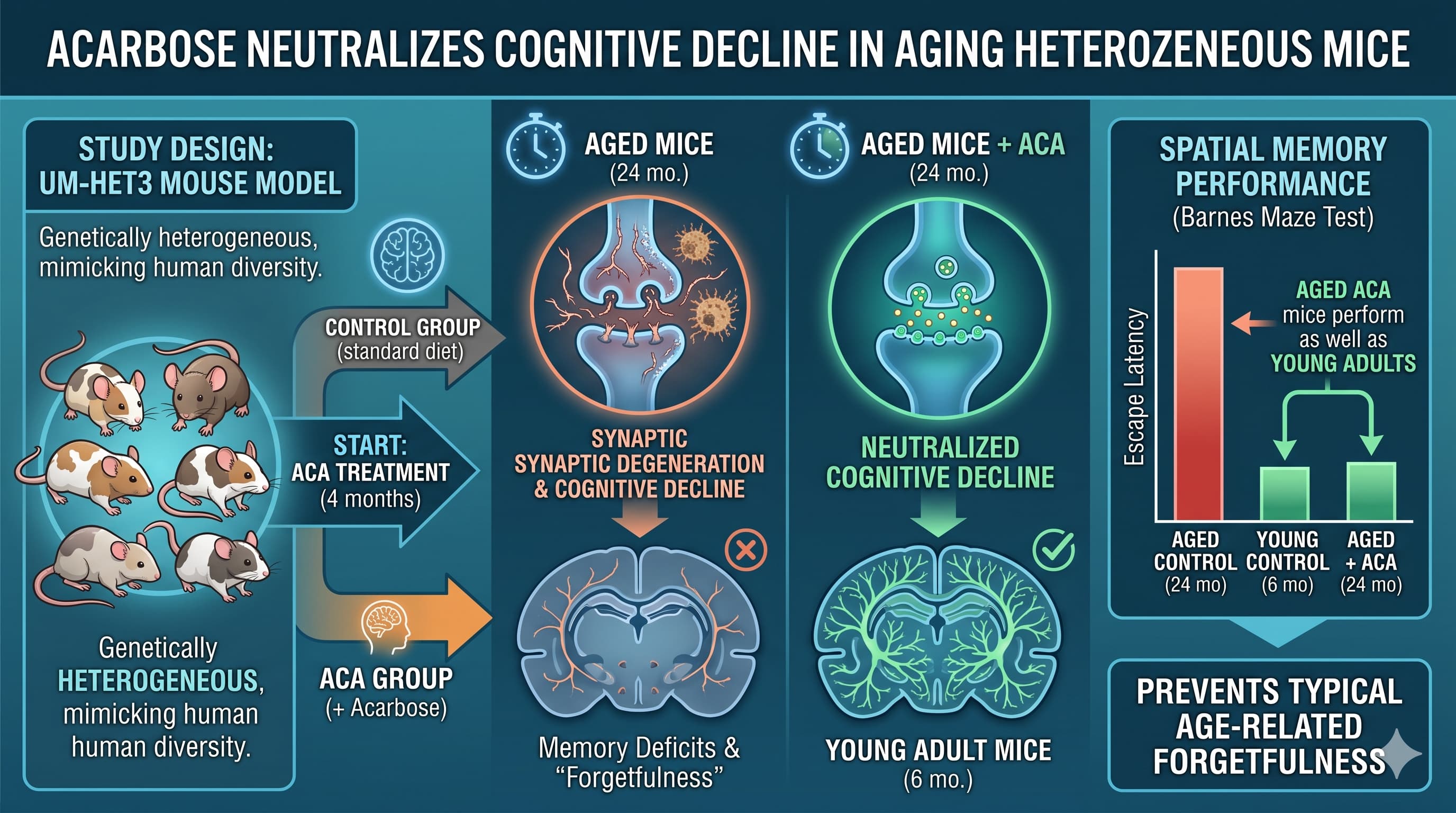
A new study from the University of Michigan suggests that the “geroscience hypothesis”—the idea that slowing the aging process can simultaneously treat all age-related diseases—may have a significant blind spot. Researchers found that Acarbose (ACA) , a common Type 2 diabetes medication and known lifespan-extender, successfully “neutralized” cognitive decline in normally aging mice but offered zero protection against Alzheimer’s Disease (AD).
The research utilized the UM-HET3 genetically heterogeneous mouse model, which better mimics human genetic diversity than standard inbred lab mice. In healthy mice, those treated with Acarbose starting at four months of age performed as well on spatial memory tests at 24 months (roughly equivalent to a 70-year-old human) as young six-month-old adults. Essentially, the drug prevented the typical “forgetfulness” associated with getting older.
However, the results took a sharp turn when applied to an Alzheimer’s-relevant model (5xFAD-HET3 ). Despite the drug’s ability to blunt glucose spikes and mimic the benefits of caloric restriction, it failed to ameliorate any cognitive deficits in the AD group. In the oldest AD mice, Acarbose may have even slightly delayed the ability to form new memories.
This study reinforces a sobering reality for the longevity community: Alzheimer’s is likely not just “accelerated aging”. While Acarbose targets metabolic pathways and peripheral glucose, AD is driven by unique “pathological protein aggregates” like beta-amyloid and tau, alongside intense neuroinflammation that seems immune to simple metabolic intervention. For those seeking to extend “brainspan,” Acarbose remains a potent candidate for maintaining healthy function, but it is not a cure-all for neurodegeneration.
Actionable Insights
-
Metabolic Priming for Brain Health: Acarbose (ACA) acts by inhibiting alpha-glucosidase, slowing carbohydrate breakdown and blunting post-prandial glucose spikes. For individuals without neurodegenerative disease, maintaining stable glucose levels appears to be a primary lever for preserving spatial memory into late life.
-
Acarbose vs. Alzheimer’s: Do not rely on ACA or basic caloric-restriction mimetics as a primary preventative for Alzheimer’s. The pathology of AD (amyloid/tau) operates through distinct mechanistic cascades that glucose regulation alone does not address.
-
Dietary Context Matters: While ACA failed in standard AD models, previous data suggests it may help if a “Western Diet” (high fat/sugar) is the primary driver of metabolic-related cognitive impairment.
-
Sex-Specific Efficacy: Historically, ACA extends lifespan more robustly in males than females. However, due to high male aggression in this study, the cognitive data is female-biased. Men may potentially see even greater cognitive benefits than those reported here.
Source:
-
Open Access Paper: Acarbose improves cognitive function in a mouse model of normal aging but not Alzheimer’s disease
-
Institution: University of Michigan Medical School, Ann Arbor, MI, USA.
-
Journal: bioRxiv (Preprint), Posted May 01, 2026.
-
Impact Evaluation: As a preprint, this paper has not yet received a formal Journal Impact Factor (JIF). However, bioRxiv is the premier repository for biological sciences, and the authors are affiliated with the NIA-funded Interventions Testing Program (ITP), an elite group in longevity science therefore this is a High-potential, pre-peer review manuscript.
Related Reading:
3 Likes
Mechanistic Deep Dive
-
Glucose and Insulin: ACA mimics caloric restriction (CR) by blunting postprandial glucose spikes. This likely preserves hippocampal function by reducing glycemic variability, which is linked to “age-related glucose dysregulation”.
-
mTOR/AMPK: While not directly measured in this paper, ACA’s CR-mimetic properties typically act through reduced mTOR signaling and increased AMPK activation.
-
Divergent Pathways: The study highlights that “normal” cognitive aging is characterized by synaptic weakening , whereas AD is characterized by overt cell death and neuroinflammation (microglial activation). ACA appears to rescue the former but has no impact on the latter.
Novelty
This is one of the first studies to demonstrate that an intervention can neutralize age-related spatial memory loss in very old (24 mo) heterogeneous mice, effectively keeping their performance “young”. It also provides a critical “reality check” for the geroscience hypothesis by showing a clear failure of an anti-aging drug in a pathological disease context (AD).
Critical Limitations
-
Sex Representation: The loss of males due to “inter-male aggression” resulted in a female-heavy study. Since ACA is known to be more effective in males, the reported benefits in normal aging may actually be an underestimate.
-
Limited Cognitive Domains: The study focused exclusively on spatial learning and memory via the Morris Water Maze. It did not assess executive function, attention, or working memory, which may respond differently to ACA.
-
Cross-Sectional vs. Longitudinal: Mice were not tracked over their lives; separate cohorts were tested at different ages. This can introduce cohort-specific variability that longitudinal studies avoid.
-
AD Model Specificity: The 5xFAD model is an aggressive, early-onset model. It is possible that ACA might have different effects on slower-progressing, “late-onset” models of AD.
Actionable Intelligence
The Translational Protocol (Rigorous Extrapolation)
-
Human Equivalent Dose (HED): * Math: In this study, mice received 1000 ppm Acarbose (ACA) in chow. A 30g mouse eating 4g of food per day consumes 4 mg of ACA, equating to an animal dose of ~133.3 mg/kg.
-
Formula: HED=AnimalDose(mg/kg)×(Kmanimal/Kmhuman). Using FDA constants (Kmmouse=3, Kmhuman=37), the calculation is 133.3×(3/37)≈10.8mg/kg.
-
Result: For a 70 kg human, this translates to ~756 mg/day. This is significantly higher than the standard clinical dose for type 2 diabetes (typical maximum of 300 mg/day, administered as 100 mg TID).
-
Pharmacokinetics (PK/PD): * Bioavailability: Systemic absorption is extremely low (<2%), as ACA acts locally within the brush border of the small intestine.
-
Half-life: The elimination half-life of the absorbed fraction is approximately 2 hours.
-
Safety & Toxicity:
-
LD50: Oral LD50 in mice and rats is estimated at >10 g/kg, indicating a very high margin of safety.
-
NOAEL: Daily doses up to 450 mg/kg in rats were tolerated without drug-related toxicity in sub-chronic studies.
-
Adverse Signals: Rare instances of elevated transaminases have been reported; monitoring liver enzymes is recommended. It is contraindicated in patients with inflammatory bowel disease, intestinal obstruction, or severe renal impairment (serum creatinine >2.0 mg/dL).
Biomarker Verification
-
Primary Target: Reduction in postprandial glucose spikes (the “blunting” effect).
-
Engagement Markers: Elevation in circulating GLP-1 (Glucagon-Like Peptide-1) concentrations following a mixed-meal test.
-
Long-term Metric: Modest reductions in HbA1c (typically 0.5–0.8%).
Feasibility & ROI
-
Sourcing: ACA is a Prescription Only (Rx) medication (marketed as Precose or generic). It is not currently available as a verified over-the-counter supplement.
-
Cost vs. Effect: * Cost: Generic Acarbose (25–100 mg) typically costs between $50 and $75 per month at US pharmacies without insurance.
-
ROI: High for “normal” cognitive aging; the data suggests it may effectively “neutralize” age-related memory decline in healthy individuals. ROI is Zero for established Alzheimer’s Disease unless a high-sugar/fat diet is a primary driver.
Part 5: The Strategic FAQ
- Why did the 5xFAD mice fail to respond despite being on the same HET3 genetic background as the responders?
- AD pathology is not merely “accelerated aging” but involves distinct drivers like amyloid-beta plaques and neuroinflammation that are seemingly decoupled from the peripheral metabolic pathways ACA targets.
- Does the aggression-led attrition of males mean we are underestimating ACA’s benefits?
- Likely yes. Since ACA has historically shown a much greater lifespan effect in males, the female-biased cohort in this study may present a “floor” version of the drug’s true potential for cognitive longevity.
- Does ACA’s low bioavailability limit its effect on the brain?
- ACA acts primarily in the gut; its cognitive benefits are likely mediated indirectly through improved glucose homeostatis and upregulated GLP-1, rather than direct blood-brain barrier crossing.
- Is the “Western Diet” a necessary condition for ACA to work in Alzheimer’s?
- Evidence suggests ACA does reverse cognitive deficits in AD models (like 3xTg) when they are exacerbated by a high-sugar “Western” diet, implying its neuroprotective role in disease may be strictly metabolic.
- How does ACA compare to Metformin for longevity?
- ITP trials show ACA has a more consistent and robust lifespan-extending effect than Metformin in mice, particularly in males.
- Does the cross-sectional design mask survival bias?
- While possible, the authors balanced cohorts. However, the 24-month group inherently represents “survivors,” which could slightly skew results toward a healthier baseline than the 6-month group.
- What is the “floor effect” mentioned regarding weight in the aged 5xFAD group?
- AD-related pathology caused weight loss in both control and ACA mice, which likely obscured the typical weight-lowering effect of the drug.
- Will lower clinical doses (300 mg/day) still provide cognitive benefits?
- This remains a knowledge gap; the HED used in the study (750+ mg/day) exceeds standard human dosing, and it is unknown if benefits scale linearly.
- What is the mechanism for the improved search strategy in normal aging?
- ACA appears to preserve synaptic integrity and prevent the age-related “weakening” of hippocampal synapses, whereas it cannot stop the cell death typical of AD.
2 Likes
N=1; I’m taking 600mg a day (2x3x100mg) so 3 100mg pills with each of my two larger meals per day. I have taken as much as 900mg in a day, but I figure that’s kind of overkill. I’ve been doing this most of the past year with no obvious ill effects and no notable or startling benefits. But various studies have convinced me (for the time being) that it is probably beneficial, and it’s cheap.
Of course there is the gas to contend with, but mine has decreased quite a bit and is very tolerable. I’ve cut down my raw potato starch to around 30grs a day, I think this helped moderate my gas production. I’ve been concerned with my gut biome for some time and the supra potato starch did max out my butyrate producers.
Now I’ve switched my focus to probiotics/fermented foods and I’m also making my own yogurt; L. casei (Yukult); L. reuteri and L. plantarium (each in different batches) and I’ll drink/consume about a cup per day, alternating throughout the week.
Of course everybody is different and I do not generally recommend acarbose, but this is just my story.
Oh the yogurt I can endorse. I’ve gone with ultra pasturized milk/cream and I skip the preheat to 180, saves alot of time in the preparation. The monthly cost of my yogurt production is approaching the cost of acarbose ($150 vs. $200 for acarbose)
Addedum: I must add a caveat here, if one was bedridden, the “unneccesary” acarbose and resulting flatulence, might well be taken as intentionally offensive to the caregiver, and it doesn’t take too many offended angry caregivers to ruin your day.
1 Like
Metabolic Sentinel: Acarbose Repurposed to Shield Vision by Resetting the Microglial “Immune Clock”
Retinal ischemia-reperfusion (IR) injury—the rapid starvation and subsequent flooding of oxygen in eye tissue—is a catastrophic event common to glaucoma and diabetic retinopathy. This process triggers a self-destructive spiral where microglia, the resident immune cells of the eye, abandon their protective roles to become pro-inflammatory “toxic” agents, ultimately killing the retinal ganglion cells (RGCs) essential for sight.
Recent research published in the Journal of Neuroinflammation reveals that acarbose (ACA), a common Type 2 diabetes medication, possesses a “dual-key” ability to halt this neurodegeneration. While traditionally used to block carbohydrate absorption in the gut, ACA was found to act directly within the retina when administered via intravitreal injection. The drug effectively “reprograms” the metabolism of overactive microglia, shifting them from a fuel-hungry, inflammatory state back toward a restorative, homeostatic profile.
The study identified a critical metabolic checkpoint: the enzyme Pkm2. Under ischemic stress, Pkm2 becomes “tagged” through acetylation at a specific site (K270), which drives inflammatory signaling and drains cellular levels of nicotinamide adenine dinucleotide (NAD)—a molecule vital for mitochondrial health and longevity. ACA interventions restored NAD levels and mitochondrial integrity by activating Sirt1 , a well-known longevity protein, which “cleans” the Pkm2 enzyme through deacetylation. This metabolic reset significantly improved RGC survival and preserved retinal thickness in mouse models. By treating neuroinflammation as a metabolic malfunction rather than just an immune overreaction, this research opens a new frontier for preserving sensory health in aging populations.
Actionable Insights
For the longevity-focused individual, this study provides three critical takeaways regarding metabolic repurposing and neuroprotection. First, it reinforces Acarbose as a top-tier candidate for “geroprotection” beyond glucose control, specifically highlighting its ability to preserve NAD pools and mitochondrial function in the face of acute stress. Second, the identification of the Sirt1-Pkm2-NAD axis suggests that interventions targeting Pkm2 acetylation (such as the experimental compound HY-113082 ) may eventually offer a synergistic approach with NAD precursors (NMN/NR) to dampen age-related neuroinflammation. Finally, the study highlights that microglial health is inextricably linked to metabolic checkpoints; maintaining systemic metabolic flexibility may be as crucial for vision as it is for metabolic health. While the study utilized direct eye injections, it provides a mechanistic rationale for how systemic metabolic drugs can influence highly specialized tissues like the retina.
Source: