I was wondering whether to add this to his trial topic or put it up as a youtube video. I thought I would get the transcript etc and then put it up as a trial. If moderators then want to merge it with the other topics then at least you have the transcript etc.
As people know I am not in any way surprised by the idea that rapamycin inhibits anabolism. We know anecdotally that this is the case and spending $724,637 is an expensive way of confirming what is already known. (I don’t know if that is USD or New Zealand)
chatGPT:
Below is a cleaned-up transcript based on the text you provided, followed by a summary and critique.
Tidy transcript
Speaker 1 (host/interviewer):
We spent 5 years and $724,637 of crowdfunded money to test whether rapamycin would improve muscle performance when combined with exercise. Our results have just been published in the Journal of Cachexia, Sarcopenia and Muscle. They really surprised me when we first calculated them. Here to discuss the results is one of the co-authors, Professor Matt Kaeberlein.
Matt, you know the preclinical rapamycin research better than almost anyone. Why are you excited about rapamycin’s potential role for human health, and why did you want to be involved in this study when I first contacted you all those years ago?
Matt Kaeberlein:
First, thanks for having me on again to discuss the results. I’ve been studying the biology of aging for a couple of decades now, and I’ve worked on rapamycin in particular over that time. Work from my lab and many others had already placed rapamycin, five years ago, as the gold standard drug that seemed to slow aging, increase lifespan, and broadly improve healthspan in laboratory animals, from yeast and worms up to rodents.
From that perspective, it seemed very reasonable to ask whether rapamycin could affect healthspan metrics in people. At that time I had also started a series of clinical trials in companion dogs, living with their owners in the real world. The results were preliminary, but directionally positive for things like heart function, activity, and quality of life. So we had some indication that rapamycin could be tested safely in a clinical trial setting, albeit a veterinary one, and that certain healthspan metrics might improve in aged dogs living in the real world.
For that reason, I still believe rapamycin is our best single-drug shot on goal if we want to target biological aging in humans. So of course it made sense to help get this clinical trial off the ground.
Speaker 1:
Some people have asked why we didn’t do a lifespan study. In humans, that would take decades. One reason we selected muscle is that if we could improve muscle function, that would likely improve frailty-related markers. Also, when I was looking through the rapamycin literature, one thing that informed the design was the idea that aged human muscle may overactivate mTOR complex 1. Rapamycin inhibits mTOR.
If mTOR complex 1 is overactivated, autophagy may be impaired. In theory, aged muscle may hold on to old, damaged components rather than clearing them away. The hope was that if we cycled rapamycin—periods of taking it to suppress mTOR and allow autophagy, followed by exercise to turn mTOR back on and rebuild—then we might get the best of both worlds. That was the basic cycling hypothesis, and it was one reason to do a muscle study rather than a lifespan study.
I also didn’t initially realize how expensive these trials would be. A lot of the five-year journey was figuring out how to fund it. Before we go into the results, is there anything else you want to say about the study design?
Matt Kaeberlein:
Two things. First, I agree there is evidence for mTOR hyperactivation, or at least suboptimal mTOR regulation, in aging muscle. One consequence is impaired autophagy, but there are others too, including mistranslation of proteins and inefficient mitochondrial function. So I’d be careful not to imply that autophagy is the only reason rapamycin might help.
Second, there is preclinical evidence supporting muscle function as an endpoint in older people. In rodents—better in rats than mice, though even in mice there are data—rapamycin can protect against age-related muscle degeneration, sarcopenia, and deterioration in body composition. So there really was preclinical rationale here.
That matters because people often assume mTOR inhibition must always be bad for muscle, since mTOR activation is needed to build new muscle. But this was not a crazy idea. There were good reasons to think it was a logical endpoint in a human clinical trial.
Speaker 1:
In designing the study, we wanted the protocol to resemble what people could actually do in real life. We enrolled 40 participants. Half took sirolimus/rapamycin at 6 mg, and half took placebo. Both groups exercised.
We delivered exercise bikes to their homes because these were older adults aged 65 to 85. We wanted a safe home-based exercise program, not something risky like high-intensity intervals that could cause injury. The study ran for 12 weeks.
Participants exercised on day 1, day 3, and day 5, then took rapamycin or placebo on day 6. The idea was that if they took rapamycin on Saturday, most of it would be out of their system by the time they exercised again on Monday.
Another thing I wanted to make sure of was that all endpoints were preregistered. The study was on a clinical trial database so people could see exactly what we planned to do and measure. There was no p-hacking or number-fudging. The endpoints and sensitivity analyses were prespecified.
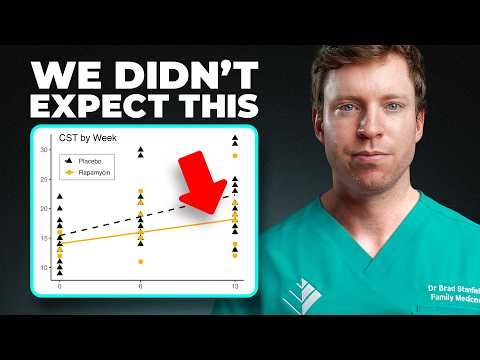
The primary outcome was the 30-second chair stand test—how many times you can stand up and sit down in 30 seconds. We chose it because it is relatively sensitive for overall muscle power and strength in older adults.
In the intention-to-treat analysis, which includes everyone in the study, the primary endpoint did not reach statistical significance. But it trended toward the placebo group improving more than the rapamycin group, which was interesting. It was not statistically significant, so we should not overinterpret it.
In the prespecified sensitivity analyses, when we looked only at people who completed the study (“case complete”), we saw statistically significant results showing the placebo group improved more than the rapamycin group. Then in the per-protocol analysis—people who took at least 75% of the study drug and completed at least 75% of the exercise plan—the difference widened further. The more people adhered to the protocol, the more the two arms diverged.
Those were not the results we predicted going in. Matt, what were your initial impressions when we calculated these together?
Matt Kaeberlein:
A couple of things. First, I want to emphasize that the trial was done to very high standards in terms of rigor. People can criticize the sample size, the duration, and so on, but in terms of design and trying to be unbiased, I think you did everything that could reasonably be asked.
Second, it is important that the primary endpoint did not hit statistical significance in the most rigorous analysis. Even though the sensitivity analyses were prespecified, they are less robust because they break randomization. That does not mean p-hacking occurred, but it does mean those analyses are more susceptible to false positives.
So from a pure statistical point of view, I have less confidence that there is a real difference between rapamycin and placebo, because the statistically significant difference only appears in the sensitivity analyses.
That said, both groups improved. My own interpretation is that the placebo group improved more than the rapamycin group—in other words, rapamycin attenuated the improvement in chair stand performance. Both groups improved directionally, but I think rapamycin blunted the gains under these conditions.
This also shows how noisy human data are. Some rapamycin users improved as much as placebo users, and some placebo participants worsened. That is why we need well-powered clinical trials, especially in generally healthy people rather than patients with a specific disease.
Speaker 1:
I’d mostly agree. We should not overinterpret the findings. The secondary muscle-performance measures—the 6-minute walk test and hand grip strength—also did not reach statistical significance, but they still trended in the same direction, with placebo improving more than rapamycin.
That starts to paint a picture that, over a 12-week period with once-weekly dosing in this population, the dosing protocol may not have been optimal. It would be interesting if we had stopped the study drug at 12 weeks but continued exercise and tested again at 24 weeks. The curves might have reversed. So perhaps the dosing schedule needed a longer washout period.
Matt Kaeberlein:
That is a reasonable hypothesis. Another point is that seeing the same direction of effect across multiple endpoints, even if not statistically significant, does not increase statistical significance, but it can strengthen one’s practical belief that there may be a real signal here.
What surprises me is that this is one of the first human studies where rapamycin seems to show a fairly clear signal of underperforming placebo. The human data overall are limited, especially for muscle function. The PEARL trial, despite interpretive challenges, suggested positive changes in body composition in women over a longer period. That trial used a compounded formulation without enteric coating, so the effective dose may have been only around 1 to 1.5 mg per week—lower than in this study.
When designing a clinical trial, you make the best guesses you can about dose, frequency, duration, and population. Then you get the results and think maybe you should have done it differently. That is just how clinical trials work.
Speaker 1:
We also measured SF-36 health-related quality-of-life scores and found no statistically significant differences. We looked at CRP, a marker of inflammation. You might expect rapamycin to reduce inflammation, but here the treatment group seemed to show the opposite. However, that looked like noise driven by two outliers with markedly elevated CRP, and CRP can rise for lots of mundane reasons, like infection or injury. So I do not think people should overinterpret that.
On safety, there were more adverse events in the sirolimus arm, but most were muscle aches and pains that likely reflected the exercise protocol itself. One notable event was a hospitalization: one participant took one dose of sirolimus and a few days later was hospitalized with pneumonia. It is unclear whether that person was already becoming ill before starting the study drug. In a small study like this, I do not think this single event should be overinterpreted.
We also looked at laboratory markers, especially HbA1c. There was a slight increase in the sirolimus group compared with placebo that did reach statistical significance, but the absolute difference was small enough that I do not think it was clinically meaningful.
Matt Kaeberlein:
That all sounds right. In organ transplant patients, rapamycin is well known to affect glucose homeostasis and insulin sensitivity, so if we were going to see side effects, we might expect effects on A1c, insulin sensitivity, or lipids. To me, this does suggest there were modest metabolic effects in this population.
That makes the whole trial moderately cautionary. Again, it is somewhat surprising because there are other data—some from larger though less tightly controlled studies, and some using everolimus—where comparable doses did not produce this kind of side-effect signal. That makes me wonder whether this particular population was especially sensitive, whether the dose was too high, or whether the formulation mattered.
My guess, though it is still just a guess, is that the dose was a little high and that spreading the dosing out over longer intervals might have reduced some of the concerning signals.
Speaker 1:
We used branded sirolimus from Pfizer to avoid the issue that some compounded generics are not enteric-coated and may be degraded in stomach acid. Pfizer had no role in the study and did not donate the drug—we bought it ourselves.
There was also no matching placebo available, so we had to put the triangular tablets into larger capsules and fill the rest with cellulose, and use cellulose-filled capsules as placebo. One possible reason for the results is that by using branded sirolimus we may have ensured participants really did receive the full 6 mg dose.
Matt Kaeberlein:
Right. Theoretically, generic sirolimus products should match the pharmacokinetics and bioavailability of the branded version, but they are formulated differently and may not be identical. Compounded versions can vary even more. In some cases, as in the PEARL trial, lack of enteric coating may mean much less drug enters circulation.
If we could do the study again with more resources, we would have measured blood rapamycin levels repeatedly throughout the trial to assess pharmacokinetics and clearance directly. We just did not have that information.
There is a new clinical study out of the University of Arizona with preliminary data on weekly dosing at 4 mg or 8 mg in about 50 people, and it includes pharmacokinetics. That should help us understand how variable weekly dosing is across individuals.
Speaker 1:
That brings us to the discussion. Contrary to the cycling hypothesis—the idea that we could periodically suppress mTOR with rapamycin and then allow exercise-driven rebuilding—we found no evidence of benefit. Instead, the sensitivity analyses suggested statistically significant blunting of functional gains in the sirolimus arm.
One possible mechanistic explanation is that we did not quite get the timing balance right. Although participants took sirolimus 24 hours after the final weekly exercise session, sirolimus has a rough half-life of about 62 hours, so there may still have been enough in the system to affect the first or even second exercise session the following week.
Matt, you had some thoughts on that half-life figure.
Matt Kaeberlein:
Most pharmacokinetic data come from organ transplant patients taking several milligrams every day, which creates steady-state drug levels. In that setting, the measured terminal half-life reflects metabolism under a different condition than once-weekly dosing.
So I’m hesitant to simply apply the 62-hour half-life from transplant settings to this context. What matters more here is what serum levels look like with weekly dosing. In unpublished data I’ve seen from Bonnie Lafleur’s study, most people on weekly dosing are below the detection limit before the next weekly dose, and they do not seem to accumulate the drug in serum over time.
That suggests the drug is cleared from serum over the week. But serum levels do not tell us what is happening inside muscle cells, liver cells, kidney cells, or other tissues. It is entirely possible that tissue levels remained high enough to inhibit mTOR in muscle several days after dosing, even if serum levels were low.
Without muscle biopsies and direct mTOR signaling measurements, we cannot know for sure whether mTOR complex 1 in muscle was still inhibited 3, 4, 5, or 6 days after the last dose. But your hypothesis is quite reasonable.
Speaker 1:
Exactly. Even if the drug was mostly gone before the next dose, we do not know what happened on day 2 or day 3. If sirolimus was still present then, that may have impaired the exercise response.
That is why in the paper we suggested future studies should test longer intervals between doses, perhaps every 3 weeks or every 6 weeks, so that the drug is definitely out of the system during most exercise sessions. The problem is that the ideal follow-up would probably be a 12-month, three-arm study—placebo, every-3-weeks dosing, and every-6-weeks dosing—which would cost millions.
Matt Kaeberlein:
I agree. But there are other hypotheses worth considering too. For example, what if after a 12-week washout, the rapamycin group would have caught up? This study looked at older adults who were not already on a dedicated exercise program and then started one. Those people should improve fairly quickly. That may be different from studying people who already exercise and asking whether rapamycin improves muscle homeostasis over the longer term.
A design with 12 weeks on weekly rapamycin followed by 12 weeks off while continuing exercise might have produced a different outcome. We simply do not know.
The broader point is that, because there has not been enough funding for the right clinical trials, we are still guessing about dose and frequency. We have a little evidence and a lot of cultural assumptions, but not enough high-quality data to know whether one week, two weeks, three weeks, or longer intervals are best.
Also, the optimal dose for brain or heart may not be the same as the optimal dose for muscle. It would not surprise me if higher doses were needed for some potential brain effects, while those same doses might interfere with muscle adaptation.
Speaker 1:
We deliberately selected people who were not already doing lots of exercise because we wanted to be able to observe a change in the chair stand test. I was also hoping rapamycin might help treat age-related anabolic resistance—that is, help older adults gain more from exercise than they normally would.
So I had hoped the rapamycin group would improve more than placebo. Even if a later washout period let them catch up, to me that still suggests the once-weekly dose was too frequent in the context of muscle performance. My central takeaway is that once-weekly dosing in older adults may be too frequent for this endpoint, and that outcomes might be better with every-3-weeks or every-6-weeks dosing.
A lot of the choice of weekly dosing was cultural. At the time, many people in the longevity field were taking rapamycin once a week.
Matt Kaeberlein:
I agree that some of it was cultural, though there is at least some trial evidence suggesting weekly dosing can have lower side effects than daily dosing while preserving some effects, such as on vaccine response. Still, much of this is hypothesis-driven.
Let me offer an alternative mechanistic possibility. Aging is associated with chronic inflammation across tissues, and that inflammation impairs normal function. From rodent data, it may take 4 to 8 weeks of fairly consistent mTOR complex 1 inhibition to reduce that inflammatory state. Then, after stopping treatment, function may improve because stem cells, immune cells, or tissues are functioning more youthfully.
So it is possible that a period of fairly robust mTOR inhibition followed by washout is needed to see some benefits of rapamycin. That might matter especially for immune-mediated or inflammation-related endpoints, while hypertrophic muscle endpoints might behave differently.
The truth is that we still do not fully understand the mechanisms behind rapamycin’s benefits even in preclinical models, so our current models may be incomplete.
Speaker 1:
I agree. It is a shame there is not enough funding in this field to answer these questions properly. I would love to run a follow-up study that gets us closer to the truth, but funding such a study is challenging.
Matt Kaeberlein:
That has been true for a long time. Still, I’m optimistic. The University of Arizona study is relatively large—more than 700 people—and should answer some important questions. My frustrated side says that should have been done ten years ago, but at least it seems to be happening now.
Hopefully, going forward, we can join forces again to answer some of the questions raised in this conversation.
Speaker 1:
I really wanted to thank you for all your support during this five- or six-year journey. I’ve appreciated being able to bounce ideas off you and have these conversations.
Matt Kaeberlein:
Right back at you. I have a lot of respect for your persistence and for putting your own resources on the line to do this clinical trial. You are truly dedicated, and I really value you as a friend and appreciate what you have done.
Summary
This video discusses the results of a small, preregistered 12-week randomized trial in older adults aged 65–85 testing whether once-weekly sirolimus/rapamycin (6 mg) combined with home exercise improves muscle-related outcomes compared with placebo plus exercise.
The core finding is negative to cautionary:
- The primary endpoint (30-second chair stand test) did not reach statistical significance in the main intention-to-treat analysis.
- In prespecified sensitivity analyses (case-complete and per-protocol), the placebo group improved more than the rapamycin group.
- Secondary endpoints such as 6-minute walk and grip strength also did not reach significance, but tended to point in the same direction.
- Both speakers interpret the overall pattern as suggesting that rapamycin may have blunted exercise-related functional gains in this specific short-term setting, though they are careful not to claim this as fully established.
The proposed original rationale was the cycling hypothesis:
- aging muscle may have excessive mTORC1 activity,
- rapamycin might transiently reduce that,
- autophagy and repair might improve,
- then exercise could reactivate anabolic signaling and rebuild muscle.
Instead, the study suggests that the chosen protocol may have interfered with exercise adaptation rather than enhancing it.
They discuss several possible explanations:
-
Dose too high
Six milligrams weekly may have produced too much mTOR inhibition for this endpoint. -
Wrong timing/frequency
Residual drug may still have been present during the next exercise sessions, potentially blunting adaptation. -
Wrong study window
A 12-week intervention may have been too short, and benefits—if any—might only appear after a later washout phase. -
Endpoint-specific effects
What is good for inflammation, immune function, brain, or heart may not be good for short-term muscle adaptation. -
Population matters
These were older adults not already in structured exercise training, so the study was testing early adaptation to exercise, not maintenance in already-trained people.
Safety was described as modestly cautionary:
- more adverse events in the sirolimus arm, though many were exercise-related aches,
- one pneumonia hospitalization after a single dose, but causality is uncertain,
- a small statistically significant increase in HbA1c, judged by the speakers to be of limited clinical importance.
The overall message from both speakers is not “rapamycin is bad,” but rather:
- this particular weekly 6 mg protocol did not help muscle function in this setting,
- and may have slightly impaired training gains,
- so future studies should test different dosing intervals, longer duration, washout designs, and pharmacokinetic monitoring.
Critique
1. Main strengths
Preregistration and rigor
This is the strongest part of the study as presented. They emphasize that endpoints and sensitivity analyses were preregistered. That matters because it reduces the risk that they searched through the data after the fact for something publishable.
Reasonable biological rationale
The study was not based on a silly premise. There was real preclinical logic:
- mTOR dysregulation in aging muscle,
- impaired autophagy,
- rodent data suggesting possible benefit for sarcopenia/body composition,
- and a plausible cycling idea.
Real-world design
A home-based exercise program in older adults is practical and clinically relevant. Testing something older adults could actually do is valuable.
Honest presentation of disappointing findings
They do not try to spin the primary outcome into a success. That is a major positive. They repeatedly acknowledge:
- the primary endpoint missed significance,
- sensitivity analyses are weaker,
- and the study should not be overinterpreted.
2. Main weaknesses
Small sample size
Forty participants is very small for a noisy human functional study. That creates three problems:
- low power to detect modest true effects,
- instability from a few outliers,
- and wide uncertainty around subgroup or per-protocol findings.
This is probably the single biggest limitation.
Over-reliance on sensitivity analyses
Even though the sensitivity analyses were preregistered, Matt is right that they are less robust because they break randomization. Once you move from intention-to-treat to case-complete or per-protocol, you are more exposed to bias from adherence, dropout, and participant differences.
So the strongest strictly statistical conclusion remains:
- no significant benefit on the primary endpoint in the main analysis.
Everything beyond that is suggestive rather than definitive.
Short duration
Twelve weeks may be enough to detect early exercise adaptation, but perhaps not enough to test a geroscience intervention whose benefits could emerge more slowly or after washout. This does not rescue the negative result, but it does limit what can be concluded.
No pharmacokinetic or tissue signaling data
This is a major mechanistic weakness. They do not know:
- serum exposure over time in these participants,
- interindividual variation in clearance,
- or whether muscle mTOR signaling was still suppressed when participants exercised.
Without blood levels, biopsies, or mTOR-pathway readouts, the mechanistic discussion remains speculative.
Exercise adaptation versus muscle aging are not the same endpoint
The study is framed partly as a test of muscle aging/frailty, but what it really seems to test is:
- how older previously undertrained adults respond over 12 weeks to starting exercise while taking weekly rapamycin.
That is narrower than “does rapamycin help aging muscle?” It may impair short-term training adaptation even if it has other longer-term tissue effects.
3. Where the speakers are strongest
They are strongest when they say:
- the primary result is negative or null,
- the overall directional pattern is cautionary,
- the dose/frequency may have been wrong,
- and better trials are needed before drawing broader conclusions.
That is a fair, disciplined reading.
4. Where the discussion becomes more speculative
Several ideas are plausible but not demonstrated here:
- weekly dosing was too frequent,
- every 3–6 weeks would work better,
- the rapamycin group might catch up after washout,
- higher doses may be better for brain but worse for muscle,
- tissue levels may persist after serum clearance.
All of these are reasonable hypotheses, but they are still hypotheses. The video generally acknowledges that, which is good.
5. My bottom-line interpretation
My reading is:
- This study does not show that rapamycin improves muscle performance when added to exercise in older adults.
- It provides some evidence consistent with mild impairment of short-term exercise adaptation under this dosing schedule.
- Because the primary endpoint was not significant in the main analysis, the evidence for harm is suggestive rather than conclusive.
- Still, the fact that the direction was similar across several endpoints makes the signal worth taking seriously.
So the practical takeaway is not “rapamycin definitely harms muscle,” but more:
A weekly 6 mg sirolimus schedule in older adults beginning an exercise program looks poorly supported by these data for improving muscle function, and may be counterproductive over 12 weeks.
6. What a better follow-up study would look like
The video itself points toward a stronger next step. Ideally:
- much larger sample size,
- placebo plus multiple dosing-interval arms,
- longer duration,
- washout follow-up,
- pharmacokinetic measurements,
- ideally some biomarker or biopsy data on mTOR signaling/autophagy,
- and perhaps separate cohorts for sedentary older adults versus already-trained older adults.
That would make the interpretation much firmer.
If you want, I can also turn this into a more concise briefing note or a table with claims / evidence / weaknesses / takeaways.