Clinical Peer Review: mTORC1 Inhibition vs. Exercise-Induced Adaptation (Stanfield et al., 2026)
I. Executive Summary
The analyzed data (Stanfield et al., 2026, Journal of Cachexia, Sarcopenia and Muscle) presents a critical challenge to the “longevity culture” surrounding off-label Rapamycin (Sirolimus) use. While Rapamycin is the undisputed gold standard for lifespan extension in diverse animal models—consistently extending life by 10–15% in rodents—this high-quality, preregistered human Randomized Controlled Trial (RCT) reveals a significant translational gap regarding skeletal muscle.
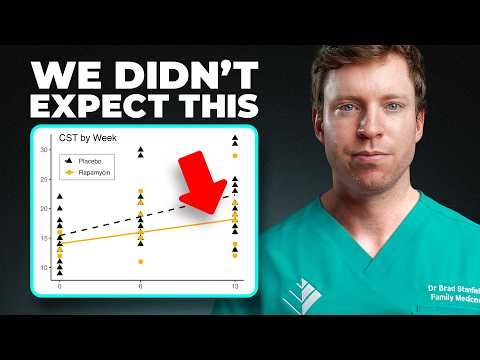
The study investigated whether a “cycling” protocol (6mg weekly, dosed 24 hours post-exercise) could overcome age-related anabolic resistance by inhibiting hyperactive basal mTORC1 and promoting autophagy, while leaving a window for exercise-induced growth. The results “floored” the investigators: Rapamycin did not enhance exercise gains; instead, it blunted them. In the most rigorous sensitivity analyses (Per-Protocol and Case-Complete), the placebo group significantly outperformed the Rapamycin group in functional metrics like the 30-second chair-stand test.
Furthermore, the trial identified specific safety signals that suggest 6mg/week may be too high for older populations. These include a statistically significant (though clinically modest) rise in HbA1c and a serious adverse event of pneumonia, highlighting the drug’s inherent immunosuppressive risk. The trial essentially confirms that the “building mode” (mTORC1 activation) required for exercise adaptation is functionally incompatible with the “maintenance mode” (mTORC1 inhibition) triggered by Rapamycin at this frequency. Future research must pivot toward significantly longer dosing intervals (e.g., every 3–6 weeks) to allow for homeostatic recovery.
II. Insight Bullets
- Direct Blunting: 6mg/week of Rapamycin significantly attenuated functional strength gains from a 12-week exercise program in adults aged 65–85.
- Cycling Failure: Dosing 24 hours after the final weekly exercise session was insufficient to prevent anabolic interference.
- Half-Life Persistence: While serum levels may clear within 7 days, the 62-hour terminal half-life suggests Rapamycin remains in tissues (muscle/liver) during subsequent exercise sessions.
- Anabolic Tension: There is a “real biological tension” between mTORC1 as a longevity target (inhibition) and its role as the master regulator of muscle protein synthesis (activation).
- Placebo Superiority: Placebo groups consistently showed greater improvement in the 30-second chair-stand test across all sensitivity analyses.
- Sensitivity Analysis Significance: When excluding non-compliant participants, the divergence between groups became statistically significant (favoring placebo).
- Immunosuppression Risk: One participant developed community-acquired pneumonia requiring hospitalization after a single dose; causality cannot be excluded.
- Glycemic Variability: A statistically significant rise in HbA1c was observed in the Rapamycin arm, confirming metabolic side effects even at intermittent doses.
- Anabolic Resistance Hypothesis: The trial was designed to test if clearing “cellular trash” via autophagy could restore muscle sensitivity; this was not supported by the 12-week data.
- Bioavailability Variance: The study used brand-name Rapamune (Pfizer) to ensure 100% bioavailability, unlike the earlier “PEARL” trial which used compounded pills with ~30% effective dosing.
- CRP Outliers: Higher average inflammation in the Rapamycin group was driven by two extreme outliers; once removed, no significant anti-inflammatory effect was noted.
- No Quality of Life Gain: SF-36 questionnaires showed no statistical difference in subjective health-related quality of life between groups.
- Suboptimal Frequency: A once-weekly 6mg dose is likely too frequent for older adults looking to maintain or build muscle mass.
- Tissue Specificity: The “optimal” dose for the brain or heart may be higher and more frequent than the optimal dose for skeletal muscle.
- Trial Cost/Rigor: The study cost ~$724,000 and was crowdfunded to ensure total independence from pharmaceutical influence.
- Nadir Levels: Preliminary data from other studies (University of Arizona) suggests most individuals clear Rapamycin from serum before the next weekly dose, but cellular levels likely persist.
- Muscle vs. Lifespan: While muscle gains were blunted, the study does not rule out Rapamycin’s benefits for other longevity hallmarks (e.g., senescent cell clearance).
- Exercise Interaction: Rapamycin may block the ERK1/2 and MNK1 signaling pathways required for contraction-induced protein synthesis.
- Short-Term vs. Long-Term: Matt Kaeberlein hypothesizes that while short-term gains are blunted, a 12-month study might show the Rapamycin group eventually catching up or exceeding placebo.
- Lack of PD Markers: The study did not perform muscle biopsies to confirm the actual degree of mTORC1 inhibition in the tissue (a noted limitation).
III. Adversarial Claims & Evidence Table
| Claim from Video | Speaker’s Evidence | Scientific Reality (Current Data) | Evidence Grade | Verdict |
|---|---|---|---|---|
| Rapamycin blunts exercise adaptation in humans. | Stanfield Trial results (2026). | Confirmed in this study and supported by acute protein synthesis data Drummond et al., 2009. | B | Strong Support |
| mTORC1 is hyperactivated in aged human muscle. | Cited as rationale for the study. | Evidence in rodents is strong; human data is mixed, often showing blunted activation post-exercise Francaux et al., 2016. | C | Speculative |
| Rapamycin is safe at 6mg weekly. | Preliminary clinical experience. | Study found HbA1c rise and a serious pneumonia case. FDA safety warnings for Sirolimus include immunosuppression, dose dependent FDA PI. | B | FDA General Safety Warning |
| Intermittent dosing (once/week) prevents side effects. | Cultural consensus in longevity community. | Blunted muscle gains and glycemic changes suggest 6mg weekly dosing is still “too much” for homeostasis Stanfield et al., 2026. | B | Unsupported |
| Rapamycin improves body composition (Lean Mass). | Referenced “PEARL” trial. | PEARL trial showed gains in women but used compounded drug with low bioavailability Moel et al., 2025. Results are inconsistent. | B | Plausible |
IV. Actionable Protocol (Prioritized)
High Confidence Tier (Protocol Changes Based on Evidence)
- Decouple Rapamycin from Hypertrophy: Do not initiate Rapamycin if the primary goal is gaining functional strength or muscle mass via a new exercise program.
- Monitor Glycemic Markers: Anyone on off-label Rapamycin should monitor HbA1c and fasting insulin quarterly, as even 6mg/week can perturb glucose homeostasis.
Experimental Tier (Refined Dosing Hypotheses)
- Extend Dosing Intervals: To avoid the “blunting” effect seen in this trial, explore dosing every 14, 21, or even 42 days. This mimics “transient” inhibition seen in animal models that successfuly reversed aging phenotypes.
- Wash-out Periods: Implement an 8-week “on” / 8-week “off” cycle to allow for muscle adaptation during the “off” phase while capturing geroprotective benefits during the “on” phase.
Red Flag Zone (Safety Data Absent)
- Avoid Compounded Rapamycin: Bioavailability is unreliable. Only use FDA-approved, enteric-coated formulations if participating in clinical research.
- Acute Infection Protocol: Stop all Rapamycin immediately at the first sign of bacterial or viral infection (fever, cough), given the hospitalization case noted in the trial.
V. Technical Mechanism Breakdown
The primary mechanism at play is the mTORC1-S6K1 Pathway.
- Anabolic Signaling: In healthy muscle, resistance exercise activates mTORC1 via the mechanotransduction pathway and IGF-1/PI3K signaling. This leads to the phosphorylation of p70S6K and 4E-BP1, which are the master switches for mRNA translation and new protein synthesis.
- The “Volume Dial” Conflict: Rapamycin functions as an allosteric inhibitor of mTORC1 by binding to FKBP12. Stanfield’s data suggests that at 6mg/week, the “volume” of mTORC1 is turned down so low—or for so long—that the muscle cannot respond to the mechanical stimulus of exercise.
- Autophagy vs. Synthesis: The “Cycling Hypothesis” aimed to promote autophagy (cellular cleanup via Lysosomal degradation) during the “off” days. However, if mTORC1 remains inhibited, the cell remains in a “catabolic/maintenance” state, preventing the “anabolic/growth” state necessary for the hypertrophy seen in the placebo group.
- mTORC2 Inhibition: Chronic or high-dose Rapamycin can also inhibit mTORC2, which regulates insulin sensitivity and the cytoskeleton. The rise in HbA1c observed in the trial is a classic indicator of potential mTORC2 interference or reduced glucose uptake in the muscle.
Interesting, I just had a nasty infection with Norovirus that started on Monday, April 13th. It started on afternoon after my morning dose of rapamycin 6 mg. I was exposed to a person with symptomatic on Saturday, April 11th, so the incubation was a perfect 48 hours. I didn’t realize they had Norovirus, they told they were have some GI issues. Have I known I was exposed to the virus I probably would have skipped the dose.
I remembered contracting Norovirus last year from my wife and it wasn’t nearly half as bad - and she didn’t even isolate. My wife didn’t contract it from me but I did sleep in a different bed.
So, yes I skipped the dose last Monday and rethinking the dosing schedule of the rapamycin. Another reason my labs from yesterday showed a reduced WBCs and hs CRP under 1. This is a very low state of inflammation which I also attribute to my super strict diet and exercise regimen. Low WBC may also be post-infectious. I wonder if someone has a very low state of inflammation… is rapamycin even still helpful ???
This is why I love this site!
I am grateful you shared your story because I was going to try my first larger dose tomorrow, with the idea of going to every 14 day dosing.
I am traveling tomorrow and it completely slipped my mind that I might have all sorts of exposure. I’ll now wait until I return.
Thx again, and sorry you got so sick.
What animal models are they referring to which exhibit the “transient” inhibition resulted in “successfully reversed aging phenotypes”? Insofar as I’m aware, the vast majority of rapamycin studies in animals were continuous exposure. The prominent studies where weekly dosing was used were the cat HCM studies where aging was not an endpoint, and MK’s dog project (where we don’t have the full results as yet).
There have been about half a dozen mouse studies with periodic, or interval dosing of rapamycin. Here is one…
I think it is. My background CRP has crept up a bit. I think I know how I can get it back below 0.15mg/L. My last few weeks results were: 0.26 0.29 0.4 0.35 0.57 0.32 1.51
I take Monday’s result (remembering I test this every week) as meaning I was infected in some way. I am not clear on what this is, however. I am due a Rapamycin dose on 1st May, but I am going to delay this until after my wife’s election result which is 8th May because I don’t want to have a low immune system whilst supporting her in an election campaign.
I take rapamycin to encourage selective mitophagy. I think this feeds into background CRP via a reduction in senescence, but senescence is in part caused by a reduction in SLC25A1 through IL-10 and the Janus Kinase. (this is a mechanistic hypothesis with some evidence).
One of the things I have not been able to get useful information on is whether it is possible to encourage selective mitophagy in the stem cell stores which are held in a hypoxic environment and to some extent a state of dormancy.
What I would suggest to people going for infrequent larger doses (which is what I do) is to build up gradually to minimise the chance of negative side effects.
I was hoping for some guidance, other than from Claude!, so thank you!!!
Recently, I’ve been dosing 5 mg rapamycin every Monday. With my workweek schedule, workouts stay lighter than the intense sessions I save for weekends.
As a carnivore dieter, I’ve sustainably dropped 40 pounds, right to my limit without calorie counting or suffering…and it’s held steady for two years. I can eat as much fatty meat as I want. At 6’6" (factoring in some age-related spinal compression), I’m 245 lbs. now; 225 was my skinny high school weight. Feeling solid at 64 years young, and with 5 mg, I’ve avoided cold sores (which I hate) or other adverse effects.
In my view, we don’t really know the optimal dose yet…Dr. Matt suggested in his Dr. Brad YouTube interview that 6 mg might be too high for older folks. So my confirmation bias says 5 mg could be better. I also wonder about dose timing around exercise: Maybe we should hit hard workouts every day except recovery, and if muscle gains are blunted post-rapa, so what?
From the mice studies and Dr. Blagosklonny’s papers, I strongly disagree. In fact I am betting my life on it
Not disparaging Dr. Stansfield, but comparing credentials with Dr Blagosklonny’s and Dr. Blagosklonny’s lifetime practice. I choose to follow Dr. Blagosklonny’s advice. Each must choose their own path to follow.
“Later, experiments in mice suggested that low doses are suboptimal”
“In mice, the higher the dose, the longer lifespan.”
"Therefore, in humans, the highest dose that does not yet cause unacceptable side In mice, the higher the dose, the longer lifespaneffects (maximal tolerated dose) may be optimal for longevity. If (unacceptable) side effects develop, the dose should be decreased.
“Side effects of rapamycin are not remarkable at all. They are even less dangerous than the side effects of most other drugs.”
You might be right! We will see. The expert who made the comment was Dr. Matt K., and I think the comment was more directed to hypertrophy versus longevity.
IMO it is a question of trough levels rather than dosing.
Dr. Alan Green took 10mg per week and he saved himself many years in life after knocking on deaths door with heart problems. Hes another good anecdote
Like @desertshores he was old as methuselah… and had significant heart issues… as with Blagosklonny and his decades of cancer, so maybe beneficial when in one 's 80’s ![]() . When I went up higher 10 -12 mg in my early 60’s…I got a negative effect… turned my MTOR1 too low. Started aging biological markers faster after a year of solid reversals. Good to monitor with tests…I use GlycanAge for inflammation and TruMe Labs for DNA Methylation.
. When I went up higher 10 -12 mg in my early 60’s…I got a negative effect… turned my MTOR1 too low. Started aging biological markers faster after a year of solid reversals. Good to monitor with tests…I use GlycanAge for inflammation and TruMe Labs for DNA Methylation.
Let’s see if I get old enough to try higher…glad I started at 6 mg at 60’s.
If I was 40… wouldn’t do more than 4 mg… 2 mg at 30. Could be a pattern 2 mg for every decade starting at 30. As I near 70… I have been popping up to 8 mg every few doses or so.
.
As i have said previously, the trough matters. mTOR has a role to play and should be uninhibited from time to time.
I wouldn’t dare go near him as an example of having benefited from RAPA. I don’t usually like anecdotes that are based on oh if he didn’t take Rapa he would have died way earlier. How would you/I know that? How about playing devil’s advocate and saying if he didn’t take Rappa he would have easily lived another 10 or so years by watching his lipids and doing SGLT2, Eze, and Pita and maybe metformin as an example. While his ills to start may have had nothing to do with Rapa, perhaps RAPA signed off his demise by wrecking his immune system? Who knows - right. Bottom line the guy dropped dead before he was even 80? I think (and was taking RAPA). That is NOT a very good sign if you were to ask me. Find me an example of a guy/gal that was taking Rapa regularly and they are (or lived) at least to a 100, then I’ll accept it. As a rule of thumb, I’d say that every time someone whose taking Rappa dies before reaching 90 (regardless of the cause) we should call that a very bad sign and every time someone that’s taking Rappa dies and is over 100 we should consider that a very good sign. If’s and but’s and other scenarios can fly around and every conceivable direction LOL. The best we have so far is @desertshores which his case is very promising, I’ll admit to that, but regardless of how much I search and search all over I have yet to come with a concrete case of a human being past 90 that has been taking rapa say in last 10 years, or even 5. Until that becomes the norm (meaning there is a lot of people that fit the category, I’m going to be VERY skeptical while still continuing to take RAPA.
Having said that I do take RAPA myself but I’m not the one to make up stories in my own mind to convince myself that RAPA will help me live longer. I’m gonna be brutally honest, while I think RAPA has probably helped some with inflammation and joint discomfort, I can also attest that couple times I have had very nasty cold like symptoms (way worse than what I would normally have when used to get sick pre RAPA days) which actually were so bad that I was thinking to myself, imagine if I’m in advance age (77+) and getting this kind of symptoms most likely won’t be able to make it. I’m almost certain that RAPA was the culprit of the worsened symptoms via destruction of the immune system, while the cold itself might or might not have been affected by it. So, keep that at the back of your mind that in case you might get hit with some nasty virus or cold etc. RAPA may not be your best friend. I’m even tempted to think that RAPA will be a contributor to one’s demise should a nasty viral infection were to occur when you are in advance age. I do however believe that for those lucky ones that have never had any bad side effects from RAPA then the benefits should far out way the risks. For unlucky ones (unfortunately I tend to be in this group) that we tend to often get side effects the opposite may be true.
Again, I’m not giving up on RAPA yet, but I’ll be very open minded and watching it closely as I go along.
I don’t know why and this is entirely based on anecdotal and how I feel but I am starting to think that a combination of PITA, EZE, EMPA, TELMISARTAN, and perhaps Metformin may offer WAYYYYYYYYYYYYYYYYYYY more benefits for longevity than Rapa(which to me means reaching about 105 years old, because beyond that there is nothing that we know of as of now that can subjectively help). I take them all and can attest to NOT being able to identify any side effects at all while absolutely getting HUGE benefits in way of lower glucose, lower inflammation, and lower lipids. Just my unbiased and untainted (by euphoria and placebo LOL) opinion. BTW, this is my earnest opinion, and it is absolutely not meant to be argumentative in any way shape or form.
If I may ask, was your renal failure due to antiphospholipid syndrome (APS) by any chance? There are several case reports of stellar improvement in the microangiopathy of APS patients when rapamycin is added to the drug regimen of these trasplant patients. The Reader’s Digest version is APS antibodies attack the intimal lining of renal microvasculature => mTOR overactivety => thickening of the intima => sluggishness of blood flow in these tiny vessels => clots forming => tissue death. Rapamycin knocks that overactivity down, the intimal thickness regresses, and blood flow is restored.
Keep in mind that regarding heart health specifically, rapamycin is heart protective. We know this from validated studies in pets. To the point that a rapamycin formulation is actually FDA approved for a heart condition in cats (HCM) - that’s about as rigorous as it gets. Matt Kaeberlein’s idea of testing rapamycin in dogs was that dogs age similarly to people, suffer from a similar range of diseases and share our exact environment, and so are an excellent model for human aging. From preliminary results (the sudy is ongoing), there are reports that rapamycin actually reverses aging of the heart, and brings it back to more youthful functionality. Therefore it seems eminently sensible that Dr. Green would select rapamycin to treat his heart condition, which he claimed helped him greatly. We all are gambling, but regarding heart health, I personally am pretty sanguine about the impact of rapamycin on my heart health. YMMV.
Agreed it’s a gamble, but I view it a as semi-informed gamble that can be mitigated with pauses etc.
A story on Dr. Green piqued my interest in rapamycin many years ago, and started dosing 5+ years ago.
That said, I’ve taken breaks and now am coming off my longest break of about 6 months and looking for advice on reintroducing. I took up running and paused to not mess with Boston training. With the race last Monday, I’m looking to both start dosing and continue training (I’m hooked ![]() ). I’ve liked my prior 6mg/10 day routine which I’d take on easy or off training days.
). I’ve liked my prior 6mg/10 day routine which I’d take on easy or off training days.
Would love feedback from the geniuses on this board.
Thanks