Part 3: Claims & Verification
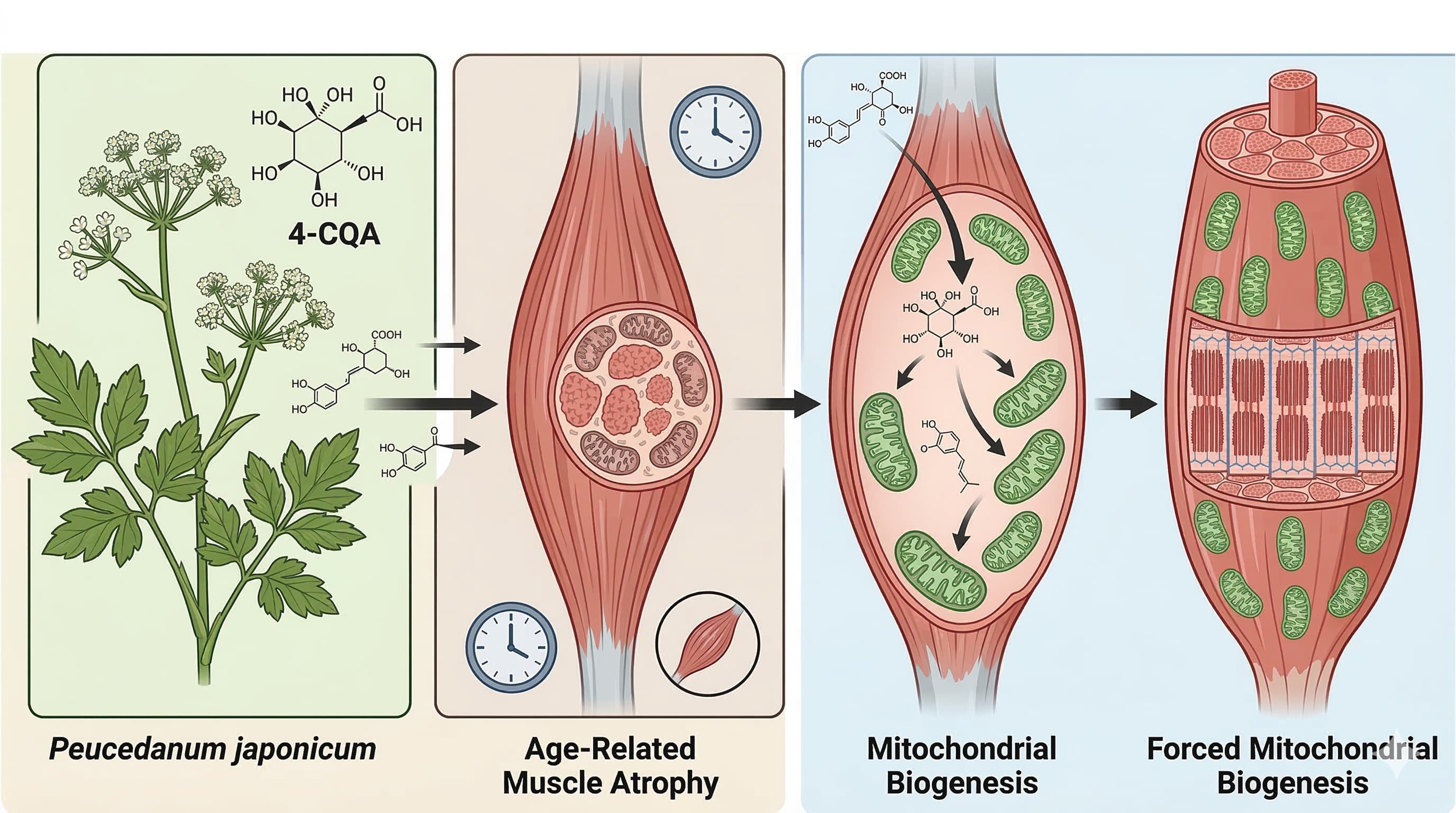
Claim 1: Peucedanum japonicum (PJ) and its metabolite 4-caffeoylquinic acid (4-CQA) reverse sarcopenic progression and increase lean body mass.
-
Evidence Level: Level D (Pre-clinical). [FLAG: Translational Gap]
-
Verification & Analysis: Live searches reveal zero human Randomized Controlled Trials (RCTs) or meta-analyses evaluating PJ or isolated 4-CQA for sarcopenia in human subjects. The primary source for this claim is a 2026 murine study. While some clinical data suggests that consuming coffee rich in chlorogenic and caffeoylquinic acids can induce minor improvements in muscle mass percentages in overweight adults, extrapolating targeted sarcopenia reversal from botanical extracts in mice to aging humans is highly speculative. Full answers require human longitudinal studies tracking skeletal muscle mass index (SMI) in sarcopenic populations.
-
Supporting Citations:
Claim 2: 4-CQA upregulates PGC-1alpha and stimulates mitochondrial biogenesis via the Nrf1-TFAM pathway.
-
Evidence Level: Level D (Pre-clinical). [FLAG: Translational Gap]
-
Verification & Analysis: The mechanistic pathway (PGC-1alpha → Nrf1 → TFAM) is a biologically verified driver of mitochondrial biogenesis. However, the claim that oral 4-CQA predictably forces this pathway in aging mammalian muscle relies entirely on in vitro assays and mouse models. There is no human biopsy data confirming that oral supplementation of 4-CQA bypasses hepatic first-pass metabolism to achieve the necessary intracellular concentrations to activate this axis in human skeletal muscle.
-
Supporting Citations:
Claim 3: 4-CQA halts muscle catabolism by suppressing the ubiquitin-proteasome system (downregulating Atrogin-1 and MuRF1).
-
Evidence Level: Level D (Pre-clinical). [FLAG: Translational Gap]
-
Verification & Analysis: Atrogin-1 and MuRF1 are established atrogenes responsible for muscle wasting. Research on various polyphenol metabolites (such as 3-HMPA, a downstream metabolite of caffeoylquinic acids) demonstrates targeted suppression of these ligases in C2C12 mouse myotubes exposed to atrophy-inducing agents like dexamethasone. However, demonstrating this effect in isolated rodent cells or aged mice does not guarantee identical proteasome suppression in human clinical applications.
-
Supporting Citations:
Claim 4: 4-CQA restores fast-twitch (Type II) muscle fibers and functions as a metabolic exercise mimetic.
-
Evidence Level: Level D (Pre-clinical). [FLAG: Translational Gap]
-
Verification & Analysis: The assertion that a botanical extract can prevent the age-related transition from fast-twitch to slow-twitch fibers is lacking clinical validation. Fast-twitch fiber preservation in humans practically requires mechanical loading (resistance training) to stimulate the necessary motor unit recruitment. Claiming 4-CQA acts as a pharmacological exercise mimetic in humans based strictly on rodent treadmill endurance and grip strength data overstates the current scientific consensus.
-
Supporting Citations:
Part 4: Actionable Intelligence (Deep Retrieval & Validation Mode)
The Translational Protocol (Rigorous Extrapolation)
-
Human Equivalent Dose (HED):
-
Calculation Parameters: The study utilized a diet containing 0.1% to 0.2% Peucedanum japonicum (PJ) extract. A standard 30-gram aged mouse consumes approximately 4 grams of food daily.
-
Animal Dose: 4 grams of food at a 0.1% concentration yields 4 mg of PJ extract per day. 4 mg / 0.03 kg body weight = ~133 mg/kg/day.
-
HED Math: AnimalDose (133 mg/kg) × (Mouse Km 3 / Human Km 37) = 10.8 mg/kg.
-
Translational Dose: For a standard 70 kg human, the theoretical effective dose ranges from 756 mg/day(based on the 0.1% diet) to 1,512 mg/day (based on the 0.2% diet).
-
Pharmacokinetics (PK/PD):
-
Bioavailability: Poor to moderate. Clinical data on chlorogenic acids shows that only about 33% of ingested caffeoylquinic acids are absorbed intact in the human small intestine. The compound undergoes extensive hepatic first-pass metabolism.
-
Half-life: Rapid clearance. Human oral administration of chlorogenic acids yields a Tmax (time to peak concentration) of 1 to 2 hours, with a very short half-life (t1/2) of 1 to 2 hours.
-
Safety & Toxicity:
-
Safety Data Absent. Precise NOAEL (No-Observed-Adverse-Effect-Level), LD50, and Phase I human safety profiles for high-dose, standardized Peucedanum japonicum extract are currently unavailable in the literature. While generic dietary chlorogenic acids are Generally Recognized As Safe (GRAS), concentrated botanical extracts require independent hepatotoxicity screening.
Biomarker Verification To verify target engagement in a human protocol, standard plasma metabolic panels are insufficient. Required verification includes:
-
Primary: Skeletal muscle biopsy utilizing RT-qPCR to measure PGC-1alpha, Nrf1, and TFAM mRNA upregulation.
-
Secondary: Immunohistochemistry of muscle tissue to quantify the ratio of Myosin Heavy Chain (MHC) Type II to Type I fibers.
-
Tertiary: Metabolomic screening for systemic decreases in ubiquitin-proteasome system (UPS) activity, specifically tracking Atrogin-1 and MuRF1 expression.
Feasibility & ROI
-
Sourcing: Highly feasible but variable. Peucedanum japonicum (often called “longevity herb” or “chomeiso” in Japan) is commercially available as a health powder or tea. However, isolating and standardizing the precise 4-CQA metabolite requires pharmaceutical-grade synthesis or high-end nutraceutical extraction.
-
Cost vs. Effect: A high-quality green coffee bean extract (the most practical proxy for chlorogenic and caffeoylquinic acids) costs approximately $15–$30 per month. The ROI is likely moderate for metabolic health but low for targeted sarcopenia reversal until liposomal or nanoparticle delivery systems are developed to bypass first-pass metabolism and drive the compound into skeletal muscle tissue.
Part 5: The Strategic FAQ
1. How does oral 4-CQA survive hepatic first-pass metabolism to reach therapeutic concentrations in human skeletal muscle tissue? It likely does not, which is the primary translational bottleneck. Data indicates that chlorogenic acids are extensively metabolized by the gut microbiome and the liver. Achieving the intramuscular concentrations seen in in vitro or murine models will practically require advanced delivery systems, such as phospholipid encapsulation, to prevent premature degradation.
2. Does 4-CQA supplementation blunt the adaptive response to actual resistance training, similar to high-dose Vitamin C or E? This is a critical, unresolved risk. 4-CQA is a potent antioxidant. Exercise-induced Reactive Oxygen Species (ROS) are the primary signaling molecules that trigger natural mitochondrial biogenesis and muscle hypertrophy. Flooding the system with exogenous antioxidants immediately post-exercise can neutralize this signal, potentially blunting the benefits of actual mechanical lifting.
3. Are the anti-catabolic effects (Atrogin-1 suppression) dependent on AMPK activation, and does this conflict with mTOR-driven muscle hypertrophy? Yes. 4-CQA drives mitochondrial biogenesis largely through AMPK activation. AMPK is a strict, direct inhibitor of mTORC1 (the primary driver of muscle protein synthesis). Therefore, while 4-CQA may prevent muscle loss (catabolism), its concurrent suppression of mTOR means it is highly unlikely to stimulate muscle growth (hypertrophy) on its own.
4. How does the 0.2% dietary inclusion rate translate to a human equivalent dose, and is that volume tolerable? The 0.2% murine diet translates to roughly 1.5 grams of extract daily for a 70 kg human. While 1.5 grams of a botanical extract is easily encapsulated and tolerable for the human GI tract, the concentration of the specific active metabolite (4-CQA) within that 1.5g extract must be standardized to match the murine exposure.
5. Does the forced mitochondrial biogenesis increase reactive oxygen species (ROS) leakage if the electron transport chain is not mechanically overloaded by physical exercise? Pre-clinical data suggests 4-CQA upregulates endogenous antioxidant enzymes (like SOD and Catalase) alongside biogenesis. However, forcing the construction of new mitochondrial factories without an accompanying demand for ATP (via exercise) risks reductive stress and minor ROS leakage in a sedentary human model.
6. Is there a paradoxical uncoupling effect in skeletal muscle mitochondria with chronic 4-CQA exposure? There is no current evidence that 4-CQA acts as a chemical uncoupler (like DNP). It increases the structural density of the respiratory chain complexes (Complexes I-V) rather than making the inner mitochondrial membrane artificially permeable to protons.
7. Why wasn’t maximum lifespan evaluated if the compound effectively reverses age-related frailty? The study was designed strictly as an 8-week functional healthspan intervention in late-life mice. Validated lifespan studies require tracking subjects until natural death, which demands significantly more funding, cage time, and distinct statistical powering (e.g., Kaplan-Meier survival analysis).
8. Does 4-CQA preferentially construct Type II (fast-twitch) muscle fibers, or is the fiber-type rescue a secondary effect of generalized mitochondrial health? It is a secondary rescue effect. Aging muscle naturally undergoes a denervation-reinnervation process that defaults to slow-twitch (Type I) characteristics. By suppressing the UPS-mediated degradation pathways that aggressively target Type II fibers during disuse, 4-CQA preserves the existing fast-twitch architecture rather than actively manufacturing new fast-twitch motor units.
9. How do the pharmacokinetics of isolated 4-CQA compare to the full-spectrum Peucedanum japonicum extract? Full-spectrum extracts contain hundreds of secondary metabolites, including coumarins, which often act as natural enzymatic inhibitors. These co-compounds can slow the hepatic clearance of 4-CQA, theoretically giving the full extract a superior pharmacokinetic profile and longer biological half-life compared to the administration of isolated, synthesized 4-CQA.
10. What specific downstream protein changes verify target engagement in clinical applications? The definitive markers are an increase in the nuclear translocation of Nrf1 and the subsequent upregulation of Mitochondrial Transcription Factor A (TFAM) within the muscle tissue, leading to an objectively measurable increase in mtDNA copy number.
Interaction Check: The Longevity Stack
-
Metformin: Synergistic. External data demonstrates that chlorogenic acids synergistically enhance metformin’s activation of AMPK and its anti-lipogenic effects in hepatic tissue. Combination improves anti-lipogenic activity (2022).
-
Rapamycin: Antagonistic for Muscle. Rapamycin strictly inhibits mTOR. Combining rapamycin with 4-CQA (an AMPK activator) will create a profound, dual-axis suppression of anabolic muscle signaling. This combination risks exacerbating sarcopenia if not heavily offset by resistance training and amino acid availability.
-
SGLT2 Inhibitors: Neutral / Additive. Both agents improve peripheral glucose disposal and metabolic flexibility through separate pathways (renal glucose excretion vs. muscular mitochondrial sink). No direct negative pharmacokinetic interactions are identified.
-
Acarbose: Neutral. Acarbose delays intestinal carbohydrate absorption. Since 4-CQA is primarily absorbed in the upper small intestine, acarbose is unlikely to interfere with its uptake, though GI transit times may be slightly altered.
-
17-alpha Estradiol: Unknown. There is no data evaluating the interaction between non-feminizing estrogens and botanical PGC-1alpha activators.
-
PDE5 Inhibitors: Potentially Synergistic. PDE5 inhibitors increase endothelial nitric oxide and skeletal muscle perfusion. This increased blood flow could theoretically act as a delivery vector, enhancing the muscular penetrance of 4-CQA during its short biological half-life.