencies. At this point, we will have to wait to see which is best.
Mikhail V. Blagosklonny
@Blagosklonny
“MD/Ph.D. Professor of Oncology In Search of Immortality” seems to like the high dose biweekly routine, while Dr. Green seems to like the lower weekly doses. (At least for his patients)
We have positive peer reviewed data for weekly administration… we don’t for fortnightly. The people I trust the most (MK & PA) take it weekly.
that’s in mice though, mice live only around 2-3 years or so
The Joan Mannick human studies were all dosed weekly (mimicking the earlier mouse studies that they were replicating in humans).
The Dog Aging study is now weekly dosing (from Matt K., to lower risk of Side effects)
We have very little data on longer dosing periods. People are exploring longer dosing periods merely as a way to reduce risk of mTORC2 inhibition and thus any associated side effects of higher doses. People exploring higher doses because in mice, the higher the dose the longer the life (the peak doses for females were finally identified in the Transient dose protocols by U. Washington.
Mannick Study See: Rapamycin Clinical Studies in Healthy People (part 2)
I think that fundamentally the dosing period is just a result of the decision on dosing amount… If you want to take the approach (higher risk, but higher potential reward) strategy of pushing up to the higher dose, then you have to go to longer washout periods to avoid inhibition of mTORC2.
I think blood tests at trough levels (just before your next dose) are key if one is testing higher doses.
But mightn’t it be important to let mTORC1 come back to its full glory before tamping it back down again?
These are all good points, but don’t seem to moving our understanding of what the full mechanism is that we are trying to influence. Building the understanding as far I can make it out are:
-
mTor1 appears to a primary regulator in the aging process - its balance of being stimulated to grow and build vs. its inhibition to allow recovery and renewal seem to be a reasonable hypotheses.
-
Something about the aging process and some may argue poor health habits cause mTOR1 be stuck in the on position or as mentioned earlier stuck in first gear. This seems to cause inflammation and then increased risk factors for age related diseases.
-
Prolonged mTOR1 inhibition can lead to a disruption of mTOR2 being made and threaten the immune system with possible side effects to follow like infection / mouth sores.
-
When we are younger, mTOR1 seems to work better - maybe from less senescence cell than as we get older. This maybe the cause of mTOR sticking in the “on” position or I like the better the analogy of being stuck in first gear since it gives more variation of regulation ?? @tongMD
-
Pulsed dosing makes sense if at least some of the above is true. Knowing trough levels seem the most useful and then time sequence of when it is best to turn off / put in the clutch ( to bio-hack mTOR’s) with Rapamycin ??
Since true biomarkers for Rapa have only some help, the trial and error for the individual technique is where we are at until our understanding of the full mechanism is achieved and then how best to manipulate it. @EnrQay Your genetic make up probably has a big impact as well @tongMD
This feels like a Rubik’s Cube that we have one side solved for , but a way to go at solving it:) Thanks to all for your input. Will try a 2 week interval next.
We need mTORC1 for growth and healing, so we definitely need it not inhibited at some point/time and duration. The general consensus in the research community is that we need to cycle mTORC inhibition.
The issue is we don’t know how much we need it to “come back”, or exactly when do we need mTORC1 not inhibited (perhaps during or after peak exercise periods), or for how long. And the exact amount we want to inhibit (or not inhibit) mTORC1 it will vary over time depending upon any wounds or other issues you need to recover from.
There is so much we still don’t know on mTOR1 modulation in humans…
Dr. Brian Kennedy covers the mTOR modulation issue in this video here:
At about a minute in, we learn that rapamycin-fed old mice regain flexibility of mTOR, which age had stuck in a narrow range - running a constant high level. Rapamycin modulates it, so it runs high when needed and low when not needed - this after the mice have been dosed for a month. Further in, he speculates about taking rapamycin for a month then stopping for six months.
Thanks for the video, RapAdmin. It’s eight minutes that goes by in thirty seconds.
Interesting and important point made by Dr Kennedy here. During prolonged use of Rapamycin MTOR is still activated by exercise and diet because the sensitivity to the stimulus has been restored.
Here we go!! This video has the content that I was hoping for discussion. I like the word he uses of “modulation” vs. just on or off. Aging, whether by shortened telomeres, increased senescence cells OR elevated inflammatory pathways, all seem to make mTOR1 stick in certain modulation ( gear, vibration or even non pulsatile ) state that it looses its feed back loop of regulation. Taking Rapamycin seems to unstick mTOR from its pro-aging state and allow it to function more as it did when younger.
I really like this model Dr. Kennedy is proposing because it gives us a better idea on mechanisms that we are trying to improve and thus better dosing strategies for now.
My take aways were:
-
Even some short exposure to Rapamycin had improvements in longevity and mTOR modulation - mice exposed to one month of Rapamycin and then not given it had a 15% improvement in longevity. This may support taking longer periods off Rapamycin
-
Rapamycin seems to NOT just turn off mTOR1C, but rather help it modulate better at being more flexable and restore it more dynamic ranges of YOUTH.
After viewing this video, I am not sure if higher doses are as important, instead, it maybe whatever dose that helps restore the proper modulation rather the highest dose possible, as long as it does not inhibit mTOR2. Using some blunt signs and symptoms - lower inflammatory states with decreased muscle pain and stiffness and some weight loss / decreased visceral fat seem to be not unreasonable markers.
@RapAdmin Great content, as always! - I attached a video below for those who like to nerd out on the mechanistic pathway.
mTOR Signaling Pathway: mTOR Complexes, Regulation and Downstream effects - YouTube
The more important (only relevant IMHO) question is how do we increase therapeutic dosing (WITH proper biomarkers), without side effects. But let’s leave that for a separate thread.
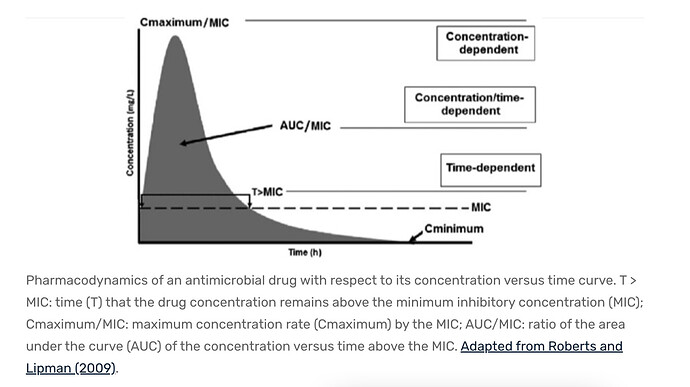
Dr B is a constant proponent for “as much as you can take without side effects”, ergo, typical high AUC for optimal drug therapy. The seminal cancer/rapamycin study used weekly dosing with target AUC maximization. Most patients on rapamycin for cancer/transplants are on daily with target trough blood (indirectly AUC) levels. The rapamycin/mice studies, the mice are subjected to typically daily very high rapamycin blood levels (but they have vastly higher metabolism, and drug elimination), have significantly reduced mTOR, and extended lifespan. The higher the dose (higher AUC), the higher the lifespan extension.
Current human translation for longevity is a complete unknown.
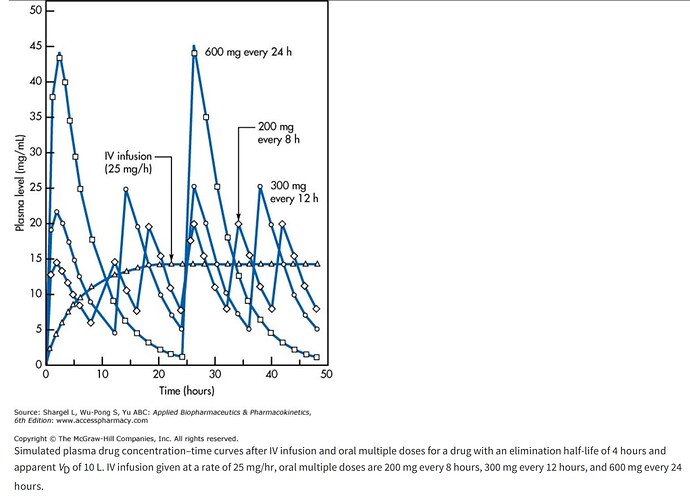
As for the dosing strategy, you can generate an infinite number of dose/time/AUC curves for rapamycin, staying just under toxic (dose limiting side effects) concentrations, but above a minimum. Most all of the studies I’ve read on humans (cancer/transplant), it’s either daily (one study of weekly). In mice, vast majority are daily.
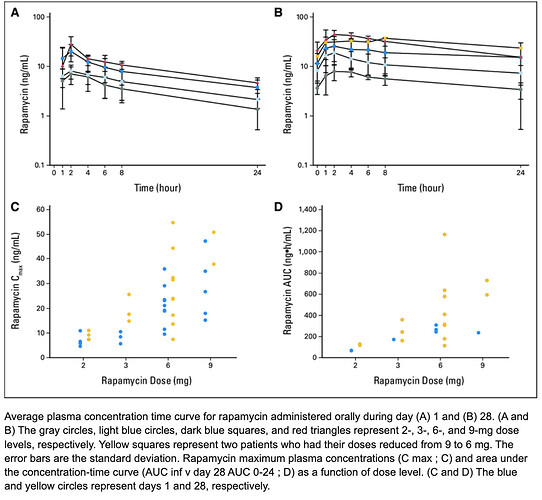
We know in a single human study, rapamycin does cross the BBB, and the higher the peripheral dose, the higher the brain concentration of rapamycin (2, 5, or 10mg daily). So this may be one rationale for a higher dose spread over a longer period (to get a short peak burst, but you dampen AUC, and lessen side effects). But I would argue for human lifespan extension, for the greatest mTOR inhibition (the main target pathway), we would want the highest AUC just below toxic, and that is more likely (ie mathematically) via lower dose but increased dosing frequency. All things being equal.
Dr Green on his 12mg two days in a row, followed by longer period, is just another iteration at dose/time/peak experimentation.
Pharmacodynamic-Guided Modified Continuous Reassessment Method–Based, Dose-Finding Study of Rapamycin in Adult Patients With Solid Tumors
One problem with dosage/frequency is, what are the adverse side effects to look for.
Taking blood tests at ~2-month intervals as I am now doing allows some things to get too far out of whack for my taste.
I was using B’s biweekly high-dose regimen. Didn’t know what to look for until I got diarrhea from 20 mg. with grapefruit juice. I actually did this 3 times to be sure of the causation.
Then I backed off to 10 - 15 mg biweekly with grapefruit juice, this stopped the diarrhea problem. Then, I decided I didn’t like the way my cholesterol markers were going. Now I am resting from rapamycin to see how my blood markers look after a washout period.
When I begin again I think I will start with 5 mg. weekly with grapefruit juice.
To each his own, you seem to have way more experience than most of us with your dosing sortees and n=1 side effects.
Cholesterol and glucose has been tossed around as benevolent dysregulation…is it? Perhaps a slight rise with good starting markers is acceptable in return for possible longevity benefits?
Lipids and lymphocytes are typically the biggest human side effects. Some people might bail on some components of CBC going below lab range.
Dr B dosent define “tolerable” re side effects re max dosing. We all have our own risk calculus, which he is saying implicitly.
Not side stepping your legitimate question, but if longevity is the prime objective, then the science says to maximize mTOR inhibition.
Uncharted territory.
I like your detailed breakdown. Thank you!
I am very interested in MOA since I think that can help guide the dosing dilemma. I think that mTOR1 inhibition is only our preliminary finding and that as we get more information that there is much more involved. I respect Dr G and Dr. B for being true pioneers, but the highest dose you can handle seems to be the trend and this seems to be missing the other moving parts of what Dr. Kennedy is referring to as reseting the modulation and not completely blocking mTOR, but helping to reset its flexibility more similar to what occurs in youth.
Biomarkers and subjective feedback are what we have. I don’t know if improvement in markers always means “good” and that a slide of markers always means “bad”. This creates an issue if you have conflict of a subjective good or bad in oposition with a biomarker that is good or bad…I feel great, but my markers look awful and or I feel like crap, but my markers look great and I should live to 130 ![]()
Always appreciate your detailed insights!
LOL I haven’t always had good markers, but I felt zero correlation with my markers as to how good I have felt. I’ll bet there are many among us that occasionally had great markers but didn’t feel that great and vice versa.