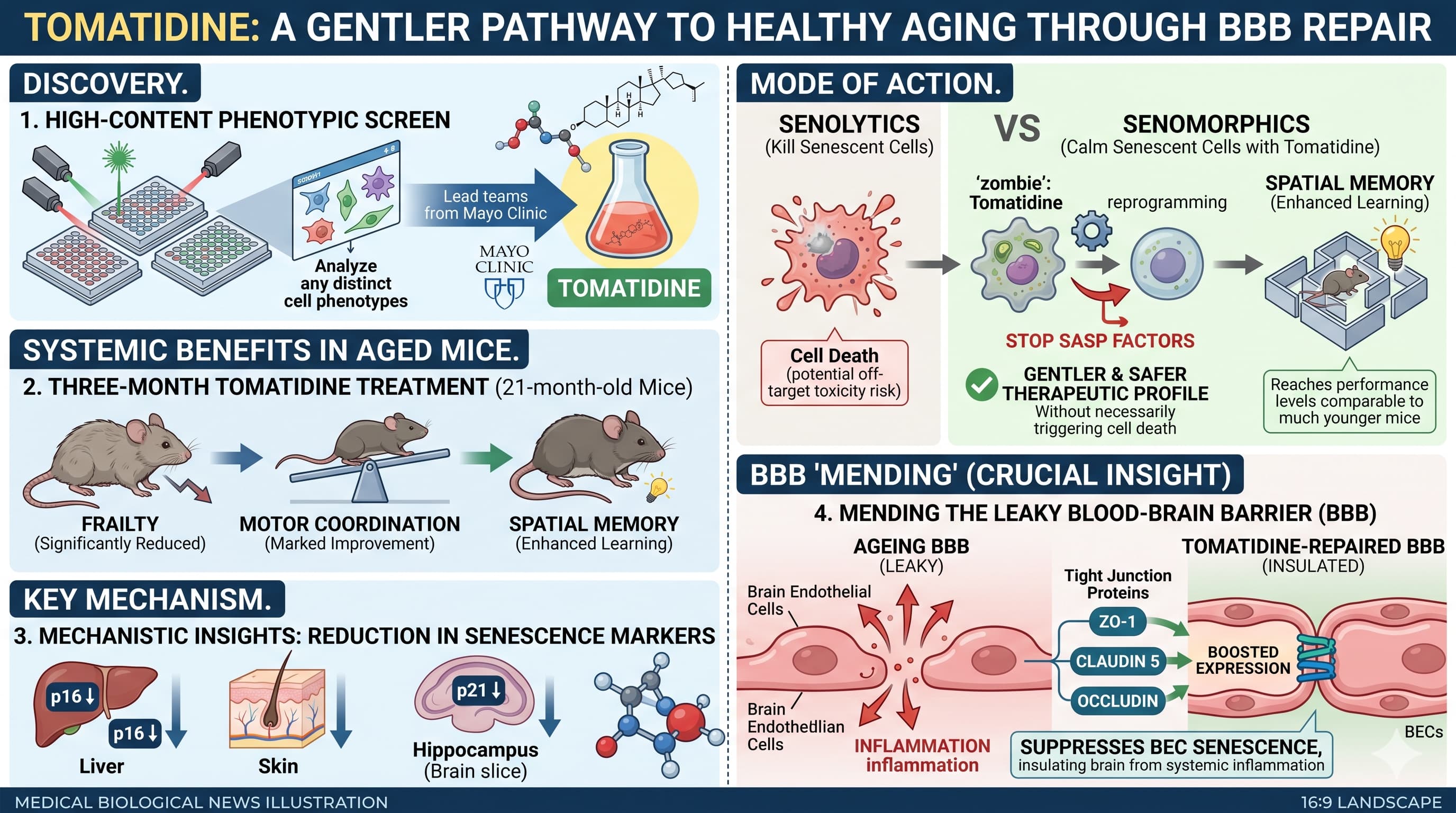
Recent research published in EMBO Molecular Medicine identifies tomatidine —a natural compound found in green tomatoes—as a potent “senomorphic” agent capable of rejuvenating the aging brain and strengthening the blood-brain barrier (BBB). As we age, our tissues accumulate senescent cells: “zombie” cells that refuse to die and instead secrete a toxic cocktail of pro-inflammatory factors known as the SASP (senescence-associated secretory phenotype). In the brain, this cellular pollution triggers neuroinflammation, degrades the BBB, and erodes cognitive function.
The researchers, led by teams from the Mayo Clinic , utilized a high-content phenotypic screen to discover that tomatidine effectively calms these “zombie” cells. Unlike “senolytic” drugs that aim to kill senescent cells—sometimes with off-target toxicity—tomatidine acts primarily as a senomorphic. It reprogrammed the cells to stop producing harmful SASP factors without necessarily triggering cell death, offering a potentially gentler and safer therapeutic profile.
When administered to aged mice (21 months old) for just three months, tomatidine produced striking systemic and neurological benefits. The treated animals showed a significant reduction in frailty and marked improvements in motor coordination and spatial memory , reaching performance levels comparable to much younger mice in learning tasks.
Mechanistically, the study found that tomatidine reduced senescence markers like p16 and p21 across the liver, skin, and hippocampus. Crucially, it targeted brain endothelial cells (BECs), which form the structural foundation of the BBB. By suppressing senescence in these cells, tomatidine boosted the expression of essential “tight junction” proteins like ZO-1 , claudin 5 , and occludin. This suggests a “mending” of the leaky BBB that typically occurs with age, effectively insulating the brain from systemic inflammation.
With its high oral bioavailability and established safety in preclinical models, tomatidine represents a promising, naturally derived strategy to extend healthspan and preserve cognitive integrity in late life.
Actionable Insights
The primary takeaway for those interested in longevity is that tomatidine is a viable, naturally occurring candidate for mitigating age-related cognitive and physical decline.
-
Source and Bioavailability: Tomatidine is the aglycone of tomatine , found primarily in unripe (green) tomatoes and the leaves of the tomato plant. Importantly, it demonstrates high oral bioavailability , meaning it can be effectively absorbed through the diet or supplementation.
-
Safe Intervention: In this study, a 3-month dietary intervention (0.05% tomatidine) in aged mice produced no detectable adverse effects or behavioral changes, supporting its favorable safety profile for late-life administration.
-
Cognitive and Physical Protection: The compound specifically targets neuroinflammation and blood-brain barrier integrity. This translates to improved spatial memory and motor coordination, suggesting it may help “insulate” the brain from the broader systemic effects of aging.
-
Senomorphic Strategy: Unlike harsher senolytics, tomatidine’s primary action is senomorphic —it reduces the toxic secretions (SASP) of senescent cells rather than just killing them. This makes it a strategic option for those looking to modulate the “cellular environment” with lower risk of tissue depletion.
Source:
- Open Access Paper: Tomatidine is a senotherapeutic compound that improves cognitive function and reduces cellular senescence in aged mice
- Institutions: Department of Physiology and Biomedical Engineering, Mayo Clinic , Rochester, MN, USA; Center for Neuroscience and Cell Biology, University of Coimbra , Portugal.
- Journal: EMBO Molecular Medicine.
- Impact Evaluation: The impact score of this journal is 11.1, evaluated against a typical high-end range of 0–60+, therefore this is a High impact journal.