This study investigates the effects of SRN-901—a novel oral combinatorial drug that consists of urolithin A, quercetin, nicotinamide riboside, alpha-lipoic acid, and Seragon’s SRN-820
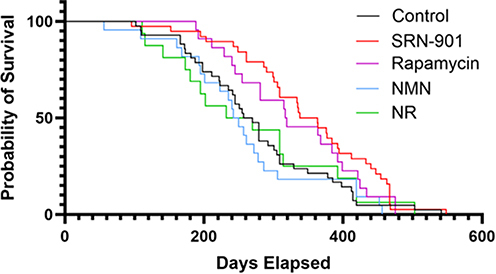
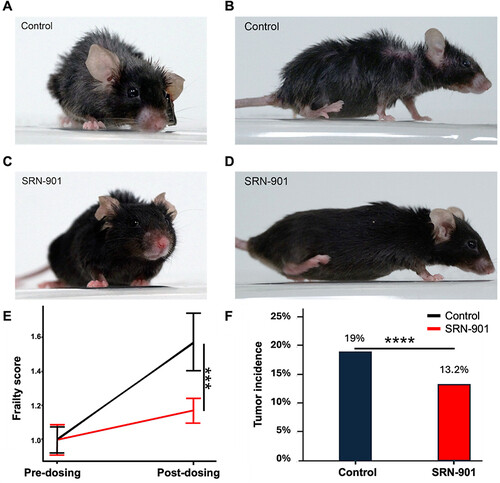
SRN-901-treated mice showed a significant increase of 33% in median remaining lifespan compared to placebo-treated mice. Cox proportional hazards analysis revealed a hazard ratio of 0.54, indicating that SRN-901 treatment was associated with a 46% reduction in the hazard of death. While rapamycin increased lifespan in adult mice, nicotinamide mononucleotide (NMN), and nicotinamide riboside (NR) did not show significant differences in median lifespan compared to placebo. SRN-901 protected mice against increased frailty during aging, with baseline-normalized scores rising to 1.17 in treated mice and 1.57 in controls, corresponding to a 70% attenuation of frailty progression between pre-treatment (D-14) and post-treatment (D128; p < 0.001). Transcriptomic analyses revealed that SRN-901 modulates gene expression across pathways implicated in aging biology, including inflammation, apoptosis, and DNA repair, as well as gene sets associated with neurodegenerative disorders, including Alzheimer’s disease. Metabolic profiling revealed that SRN-901 was associated with attenuation of several age-related metabolic shifts, resulting in a blood metabolite profile that more closely resembled that of younger mice. The upregulation of glutathione metabolism and other longevity-related pathways underscores SRN-901’s role in enhancing cellular defenses against oxidative stress and maintaining metabolic health.
The 500 mg/kg/day SRN-901 dose was selected by combining nicotinamide riboside, quercetin, urolithin A, and alpha-lipoic acid in equimolar concentrations, consistent with doses previously reported in the literature to be well tolerated and to influence metabolic or aging-related pathways, together with Seragon’s SRN-820 at an internally established percentage that produced robust target engagement without gastrointestinal intolerance in prior exploratory studies. NMN (300mg/kg) and NR (300mg/kg) were purchased from Effepharm, and fed through oral gavage 6 days a week. Rapamycin was mixed in food with a concentration of 14.4 ppm. All groups (SRN-901, NMN, NR, Rapamycin, and Placebo), underwent an identical handling and gavage schedule (same frequency, time-of-day, and volume) by trained staff using a standardized procedure using either group-specific drug or vehicle (water) and no deviation events were recorded.
I wonder why they didn’t compare to urolithin A alone…