Rapamycin, a compound originally discovered in the soil of Easter Island, has transitioned from a niche immunosuppressant to the most reproducible lifespan extension drug in scientific history. The “Big Idea” explored by Dudley Lamming in GeroScience is that we are now at a critical juncture: moving from successful animal studies to human clinical applications.
For almost a century ago, calorie restriction (CR) has been identified as a robust intervention for extending lifespan and healthspan, a discovery that captured the imagination of both scientists and the public. If the powerful mechanisms engaged by CR can be uncovered and harnessed through a pill, humans might be able to live longer and healthier lives. In fact, studies over the past two decades have now shown that rapamycin extends lifespan and probably extends healthspan in animals. In this new paper Dudley Lamming concludes that rapamycin — or molecules that similarly act to inhibit mTOR — may yet realize the century-old dream of extending human healthspan and lifespan with a small molecule.
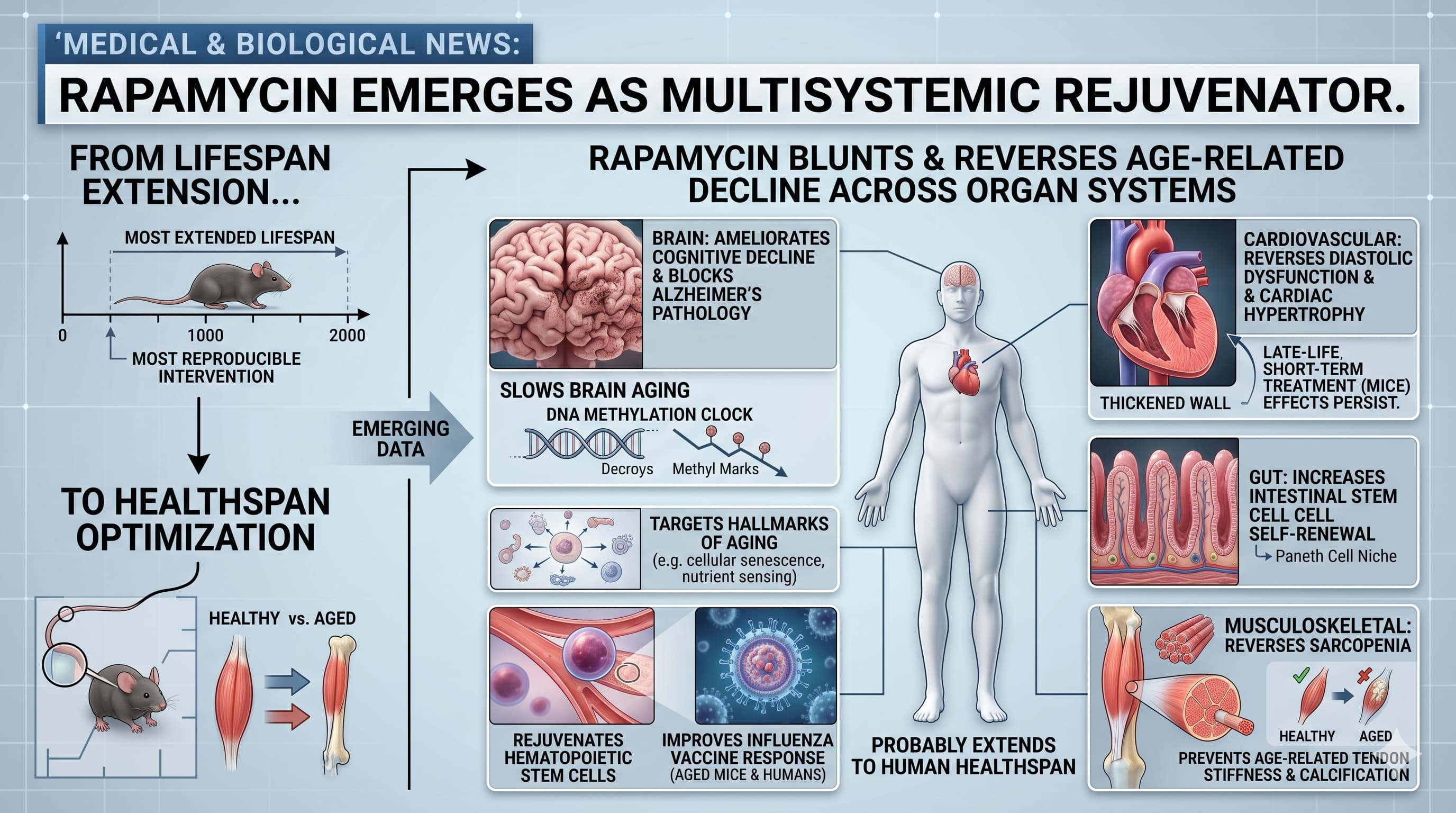
Evidence that Lamming cites includes emerging data that suggests how rapamycin may function as a multisystemic rejuvenator, blunting or reversing age-related declines across nearly every major organ system.
The evidence that rapamycin “probably” extends human healthspan is grounded in extensive rodent and preliminary mammalian studies. In the brain , rapamycin has been shown to block Alzheimer’s-type pathology, ameliorate cognitive decline, and slow brain aging as measured by DNA methylation clocks. The cardiovascular benefits are equally striking: late-life, short-term treatment in mice has reversed diastolic dysfunction and cardiac hypertrophy, with improvements persisting for months after the drug was stopped.
Beyond the vital organs, the drug targets the “hallmarks of aging” at a cellular level. It rejuvenates hematopoietic stem cells and improves the response to influenza vaccination in both aged mice and humans. In the gut , it increases the self-renewal capacity of intestinal stem cells via the Paneth cell niche. Even the musculoskeletal system sees benefits; rapamycin has reversed sarcopenia in rats and prevented age-related tendon stiffness and calcification in mice.
The primary challenge identified is the “mTOR Dualism.” Rapamycin acts on two complexes: mTORC1 and mTORC2. Inhibiting mTORC1 is largely beneficial, promoting autophagy (cellular cleanup) and extending lifespan. However, chronic or high-dose administration also inhibits mTORC2, which can lead to metabolic dysfunction, including insulin resistance and hyperlipidemia.
The geroscience community is currently focused on “intermittent dosing” to exploit this gap—inhibiting the “bad” aging pathways while sparing the “good” metabolic ones. While preliminary results in dogs and humans suggest safety and potential benefits in cardiac and immune function, the field still lacks large-scale, long-term data to prove that rapamycin actually extends human healthspan. Thankfully, new well-funded studies have now been initiated to address this key issue, and much more detailed information will be available over the coming years.
Actionable Insights
The practical applications of this research focus on optimizing the benefit-to-risk ratio through targeted and timed interventions. One of the most promising “take-home” messages is the efficacy of topical administration. Low-dose rapamycin cream has been shown to improve skin appearance, reduce the senescence marker p16, and increase collagen VII without reaching detectable levels in the blood, thereby avoiding systemic side effects.
For systemic longevity, the shift toward intermittent dosing (e.g., weekly) aims to capture the benefits of mTORC1 inhibition—such as enhanced autophagy and stem cell function—while sparing mTORC2, which is essential for insulin sensitivity and metabolic health. Preliminary human data from the PEARL trial suggests that such regimens are generally well-tolerated and may even increase lean mass in women.
However, practitioners must exercise caution regarding musculoskeletal interactions. While rapamycin may prevent tendon aging, its effect on osteoarthritis is mixed, with some evidence suggesting it could aggravate joint pathology through feedback activation of Akt signaling. Additionally, because it may blunt certain exercise-induced muscular adaptations, the timing of rapamycin in relation to physical training requires careful consideration to avoid diminishing performance gains.
Source:
- Open Access Paper: Bench to bedside: is rapamycin headed for the docTOR?
- Institution: Department of Medicine, University of Wisconsin-Madison.
- Country: USA.
- Journal Name: GeroScience.
- Impact Evaluation The impact score of this journal is 5.6 (CiteScore/Journal Impact Factor), therefore, this is a High impact journal within the specialized fields of geroscience and the biology of aging.