Yes:
“The subjects in the astaxanthin group were instructed to take one 20 mg astaxanthin capsule (Marine Product Tech. Inc., Seongnam, South Korea) once daily after breakfast for 12 weeks.”
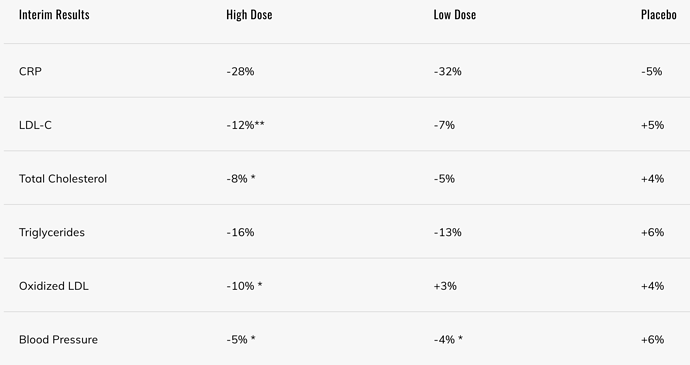
Effect of Astaxanthin on Blood Lipid Profiles
Total cholesterol, triglycerides, HDL cholesterol, LDL cholesterol, ApoA1, ApoB, and ApoA1/ApoB ratio between placebo and astaxanthin groups at both baseline and at 12 weeks are listed in Table 2. The lipid concentrations were not significantly different between the two groups except LDL cholesterol, ApoB, and ApoA1/ApoB ratio. In the astaxanthin group, the LDL cholesterol, ApoB, and ApoA1/ ApoB ratio at 12 weeks were significantly lower (by 10.4, 7.59, and 8.22%, respectively) than at baseline.
Patient Compliance and Adverse Events
All subjects in the two intervention groups completed the study. According to the count of remaining capsules, adherence rates were 93.4 and 92.9% in the astaxanthin
In the astaxanthin group, gastrointestinal adverse events were observed: fecal color changed to red (n=2, 14.3%) and bowel movements increased (n=2, 14.3%). Changes in fecal color to red could have been due to the reddish color of astaxanthin. In the placebo group, muscle fatigue (n=2, 15.4%) and abdominal discomfort (n=1, 7.69%) were observed. Subjective symptom improvement after astaxanthin intervention included decreased fatigue (n=4, 28.6%), body weight loss (n=3, 21.4%), and improved skin condition (n=2, 14.3%). There were no abnormal changes in the results of renal and hepatic function tests during the study period.
Discussion:
…
One particularly interesting finding is the possibility that astaxanthin prevents body weight gain. The changes in BMI and body weight in the two intervention groups support this hypothesis. Body weight and BMI hardly changed in the astaxanthin group, whereas they both increased in the placebo group (Table 1). A previous animal study also showed that astaxanthin inhibited increases in body and adipose tissue weights in obese mice fed a high-fat diet [11]. These findings may be meaningful for overweight or obese individuals. However, there was not a significant difference in our study; thus, more studies are needed to evaluate whether astaxanthin truly has a body weight-lowering effect.
Protective effects of astaxanthin, including antioxidant activity, have been demonstrated in many studies. However, information about the biological mechanism of astaxanthin is still not sufficient. We can speculate based on previous clinical results and pharmacokinetic data, but further studies are required to determine the mechanism of astaxanthin in humans.
In summary, 12 weeks of supplementation with astaxanthin significantly lowered LDL cholesterol and ApoB in overweight subjects. Also, OS biomarkers including MDA, ISP, SOD, and TAC were improved by astaxanthin. Thus, astaxanthin supplementation is apparently safe and effective for prevention of lipidemic diseases and oxidative damage in healthy overweight subjects.