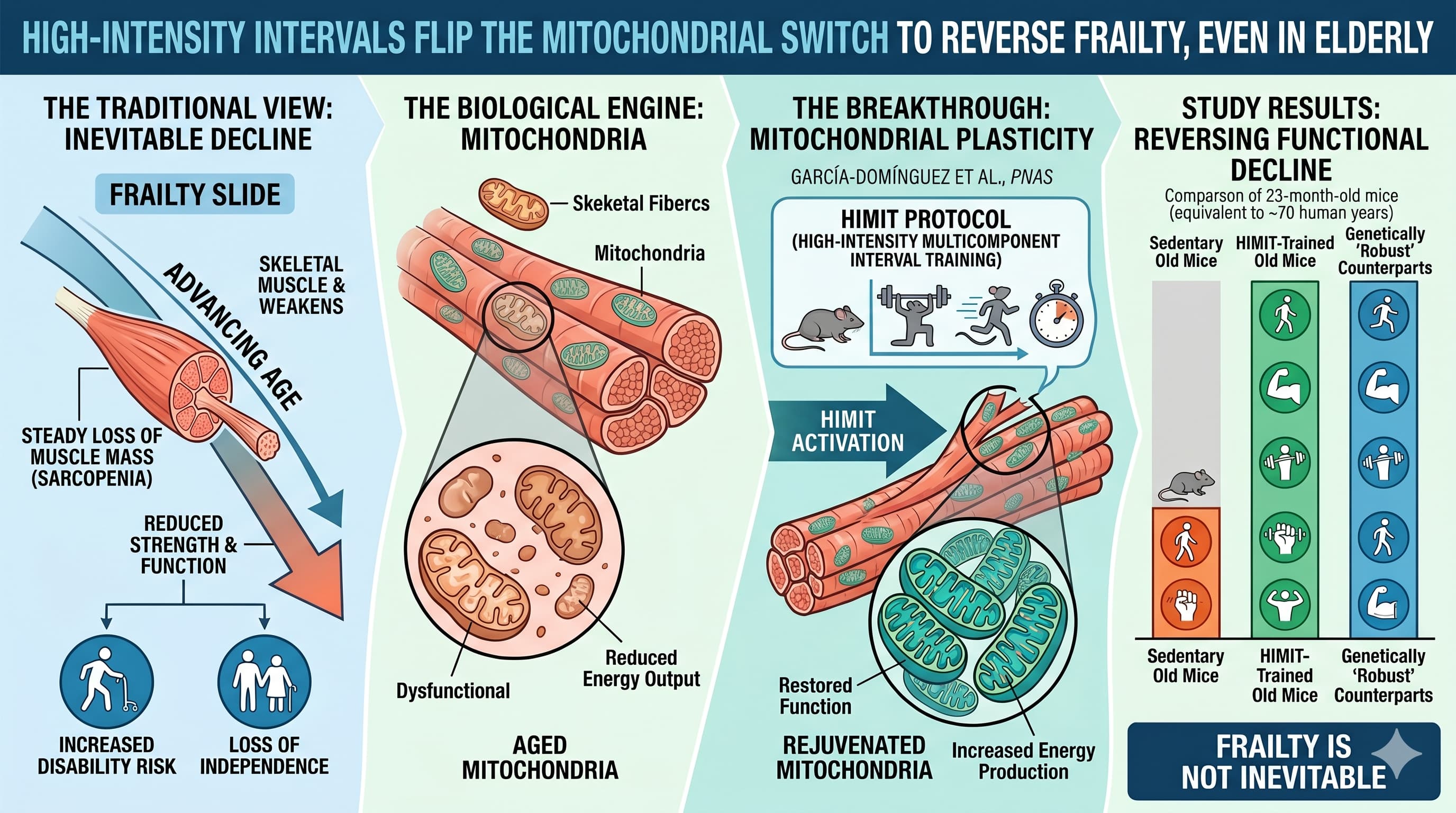
Frailty is often viewed as an inevitable slide into disability, driven by the steady loss of muscle mass and strength. However, new research published in PNAS by García-Domínguez et al. suggests that the “biological engine” of our muscles—the mitochondria—retains a surprising level of plasticity even in extreme old age. By utilizing a high-intensity multicomponent interval training (HIMIT) protocol, researchers demonstrated that 23-month-old mice (roughly equivalent to 70-year-old humans) could effectively reverse functional decline and match the performance of genetically “robust” counterparts.
The study identifies mitochondrial remodeling as the primary driver of this reversal. Aging typically degrades the structural and enzymatic efficiency of mitochondria, leading to energy deficits and increased oxidative stress. The research showed that high-intensity exercise forces these organelles to reorganize into “supercomplexes,” significantly boosting their energy-producing capacity (OxPhos) while simultaneously lowering the production of harmful reactive oxygen species.
Crucially, the team proved that these benefits are not merely correlations. By using a muscle-specific knockout model (Usmg5) that lacks a key subunit for ATP production, they demonstrated that without functioning mitochondria, exercise fails to improve physical performance. This confirms that mitochondrial integrity is the mechanical prerequisite for healthy aging.
The findings were further validated in human subjects ranging from 17 to 99 years old. Older individuals who maintained high physical function exhibited mitochondrial signatures nearly identical to younger adults, characterized by high G6PD activity and preserved respiratory complex function. In contrast, sedentary older adults showed a marked shift toward inefficient glycolytic metabolism and increased systemic inflammation. This research shifts the geriatric paradigm from managing decline to actively “remodeling” the cellular infrastructure of aging muscle.
Actionable Insights
-
Intensity is Essential: The HIMIT protocol—combining resistance (ladder climbing), motor coordination (rotarod), and high-intensity cardiorespiratory intervals—successfully reversed frailty where moderate activity often fails.
-
Targeting G6PD: Glucose 6-phosphate dehydrogenase (G6PD) emerged as a critical antioxidant enzyme. Strategies to maintain G6PD activity, whether through specific exercise or potential future compounds, appear vital for protecting muscle from oxidative lipid damage.
-
Function Over Age: The human data indicates that chronological age is a poor predictor of muscle health compared to “functional status”. High-functioning octogenarians maintain mitochondrial volumes and antioxidant defenses comparable to those in their 30s.
-
Mitochondrial Plasticity: Because mitochondria remain adaptable throughout the lifespan, it is never “too late” to initiate high-intensity interventions to rescue locomotor function and reduce the risk of falls and dependency.
Context
2 Likes
Mechanistic Deep Dive
The study maps functional reversal to specific molecular pathways:
-
Mitochondrial Dynamics & Supercomplexes: Exercise training significantly enhanced the assembly of respiratory supercomplexes (Complexes I, III, and IV). This structural remodeling improves electron transfer efficiency and reduces electron leakage. [Confidence: High]
-
Antioxidant Defense (G6PD): High-intensity exercise increased the activity of G6PD, which is critical for NADPH production and glutathione-mediated ROS detoxification. This led to a marked decrease in malondialdehyde (MDA), a marker of lipid peroxidation. [Confidence: High]
-
OxPhos Plasticity: Proteomic analysis (MitoCarta 3.0) confirmed a broad enrichment of Oxidative Phosphorylation pathways in both the trained mice and high-functioning “Older with Good Function” (OGF) humans. [Confidence: High]
-
Immune/Inflammatory Axis: Poor physical function in humans (OPF) correlated with an upregulation of immune effector processes and chronic low-grade inflammation, a phenotype partially mitigated by the metabolic health of the OGF group. [Confidence: Medium]
Novelty
This paper moves beyond mere correlation by proving that intact mitochondrial function is a causal requirement for exercise-induced rejuvenation. The use of the Usmg5 mKO model demonstrates that without the ability to dimerize ATP synthase and maintain efficient OxPhos, the physiological benefits of training are abolished. Additionally, the high-throughput proteomic comparison between “robust” transgenic mice and high-functioning humans provides a rare “concordance map” for healthy aging.
And a related type of study:
Lifting the Weight of Age: Why Your Mitochondria Care More About the Gym Than Your Mirror
Resistance training (RT) has long been the gold standard for combating sarcopenia—the age-related decline in muscle mass and strength that costs the U.S. healthcare system over $40 billion annually. However, a new narrative review from researchers in China reveals a frustrating “translational gap”: while we understand how lifting weights builds muscle in young rats, those molecular pathways often stutter or fail in the bodies of older humans.
The “Big Idea” here is that muscle mass is a fickle metric for longevity. The study highlights that while older adults frequently fail to see significant muscle size increases (hypertrophy) due to “anabolic resistance,” they still gain substantial strength and functional independence. The secret lies not in the size of the fiber, but in the health of the engine. Mitochondrial quality control (MQC) emerges as the most consistent biological winner across species. RT forces the body to purge broken mitochondria and streamline energy production, effectively “re-coupling” respiration even when the scale doesn’t budge.
Furthermore, the review introduces the Hippo-YAP pathway as the potential “mechanostat” of the muscle—a molecular hub that senses physical tension and tells satellite cells (muscle stem cells) whether to wake up and repair or stay dormant. While promising, this remains largely hypothetical in humans. For the aging population, the message is clear: the biological benefits of the gym are happening at a cellular level that your bathroom scale is currently unable to detect.
Actionable Insights
For those seeking to maximize healthspan and combat sarcopenia, the following practical protocols are supported by the evidence:
-
Volume Over Intensity for Anabolic Resistance: To overcome the “blunted” protein synthesis seen in aging, doubling training volume (more sets) is more effective than simply increasing the weight. Low-load RT (40% 1RM) performed with higher sets can produce hypertrophy comparable to high-load training (80% 1RM) in older adults.
-
The 12-Week Threshold: Meaningful improvements in mitochondrial respiration and muscle function require a minimum of 12 weeks of consistent adherence.
-
Frequency and Recovery: Aim for 2–3 sessions per week. Critically, older muscle requires at least 48 hours of recovery between sessions to manage neuromuscular fatigue and optimize adaptation.
-
Strategic Compound Movements: Prioritize multi-joint exercises (e.g., leg press, rows) that target the lower limbs, as these are the primary drivers for maintaining gait speed and preventing falls.
-
Personalized Intensity: Robust seniors should target 60–80% of their one-repetition maximum (1RM), while frail individuals should start at 50–55% 1RM, focusing on movement control before adding load.
Context
1 Like