I was the one who started the food poisoning thread, though this went on weeks after rapamycin stoppage (hopefully not the rebound, !)
Every three weeks? interesting schedule
Yes, it was rookie longevity stuff by myself, to take everything at the same time (including prescripts statins/jardiance/acaarbose at the same time out of laziness
Also, in regards to this rebound situation, I’m thinking of either going daily, or real low 1mg/weekly
The rebound occurs in the study only if used intermittently? yes? And I’m starting to think my fat loss (I was looking good) occurred when I was using daily. Some studies I’ve seen on other animals found a dose dependent positive response. So taking 1 mg daily at 45 years old, even if it ends up being 5x the dose because of the supplements I take around it, might not hurt?
Also is 1mg weekly too low to do much?
Going daily with 1mg will give you mouth ulcers. I tried that. Did not work for me. And 1mg per week will hardly do anything imo.
1 mg daily with absorption enhancers could make your dose effectively 3-5X higher. That much daily might not be a good idea. If you do 1 mg plus the enhancers - GFJ, Metformin, fatty meal - that might put you in the 3-5 mg weekly dose which may be low but fine. If you feel it is too low, you could double it and you’d be in the ballpark with most people and would not need to worry about rebound.
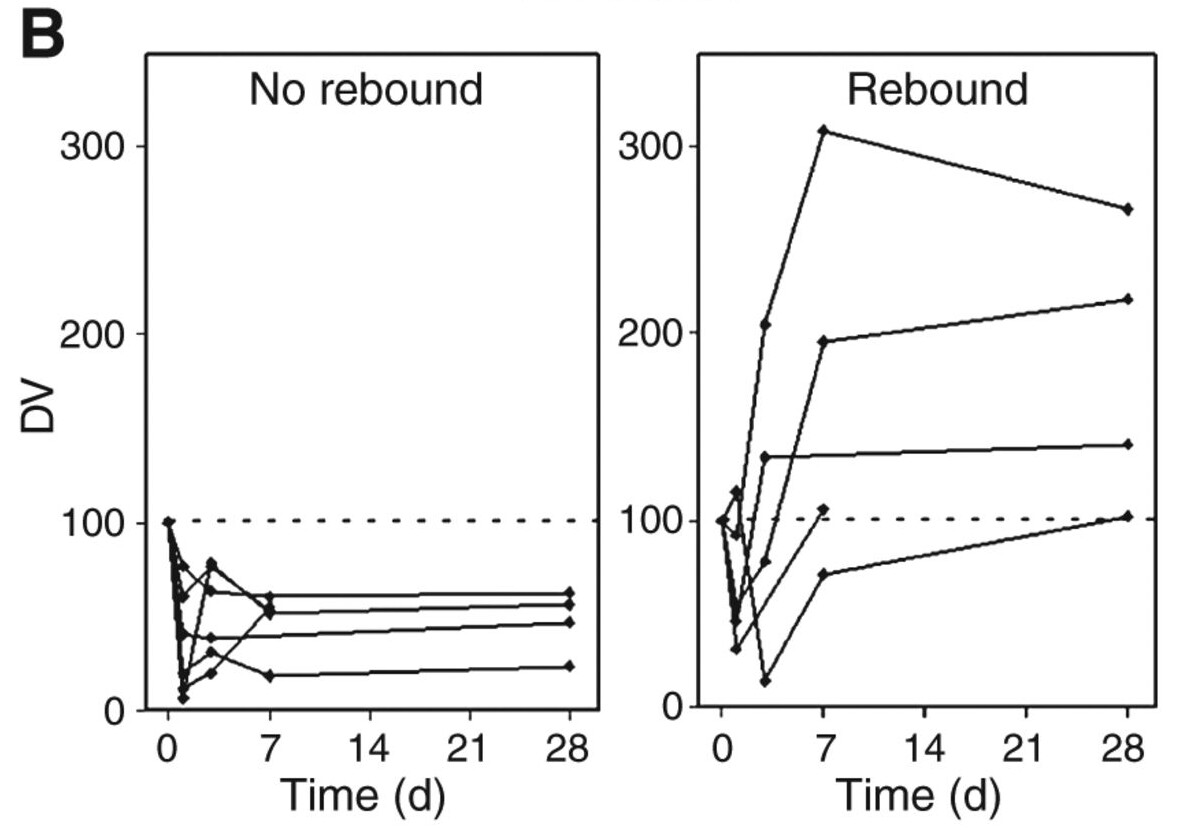
@sunshine4 As far as I can tell from that study they took all the subjects that appeared to have rebound (which I think is variation in their S6 kinase phosphorylation assay) and graphed them together (regardless of dose schedule) then they labeled the graph “rebound” (Fig 2B). Then they took all subjects that didn’t appear to have rebound and graphed them together (a mixture of intermittent and constant dosing) and labeled that graph “no rebound” (Fig 2B). It’s not clear to me that “rebound” is anything more than variability in their ability to measure the amount of S6 kinase phosphorylation.
This study only investigated weekly dosing:
Materials and Methods
Dosing cohorts
Sirolimus was administered once weekly as a 1 mg/mL oral solution (sirolimus alone and sirolimus plus grapefruit juice studies) or as a 1-mg tablet (sirolimus plus ketoconazole study).
This is incorrect. Noise/variation/variability would look like this:
Instead, what (Cohen et al. 2012) saw was a delayed surge in phospho-p70S6K for a subset of participants. The trend was consistent across four to five measurements (from day 0 to day 28):
This also wasn’t an unusual or finicky assay. They performed Western blots to assess phospho- and total p70S6K. Westerns have been a staple in molecular biology labs since the 1980s.
Please see my previous post for more context. In short, though, they performed the rebound analysis in participants where they had at least four time points of phospho- and total p70S6K.
Lastly, the mTOR rebound is a well-established phenomenon in cancer research—it just doesn’t go by that name. See, for example: Suppression of feedback loops mediated by PI3K/mTOR induces multiple overactivation of compensatory pathways: an unintended consequence leading to drug resistance.
So. It seems there is a rebound effect that affects some people who take Rapamycin.
Without reading through all the documentation what are the circumstances in which such a rebound occurs (if it occurs for any one of a number of individuals)?
mTORC1 Rebound Effect:
So many things are a double-edged sword: There is opposition and rebuttal in all papers published. There is no guarantee that anything we read is true.:
The rebound effect isn’t all bad mTORC1 activation may help with muscle hypertrophy.
And why I am now much more interested in peak values than AUC in taking rapamycin.
“Even though there is more to muscle growth than activating mTOR, coaches and athletes should be trying to increase mTOR activity as much as possible.”
“If humans are given the immunosuppressant rapamycin (blocks mTOR) before they perform their strength training, there is no increase in protein synthesis (Drummond et al., 2009). This demonstrates that mTOR is required for the increase in muscle protein synthesis after resistance exercise.”
“Skeletal muscle hypertrophy largely depends on protein turnover, defined as balance between protein synthesis and degradation. Activation of mTOR signaling positively affects processes of protein transcription and translation, and inhibits autophagy, leading to increase in size of muscle fibers.”
“However, in adult mammals, resting mTORC1 activity is reduced and does not appear to be necessary for the maintenance of muscle mass since the suppression of resting mTORC1 activity in adult muscle by rapamycin or raptor deletion does not induce muscle loss but does cause a slight reduction in basal protein synthesis.”
However the same paper bottom line:
“Although activation of mTORC1 may not be required for the maintenance of muscle mass in adult animals, it does appear to be a critical pathway for the induction of adaptive muscle growth, especially in response to mechanical overload.”
“mTOR signaling pathway and mTOR inhibitors in cancer: progress and challenges”
“Autophagy and apoptosis of cancer
On the one hand, autophagy can keep the genome stable by removing damaged organelles and misfolded proteins, so it can inhibit the growth of cancerous cells [46]. On the other hand, autophagy provides the tumor with more nutrients, which strengthens the tumor’s ability to cope with extreme environments [47, 48]. In addition, the unlimited proliferation of tumors is partly due to the inhibition of tumor cell apoptosis.”
https://www.gssiweb.org/docs/default-source/sse-docs/sse123_baar_sse_bleed.pdf?sfvrsn=2
mTOR signaling pathway and mTOR inhibitors in cancer: progress and challenges | Cell & Bioscience | Full Text integrates%20 signals%20from%20 multiple,proliferation%20and%20survival%20%5B1%5D.
https://www.sciencedirect.com/science/article/abs/pii/B9780123964540000229#:~:text=Skeletal%20 muscle%20 hypertrophy%20 largely%20 depends on%20size%20of%20 muscle%20 fibers.
Over time, we’ll certainly gather more evidence on how individual molecules affect CYP3A4 and other cytochromes—but short of a massive leap in computational biology, you will not see any studies that assess the effects of these molecules in combination. I would not recommend waiting to dose rapamycin (a well evidenced intervention) due to the unknown modulating effects of molecules with substantially less evidence behind them.
Within reason, I think the timing of a supplement stack matters more than the size. The simplest option—and the one I practice myself—is to give the rest of your supplement stack a break on the day you take rapamycin.
Based on the evidence I’ve found so far, high doses of rapamycin (> 20 mg) cause the rebound. If you’re still concerned and want to buffer against the possibility of a rebound, then consider pairing rapamycin with metformin. See my prior posts in this thread for the rationale.
Probably. In (Kemp Bohan et al. 2021), the only notable effect I recall was that weekly dosing with 0.5 mg (n = 8) and 1.0 mg (n = 3) trended toward an increase in naïve T-cell populations. This was a very small study, but it’s at least one data point.
So, a dosage that would probably avoid the rebound effect would be 20 mg or less weekly + Metformin (just to be safe). Going above this risks rebound.
20 mg would be about 5-6 mg + GFJ.
Rebound is especially bad for cancer.
Thanks. I already skip the supplements on my once a week work out day to avoid mixing dietary anti oxidants with what’s going on in my body, I will just take it then.
The synergistic effects of metformin sound appealing, and its a cheap drug, but I already take jardiance/acarbose for life extension as a non prediabetic, adding metformin too ? Might work, but maybe send my blood sugar too low? I’ll look into it
@McAlister The photo of the figure you posted says it all. Who titles a figure with the conclusion? ie rebound, no rebound? Each time point has a single value with no error bars (despite demonstrating significant error in their assay in figure 1). There are at most 3 time points that show “rebound” in one patient and two other subjects show two time points each (not 5 per subject). They need to do replicates for each time point that include not only repeating the assay but repeating the prep of the protein. There can be significant error when quantifying signal off westerns as well. Their variability likely does look like the graph you posted (red) if you graph the value obtained for one time point and graph it against replicates. (ie value on Y and replicate 1-5 on X axis). See Figure 1 from the paper where they do look at the error of their assay for d2 time points as a representative, the error bars representing the SD of their assay are huge.
I’m just reading the paper… it says “rebound” and “no rebound” nothing more. I wouldn’t make too much of it, it’s likely noise in their assay. It’s unfortunate, it would be nice to see a definitive study.
This is Figure 1 showing the variability in their assay. Pretty sure if they had done replicates of the “Pre” time point the error bars would overlap.
Okay. I think I better understand how you’re processing this paper and coming to your conclusion. Let me clarify a few points and see where that gets us.
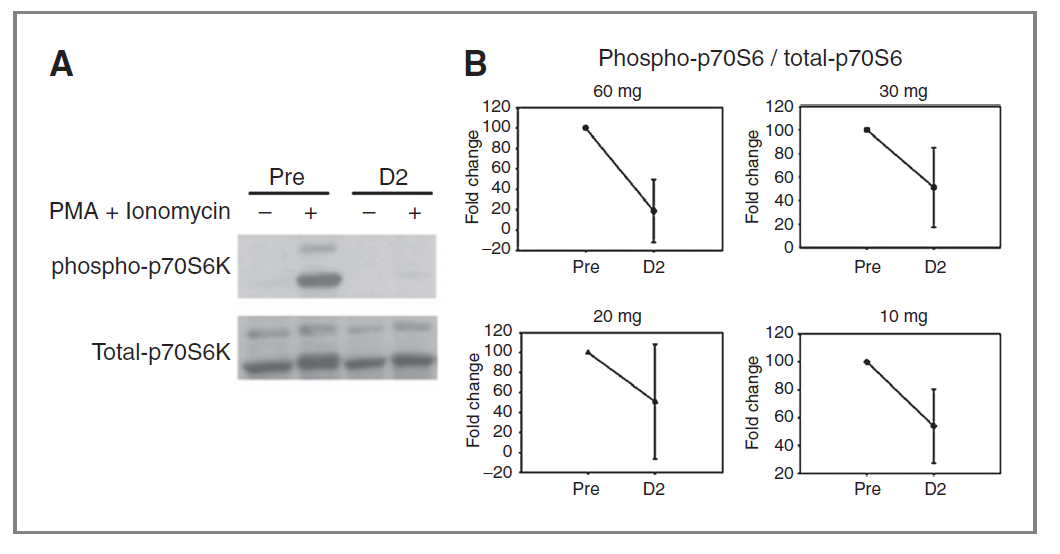
Figure 1B displays the fold change of phospho-p70S6 / total-p70S6 from day zero (prior to dosing rapamycin) to day two. The error bars shown at D2 are not variations in the assay within the same person, they are variations across all the different participants. This is not surprising, as rapamycin is known to have widely different pharmacokinetics and pharmacodynamics from person to person.
There are no error bars for the “Pre” measurement because it’s a fold change—every participant’s initial measurement is 100%.
That would be great, but keep in mind this rebound effect was an ancillary finding. The purpose of the study was to investigate “weekly sirolimus alone or in combination with either ketoconazole or grapefruit juice”.
Assuming I take 20mg of rapamycin with olive oil at once, how many days would I have to take metformin in order to stop the rebound from occuring?
@McAlister So lets take the 20mg cohort. I agree these are values from multiple subjects day 2 S6 P levels (but don’t you agree it is weird to plot the mean and SD of multiple subjects as a single point?). A quick look at their other data does not support that blood levels for Sirolimus varied dramatically within the 20 mg cohort (Cmax was 19.27 with a SD of 5.69 ng/ml Table 4) yet we are seeing 100 fold variation in their S6 assay. I’m still not sure what the “pre” dot represents, why is it plotted at 100 fold change for all dose cohorts, seems like it should be plotted at 0?
Also, look at their western and keep in mind these are day 2 signals barely visible on the blot. Do you think that a day 7 signal (where they began to see “rebound” in most of their subjects) is within the dynamic range of that assay? My guess is error bars get very big out at day 7 where blot background signal noise is comparable to the signal to phosphorylated S6 in the “no rebound” subjects and 300x the “pre” level in one “rebound” subject. But we don’t know because they didn’t measure them or didn’t perform replicates.
Anyway, not saying rebound is good or bad, or exists or doesn’t exist, just pointing out this is an incredibly poor description of an important phenomenon that may or may not be occurring at these doses and time frames. I wouldn’t use this paper to justify changing dose or dose frequency to enhance or avoid rebound because I don’t think their data supports their conclusions or the assumptions people make about what doses cause or don’t cause rebound.
Hi, I am new to the group, and with low Rapamycin exposure.
My dosing was 3mg every month, for six months now. I take metformin 5-6 days a week.
Maybe this is underdose, but I like to be cautious. For a long time I resisted statins. Now I take 10 mg rosuvastatin once in three days, and cholesterol went down from 6.5 to 4.9.
Now I am worried with the Rapa rebound. I was hoping for the intermittent mTOR inhibition only.
So I will stop now and follow the developments. Kind regards.
3 mg a month won’t cause MTOR rebound. You need to break a level of about 20 mg to have the potential of having a rebound effect. Even then there is only a 50% chance once you hit a 40 mg dose.
Yes - just confirming what DeStrider mentioned. Here is the full analysis on rapamycin rebound… you don’t need to worry at that dosing protocol: Rapamycin / MTOR Rebound effect in 3/12 non-GF and non-Keto patients