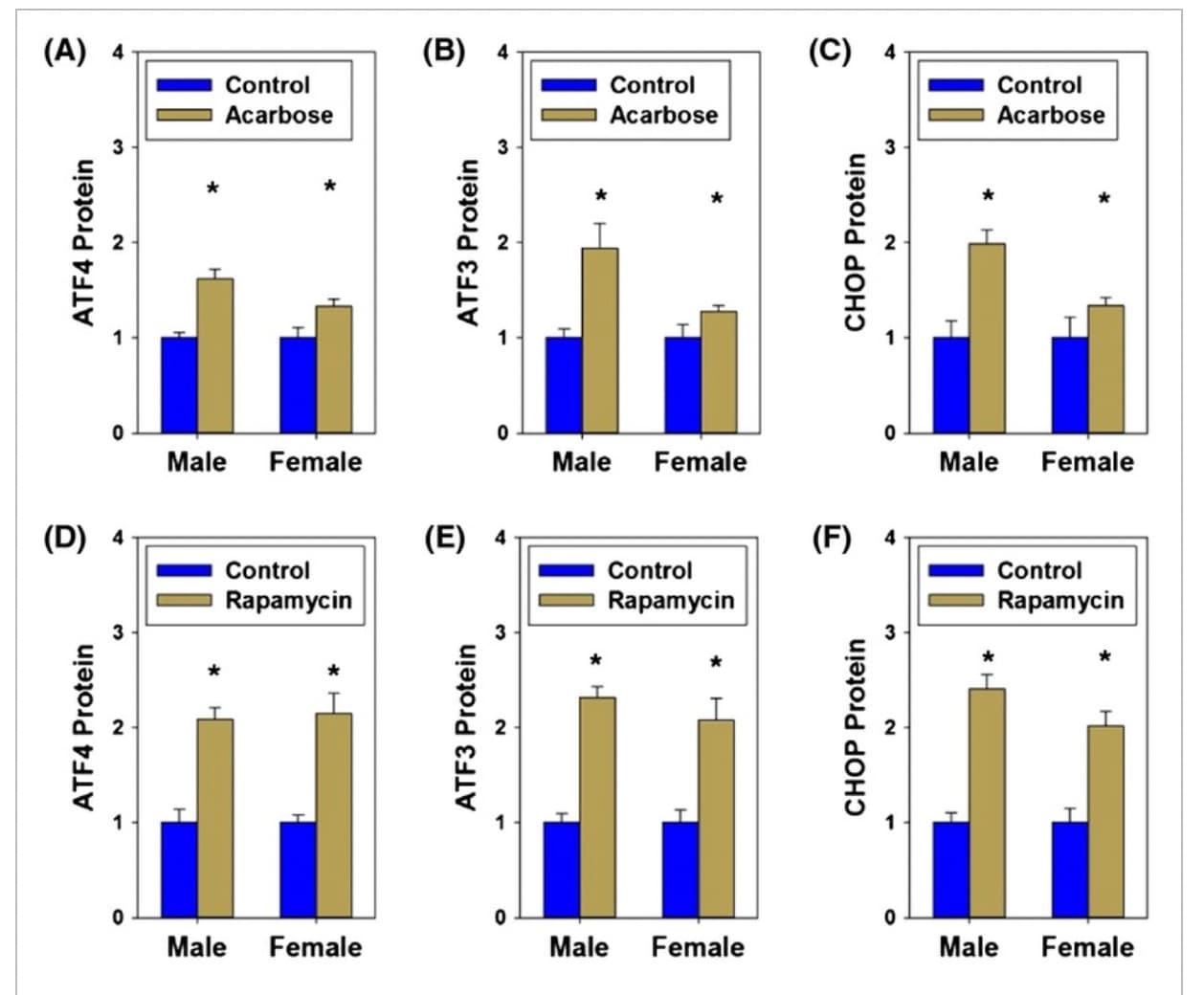
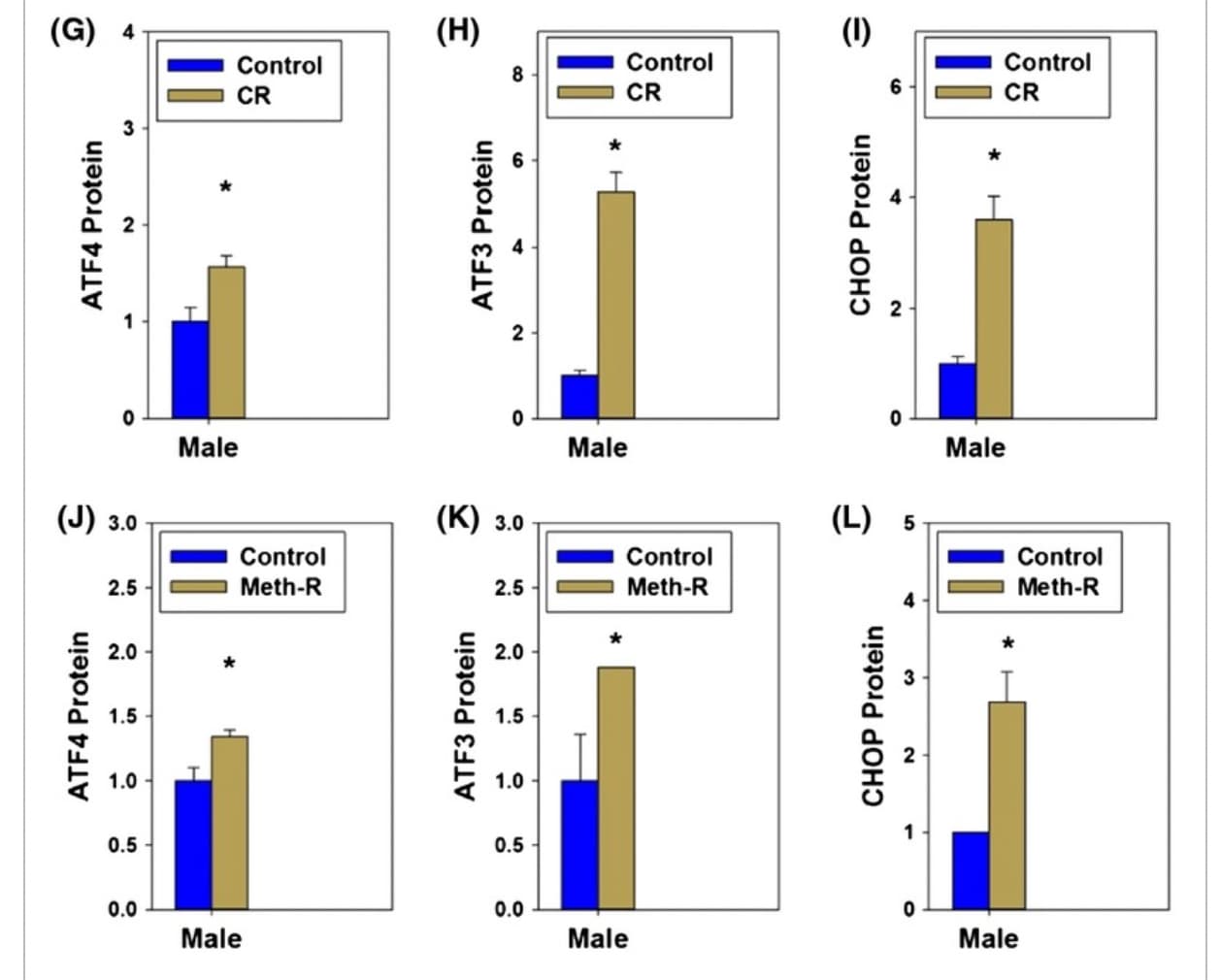
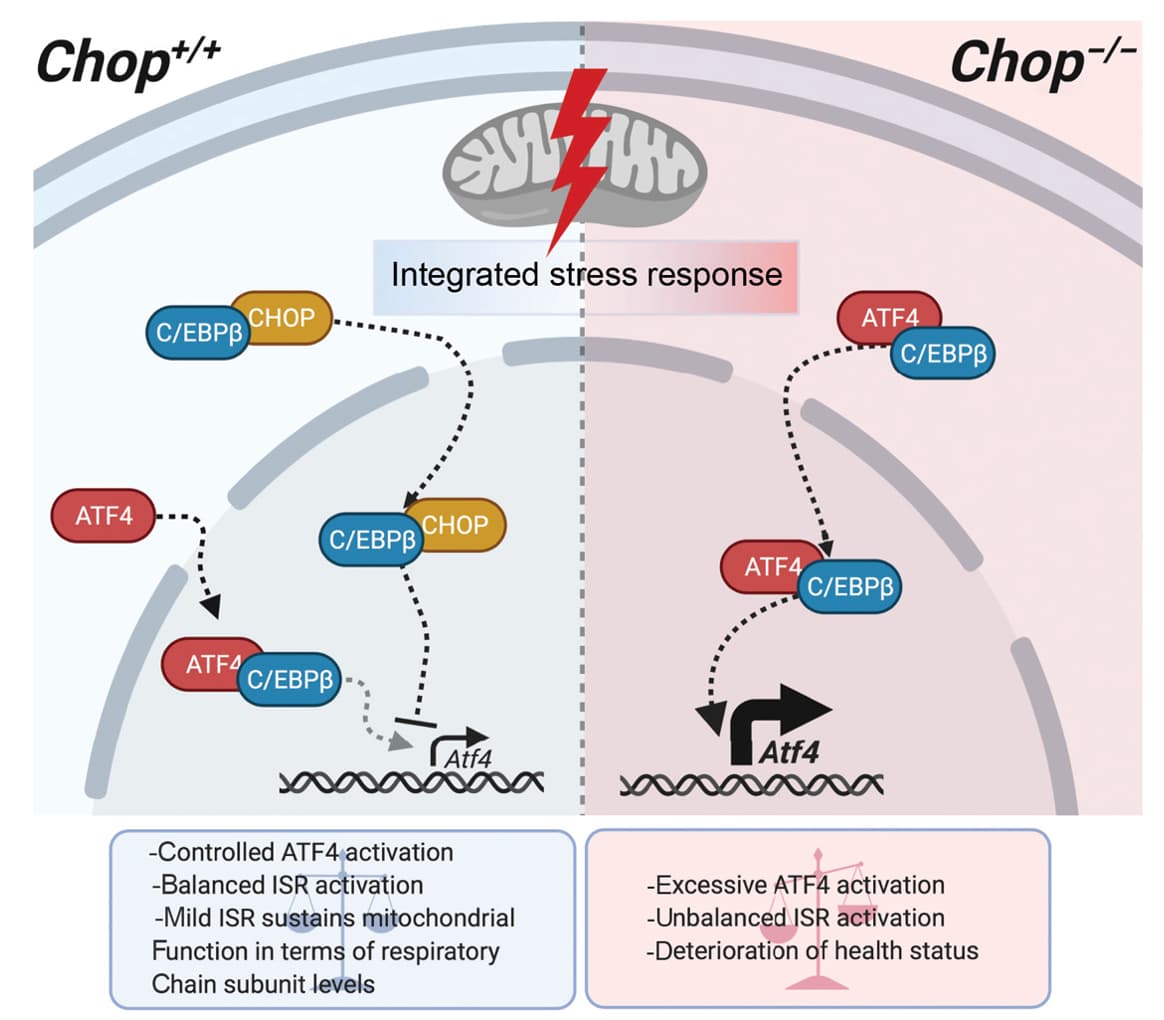
Last year it was announced that ISRIB was out-licensed from UCSF to Alphabet / Google’s Calico, based on the great pre-clinical data that UCSF had developed, in terms of brain function / memory results.
Normal mice put inside a watery maze took more than a minute to locate the submerged platform that would let them escape. But these mice — which had been injected with a curious new molecule — found it in an average of just 16 seconds. (source)
Just a few doses of an experimental drug can reverse age-related declines in memory and mental flexibility in mice, according to a new study by UC San Francisco scientists. The drug, called ISRIB, has already been shown in laboratory studies to restore memory function months after traumatic brain injury (TBI), reverse cognitive impairments in Down Syndrome, prevent noise-related hearing loss, fight certain types of prostate cancer, and even enhance cognition in healthy animals.
Source: Drug Reverses Age-Related Mental Decline Within Days
Full Research Paper: Small molecule cognitive enhancer reverses age-related memory decline in mice
I’ve not seen any human clinical trial data, and there are no clinical trials of ISRIB listed on the ClinicalTrials.gov website.
ISRIB seems to be available easily from lab / reagent supply companies, and I’ve seen that some people are starting to do their own personal clinical trials with it. With no human clinical data at all on this compound, that seems a little too aggressive for me personally.
However, in this January, 2021 update it was said:
Human Trials In The Offing
A review co-authored by Walter and published last year in Science (DOI: 10.1126/science.aat5314) provides an overview of diseases of the brain associated with ISR activation. In addition to TBI and age-related cognitive decline, the list included frontotemporal dementia, Alzheimer’s disease, Parkinson’s disease, Huntington disease, amyotrophic lateral sclerosis, multiple sclerosis, Down syndrome (characterized by a high incidence of Alzheimer’s), Charcot-Marie-Tooth disease (a group of inherited disorders causing nerve damage), vanishing white matter disease (genetic disorder where the body doesn’t make enough myelin), and prion disease (involving abnormally folded proteins).
Recent evidence has shown that the ISR serves as a “universal regulator” of long-term memory formation, they write. Inhibiting the ISR enhances this process, while activation of the IRS prevents it.
The good news, is that in mice at least, it was highly non-toxic.
Since ISRIB has proven to be completely non-toxic in mice when working with a ISRIB derivative for TBI applications, Rosi says she and Walters are “really motivated to… push it to humans as soon as possible. Every day, I get at least five to 10 emails from people suffering asking if they can be in any clinical trials.”
An Educational Overview of ISRIB
Source: Promise of ISR Inhibitors In Treating Age-Related Cognitive Decline
Related Research papers:
ISRIB Related Reading
From: For Researchers | Cognitive Vitality | Alzheimer's Drug Discovery Foundation
ISRIB.pdf (304.4 KB)