The Down Syndrome is “progeroid”, that is, it accelerates aging. Thus people with DS would be good subjects for clinical trials with rapamycin as an anti-aging agent, all the more because these people often already are under medical care.
As rapamycin (sirolimus) is now considered quite safe, at least at specific (pulsing) dosing, wouldn’t it be a good idea to give it to people with Down Syndrome and see how it helps them?
New study on DS and accelerating aging
Accelerated epigenetic aging in newborns with Down syndrome
Accelerated aging is a hallmark of Down syndrome (DS), with adults experiencing early-onset Alzheimer’s disease and premature aging of the skin, hair, and immune and endocrine systems. Accelerated epigenetic aging has been found in the blood and brain tissue of adults with DS but when premature aging in DS begins remains unknown. We investigated whether accelerated aging in DS is already detectable in blood at birth. We assessed the association between age acceleration and DS using five epigenetic clocks in 346 newborns with DS and 567 newborns without DS using Illumina MethylationEPIC DNA methylation array data. We compared two epigenetic aging clocks (DNAmSkinBloodClock and pan-tissue DNAmAge) and three epigenetic gestational age clocks (Haftorn, Knight, and Bohlin) between DS and non-DS newborns using linear regression adjusting for observed age, sex, batch, deconvoluted blood cell proportions, and genetic ancestry. Targeted sequencing of GATA1 was performed in a subset of 184 newborns with DS to identify somatic mutations associated with transient abnormal myelopoiesis. DS was significantly associated with increased DNAmSkinBloodClock (effect estimate = 0.2442, p < 0.0001), with an epigenetic age acceleration of 244 days in newborns with DS after adjusting for potential confounding factors (95% confidence interval: 196–292 days). We also found evidence of epigenetic age acceleration associated with somatic GATA1 mutations among newborns with DS (p = 0.015). DS was not associated with epigenetic gestational age acceleration. We demonstrate that accelerated epigenetic aging in the blood of DS patients begins prenatally, with implications for the pathophysiology of immunosenescence and other aging-related traits in DS.
Newborns…wouldn’t have any ex uterus epigenetic interaction. Clearly, the dysregulated somatic mutations in and of themselves, lead to massive “epigenetic” signals in the genome.
Rapamycin should not impact these intrinsic mutations, but perhaps the proliferation of damaging, misfolded proteins and abnormal “myelopoiesis” in the DS phenotype. We already know chronic Rapamycin can impact hematopoietic stem cells. Using rapalogs, it’s likely a balancing act between competing positive/negative interactions in these accelerated aging models.
Mice only die of cancer, Rapamycin extends lifespan. More studies being published showing somatic mutations (eg. cancer) and associated epigenetic amplification control longevity. Large GWAS studies of centenarians show they have better DNA repair/suppression genes, and simply delay mortality by fighting back accelerated aging.
Perhaps we will learn much about mTOR inhibitors and longevity pathways in these accelerated aging models.
What’s Happening TODAY With the Progeria Clinical Trials?
The current trial involves 2 drugs: Zokinvy (lonafarnib) and a new drug, everolimus.
"Phase 1, to determine the safe and appropriate dosage of everolimus, began in April 2016 and was successfully completed in June 2017. Phase 2, which is testing the effectiveness of the 2-drug combination, began in July 2017. Sixty children from 27 countries have enrolled! We expect the trial visits to end within the first few months of 2022, followed by several months of data analysis and eventual publication of results in a peer-reviewed, scientific publication.
Everolimus is a form of the drug rapamycin; everolimus can be more easily given to children with Progeria because it requires fewer blood draws to measure drug levels. While Zokinvy (lonafarnib) may block the toxic progerin from developing, rapamycin appears to allow cells to more rapidly clear out progerin. Thus with rapamycin targeting a different pathway than Zokinvy (lonafarnib), the combination may prove to be a “one-two punch” to Progeria – hopefully a better treatment than Zokinvy (lonafarnib) on its own.
The Science Behind the Addition of this Second Drug
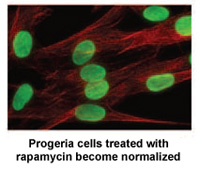 Rapamycin is an FDA-approved drug that has previously been shown to extend the lives of non-Progeria mouse models. A study* by researchers at the NIH in Bethesda, MD and Massachusetts General Hospital in Boston demonstrates that rapamycin decreases the amount of the disease-causing protein progerin by 50%, improves the abnormal nuclear shape, and extends the lifespan of Progeria cells in the laboratory.
Rapamycin is an FDA-approved drug that has previously been shown to extend the lives of non-Progeria mouse models. A study* by researchers at the NIH in Bethesda, MD and Massachusetts General Hospital in Boston demonstrates that rapamycin decreases the amount of the disease-causing protein progerin by 50%, improves the abnormal nuclear shape, and extends the lifespan of Progeria cells in the laboratory.
Rapamycin is known for its anti-aging properties in mice. These findings are part of a growing list of studies that help to validate the theory that finding the cure for Progeria may also benefit the entire aging population."