Part 4: Actionable Intelligence
The Translational Protocol (Rigorous Extrapolation)
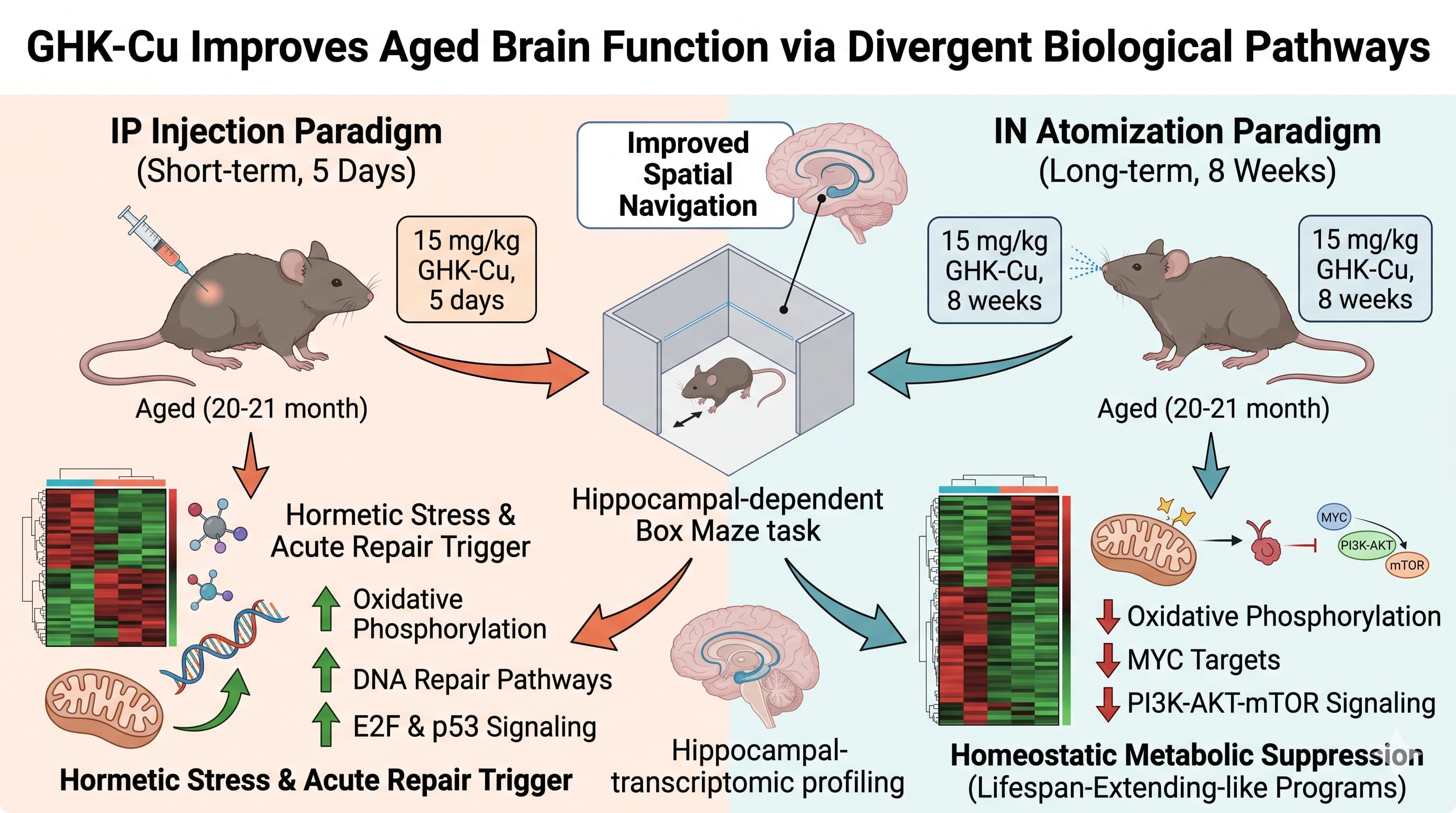
- [cite_start]Human Equivalent Dose (HED): The murine dose utilized in both paradigms was 15 mg/kg[cite: 144]. To translate this to human parameters, we apply the FDA’s Body Surface Area (BSA) normalization method.
- Calculation: Animal Dose (mg/kg) x (Mouse Km / Human Km)
- Math: 15 x (3 / 37) = 1.21 mg/kg.
- For a standard 70 kg human, the calculated HED is 84.7 mg/day. This dose is exceptionally high compared to standard clinical and cosmetic peptide protocols, which typically range from 1 to 5 mg daily.
-
Pharmacokinetics (PK/PD):
-
Half-life: Plasma half-life of systemic GHK-Cu is short, generally cited in external literature as 2 to 4 hours.
-
Bioavailability: Highly bioavailable via parenteral routes. [cite_start]Intranasal administration specifically bypasses first-pass hepatic metabolism and the blood-brain barrier (BBB) via olfactory and trigeminal nerve pathways[cite: 396].
-
Safety & Toxicity:
-
NOAEL / LD50: Safety Data Absent in this study.
-
Phase I Safety Profile: While topical and low-dose injectable formulations demonstrate high tolerance and minimal systemic toxicity, there is no formal Phase I safety data for chronic 85 mg/day intranasal delivery in humans.
-
Liver/Kidney/CYP450: High-dose systemic copper chelate therapy requires strict monitoring of ceruloplasmin and unbound serum copper to prevent heavy metal toxicity or Wilson-like hepatic accumulation.
Biomarker Verification
To verify target engagement in humans without direct hippocampal transcriptomics, peripheral proxy markers must be monitored:
-
For Intranasal GHK-Cu (Metabolic Dampening): Monitor systemic proxies for mTOR suppression and mitochondrial load. Primary markers include IGF-1, fasting insulin, and targeted metabolomic panels evaluating oxidative phosphorylation byproducts.
-
For Intraperitoneal GHK-Cu (Acute Stress/Repair): Track acute phase reactants such as hs-CRP, IL-6, and localized inflammatory cascades.
Feasibility & ROI
-
Sourcing: GHK-Cu is readily available as an over-the-counter topical, a compounded injectable (prescription), and a bulk lyophilized powder from chemical suppliers.
-
Cost vs. Effect: Achieving the 84.7 mg/day HED equates to approximately 2.5 grams per month. Procuring bulk powder costs roughly 60 to 150 USD for this volume. However, sourcing this via clinical compounding pharmacies would elevate the cost to thousands of dollars monthly. Given the translational uncertainty, the ROI for an 85 mg/day intranasal protocol is currently speculative.
Part 5: The Strategic FAQ
1. The 15 mg/kg dose is massive when translated to a human protocol. Was this selected to account for poor atomization efficiency, and what is the actual CNS concentration? The 15 mg/kg dose was selected based on established efficacy and safety relative to known copper toxicity thresholds. However, the researchers failed to measure pharmacokinetic brain concentrations. Without direct tissue quantification, it is unknown if the CNS exposure was high, or if the large systemic dose simply compensated for low anatomical delivery efficiency across the nasal mucosa.
2. Could the divergence between IN and IP molecular effects be purely pharmacokinetic rather than biological remodeling? Yes. The authors acknowledge this limitation. IP administration creates rapid systemic spikes and clearance, triggering acute stress-response pathways. IN delivery provides a sustained, lower-level exposure over 8 weeks. The transcriptomic divergence could simply reflect the difference between acute chemical shock and steady-state adaptation.
3. If IN GHK-Cu suppresses oxidative phosphorylation and mTOR, does it blunt exercise adaptations or synaptic plasticity in healthy subjects? Suppressing MYC targets and PI3K-AKT-mTOR signaling creates a low-growth state. While this is protective against age-related hyperfunction, it theoretically risks blunting the anabolic responses required for new synaptic formation, muscle hypertrophy, or exercise adaptation in subjects who are not already biologically aged.
4. How do you rule out that the IP “repair” signature isn’t just a toxic stress response to copper spiking? The study cannot rule this out. The short-term IP dosing triggered DNA repair and inflammatory TNF-alpha/NF-kB pathways in females, which are canonical responses to metabolic stress or tissue injury. It is plausible the IP method induced a mild, transient toxicity that improved maze times through acute stress-arousal rather than genuine neural rejuvenation.
5. Why was there no young control cohort used to benchmark “youthful” transcriptomics? The study lacked a young reference cohort. Consequently, it is impossible to determine whether IN GHK-Cu restored the aged hippocampus to a younger baseline, or if it simply pushed the tissue into a novel, alternative compensatory state that functions better in a spatial maze.
6. GHK-Cu is highly stable, but does atomization for IN delivery degrade the copper chelate bond before it reaches the olfactory bulb? GHK-Cu is a robust complex, but the biochemical environment of the human nasal mucosa (pH variations, mucociliary clearance) poses physical and enzymatic barriers not fully modeled in murine anatomy. Delivery efficiency of the intact chelate remains unquantified here.
7. Did the study monitor systemic copper toxicity parameters during the 8-week protocol? No. The paper does not report systemic toxicity panels, liver enzymes, or serum copper metrics. Extrapolating a continuous, high-dose copper-complex therapy without this data is hazardous due to the risk of copper accumulation.
8. Is the improvement in the Box Maze specific to hippocampal neurogenesis, or simply an overall reduction in age-related anxiety? The Box Maze relies heavily on spatial learning. While the study found changes in GFAP and synaptophysin, confirming structural modulation, the behavioral assay does not strictly isolate memory from reduced anxiety or improved locomotion. The performance improvement could partially stem from modified behavioral states.
9. How does GHK-Cu interact with established gerotherapeutics that also suppress mTOR? Interaction Check: Both IN GHK-Cu (in females) and rapamycin suppress mTOR signaling. Co-administration could lead to excessive down-regulation of mTORC1, potentially blunting necessary cellular repair, immune function, and wound healing. Stacking this with metformin or SGLT2 inhibitors requires extreme caution to avoid severe metabolic dampening or clinical frailty.
10. What specific downstream clinical data is required before off-label IN GHK-Cu should be deployed for age-related cognitive decline? Clinical translation requires human CSF pharmacokinetic assays to prove BBB bypass, a rigorous dose-escalation Phase I trial to establish a safe human NOAEL for chronic dosing, and an RCT utilizing precise neurocognitive testing (e.g., MoCA) to confirm that murine transcriptomic shifts yield functional neuroprotection in humans.