Dr B & others have spoken of the possibility that high intermittent doses can lead to surges in mtor during the period in which no rapa is taken. The question would be what dose regime, if any, would mitigate this risk without exposing the body to excessive rapamycin levels.
The problem is that I don’t understand what “surges” means. Can you explain that.
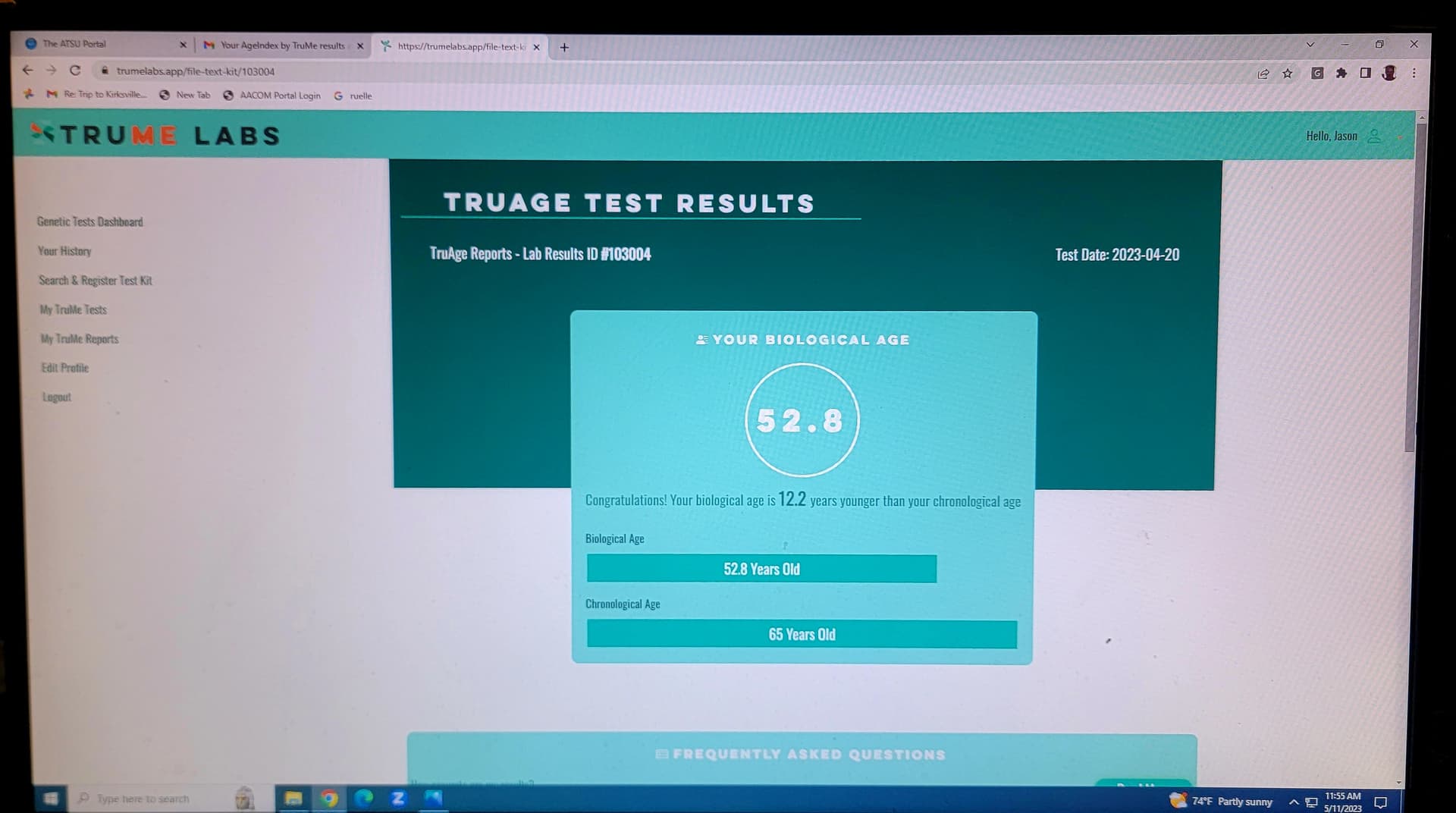
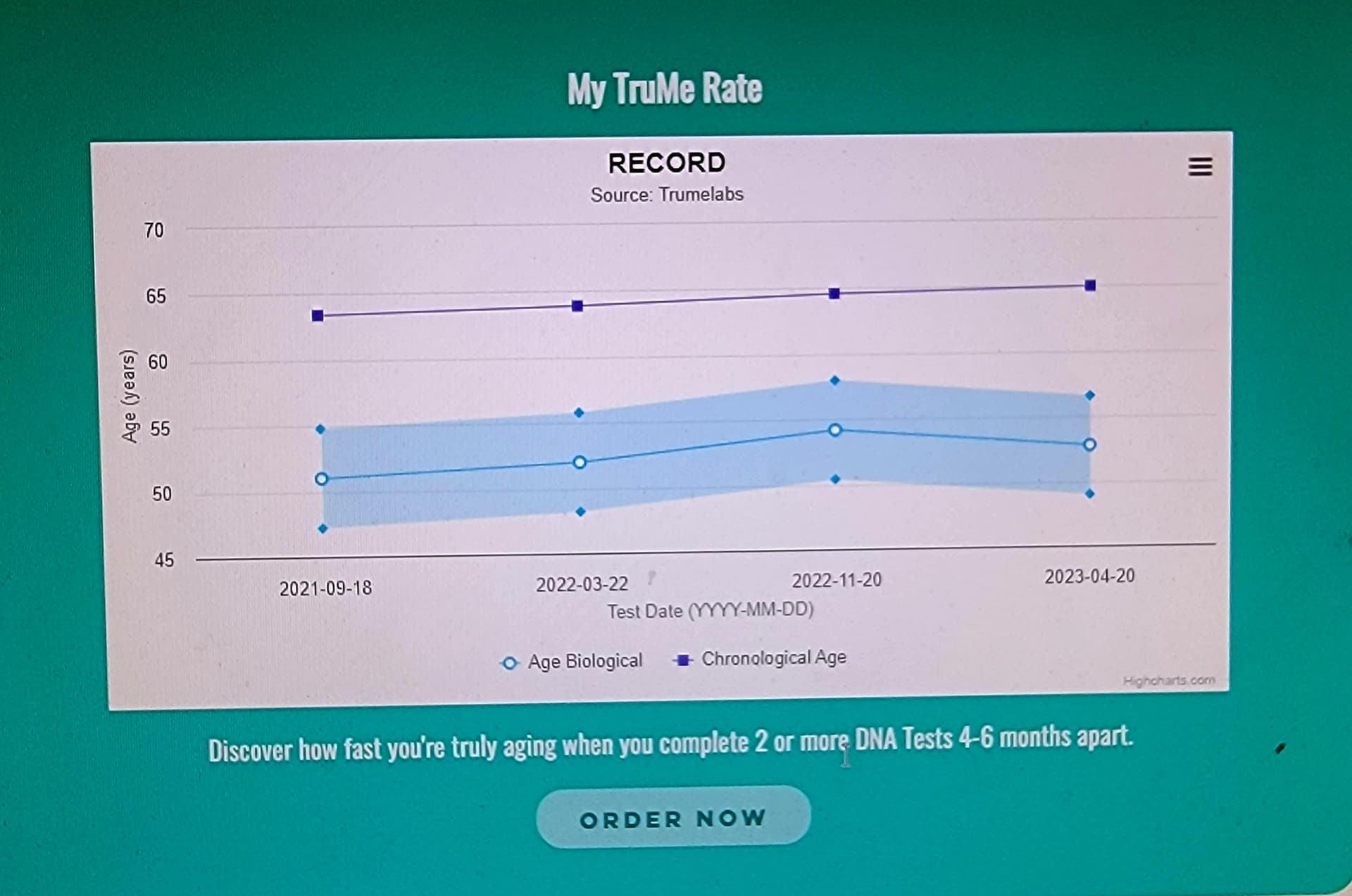
I have taken the TruMe epegentic methylation spit test 4 times. Spikes showed me aging faster during the 7-months higher rapamycin dose 36 mg every 10-days.
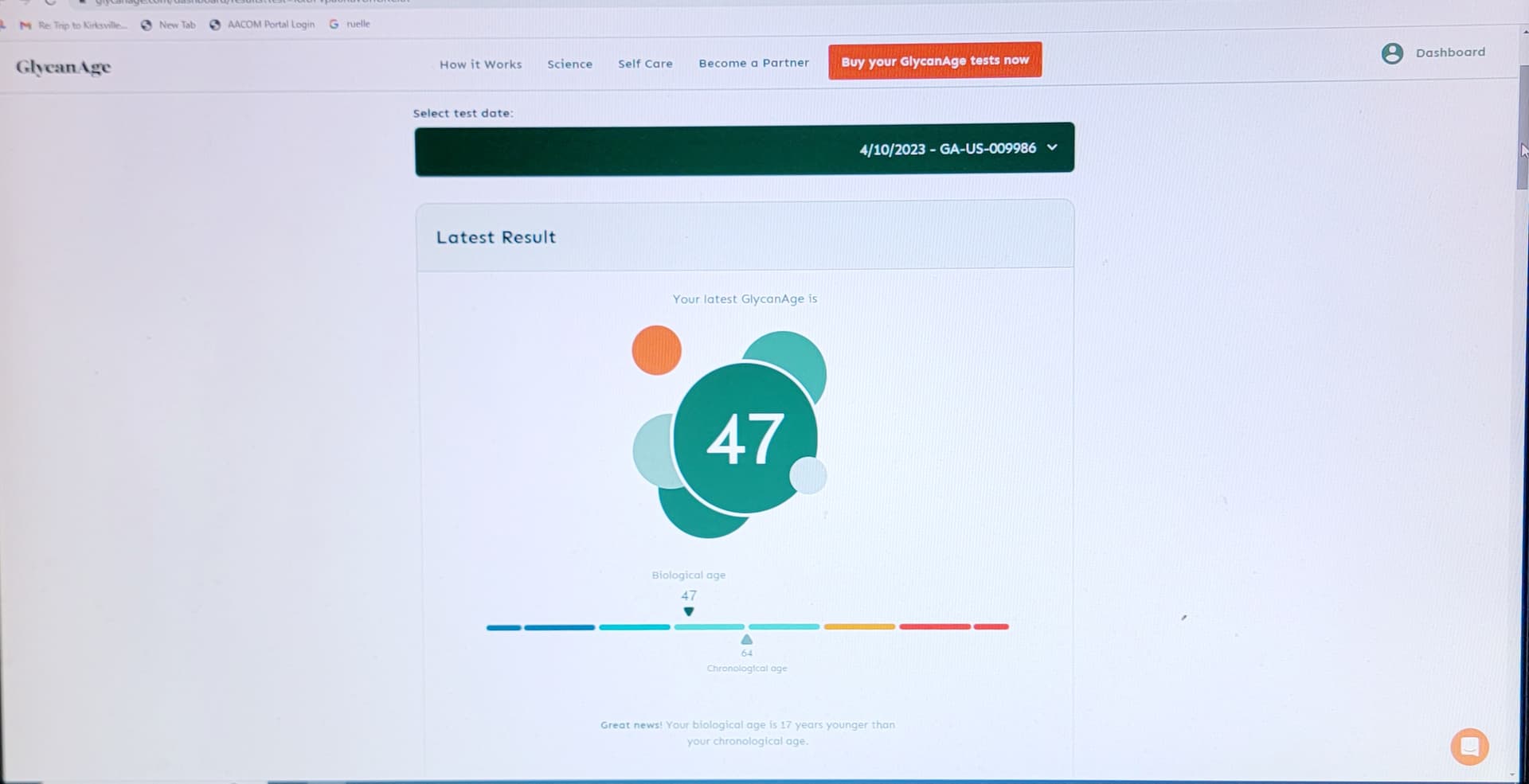
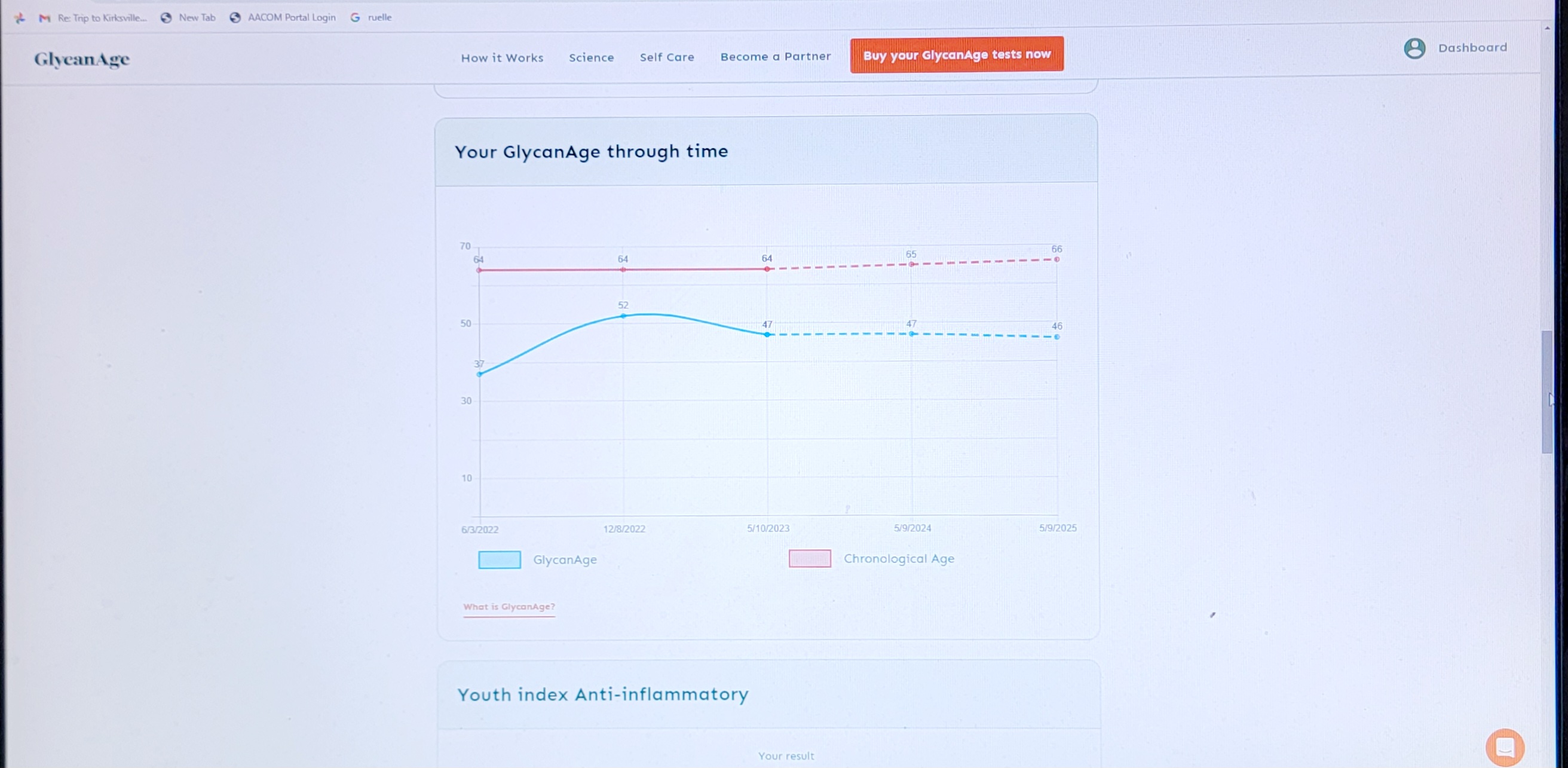
And,GlycanAge test blood - glycans on proteins. Again, a faster aging at the higher dose of 36 mg for 7 months. The two test for different biolgical changes correlate perfectly. Once I reduced back to 6 mg weekly, the biological aging reversed to younger again. Interesting - yes!
TruMe over the past 2.5 years
GlycanAge test over the past 2.5 years - I don’t think more rapamycin is better - it is too much.
Im speaking of the risk that in an intermittent dosing regime, the body will make up for the initial mtor suppression in the period when no rapamycin is taken & surge to higher than normal levels. This would presumably happen when blood levels approach their trough levels. This could in theory negate the benefits of rapa or even render it harmful.
@jimo3 & @CTStan
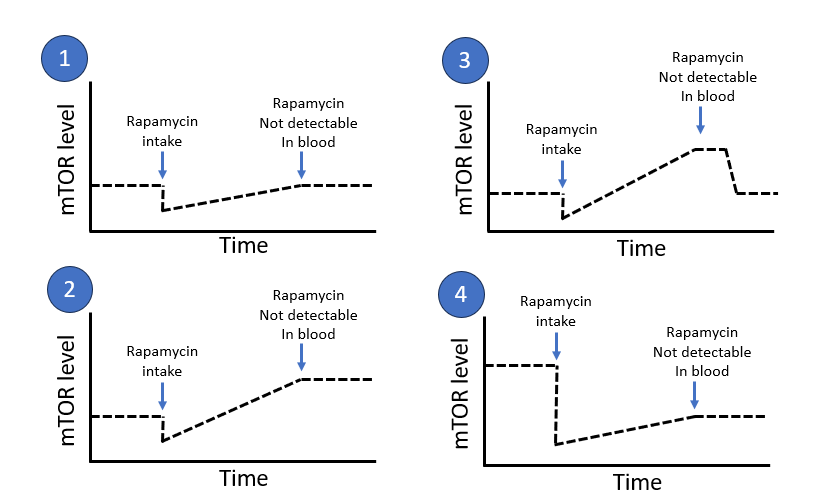
I’m starting to understand what you are pointing out but I want to understand a little bit more exact what effect we are talking about. I made a quick sketch of four possible scenarios. Which of these scenarios are we talking about.
The question needs to be addressed to Dr B (& others) as he’s the one who speculated there could be a risk from a spike. See the quote provided by CTStan above.
Total uneducated speculation but I’d suppose it would be something like 3.
I dont believe this photo is current.
In response to P.D Mangan,he replies “before disease. in Italy. I was dressed like you on your photo”
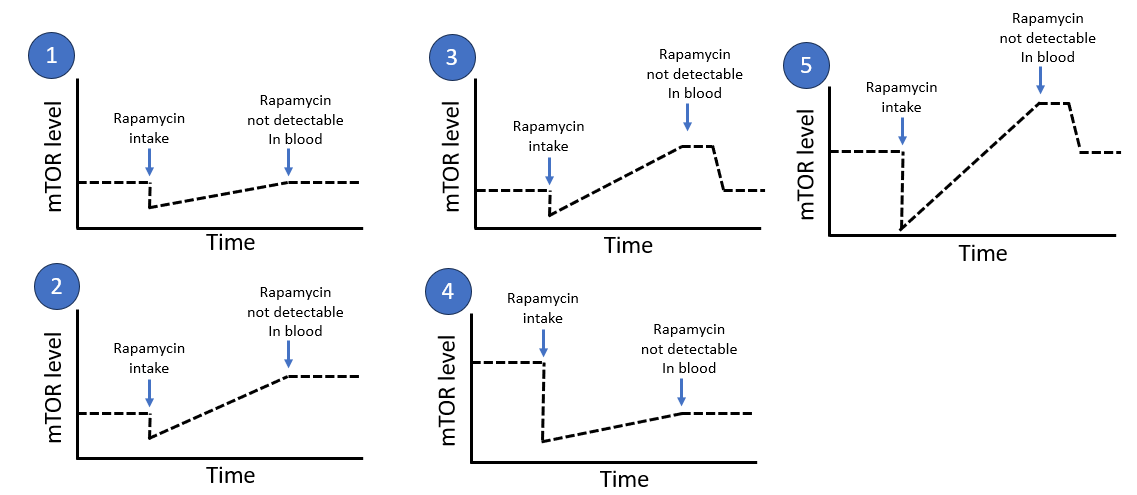
Great, now we are talking and this is the importance of specifing what we mean with the words we use because they can be interpreted in different ways ![]() I think scenario 3 is very interesting one and good aligned with some anecdotal things from others and also own experience when doing extended fasting and starting eating again. In some cases I feel that I have got some kind of anabolic peak in muscle building after a extended fast. The same thing seems also happen with glucose after a extended fast. Which is called pseudo-diabetes. Blagosklonny talks about this for example in this paper.
I think scenario 3 is very interesting one and good aligned with some anecdotal things from others and also own experience when doing extended fasting and starting eating again. In some cases I feel that I have got some kind of anabolic peak in muscle building after a extended fast. The same thing seems also happen with glucose after a extended fast. Which is called pseudo-diabetes. Blagosklonny talks about this for example in this paper.
Very interesting topic. It’s like the body gets temporary more sensitized after we have pushed our body in to a more catabolic state with low mTOR and high ampk. Like with fasting you can break a fast in a good or a bad way. Same thing is probably the case with rapamycin. Interesting!
To my knowledge the evidence is contradictory as to whether mTOR rebound after cessation of rapamycin exceeds the level before rapamycin is initiated.
The guess is that your image 2 or 3 may result.
The fear is that negative consequences could occur, including increased cell proliferation, new tumors or regrowth of existing tumors.
Since the issue was raised by Blagosklonny, he likely has an opinion on how to reduce the risk of rebound.
I think its good to add the first paragraph here to what you (@CTStan) quoted and it’s about very high intermittent doses Blagosklonny is talking about.
In theory, high intermittent dose of rapamycin (for example, 30 mg every 3 weeks) may produce a high peak level to ensure that even rapamycin-resistant cells will be targeted. (Probably, everolimus is better for this purpose because of short half-life). A high peak concentration may affect neurons, protected by the blood brain barrier, and stem cells in their niches. A high single dose of rapamycin was shown to maintain lower body weight by shifting the set point long-term in rats [25].
However, intermittent therapy may have some disadvantages. Such schedules include drug-free periods. During these periods, mTOR can be over-activated in compensation and may, in theory, cause acute harmful events. (I believe that rebound of mTOR in endothelial cells may increase thrombosis, arterial permeability and arterial spasm)
So I would like to add one more scenario 5 here which seems more aligned with what he is talking about as I interpret it. It’s about creating a deep peak but the risk is that it bumps up in the other direction (high mTOR) when it comes back and after that normalizes. It’s almost like scenario 3 but you push more in scenario 5.
For some years ago I dig a lot in the fasting topic and the same thing exists there. But the fasting community seldom talk about mTOR. They talk mostly about activating autophagy (which mTOR inhibition does). But in that community they also got very concerned about the potential cancer stimulating effect of fasting because it was not that simple that fasting only prevents cancer or improves cancer treatment. It can also make cancer grow. It’s a powerful double edge sword. Below are some quick quotes from some studies that I looked into during that time. This is a very interesting topic but I would say that this is so much bigger than just the longevity interventions fasting and Rapamycin. This double edge sword effect is connected to a lot of longevity interventions which many people use. So it’s not that easy to just stop taking Rapamycin and stop doing fasting if a person is concerned about the cancer effect. There are most likely lots of other longevity interventions to also stop doing because many are connected with activating a catabolic state with high ampk and high autophagy and low anabolism (low mtor etc).
autophagy plays a paradoxical role in tumorigenesis, depending on the stage of tumor development. Early in tumorigenesis, autophagy is a tumor suppressor via degradation of potentially oncogenic molecules. However, in advanced stages, autophagy promotes the survival of tumor cells by ameliorating stress in the microenvironment. … Autophagy is reported to stop tumor cell metastasis ([103], [104]), but other authors suggest that autophagy favors metastasis ([105], [106]).
Source:The Double-Edge Sword of Autophagy in Cancer: From Tumor Suppression to Pro-tumor Activity - PubMed
Autophagy can either cause cancer cell death or promote tumor survival during colorectal cancer.
Source:Autophagy is a double-edged sword in the therapy of colorectal cancer - PubMed
However, excessive autophagy may lead to apoptosis. Furthermore, abnormal autophagy may lead to carcinogenesis and promote tumorigenesis in normal cells. In tumor cells, autophagy may provide the energy required for excessive proliferation, promote the growth of cancer cells, and evade apoptosis caused by certain treatments, including radiotherapy and chemotherapy, resulting in increased treatment resistance and drug resistance. On the other hand, autophagy leads to an insufficient nutrient supply in cancer cells and the destruction of energy homeostasis, thereby inducing cancer cell apoptosis. Therefore, understanding the mechanism of the double-edged sword of autophagy is crucial for the treatment of cancer.
Source: Autophagy-related signaling pathways are involved in cancer (Review) - PubMed
In cancer, autophagy is highly context-specific depending on the cell type, tumour microenvironment, disease stage and external stimuli. Recently, the emerging role of autophagy as a double-edged sword in cancer has gained much attention. On one hand, autophagy suppresses malignant transformation by limiting the production of reactive oxygen species and DNA damage during tumour development. Subsequently, autophagy evolved to support the survival of cancer cells and promotes the tumourigenicity of cancer stem cells at established sites.
Autophagy can suppress or promote tumors depending on the developmental stage and tumor type, and modulating autophagy for cancer treatment is an interesting therapeutic approach currently under intense investigation. Nutritional restriction is a promising protocol to modulate autophagy and enhance the efficacy of anticancer therapies while protecting normal cells.
Source: Autophagy and intermittent fasting: the connection for cancer therapy? - PubMed
The modulation of autophagy plays dual roles in tumor suppression and promotion in many cancers. In addition, autophagy regulates the properties of cancer stem-cells by contributing to the maintenance of stemness, the induction of recurrence, and the development of resistance to anticancer reagents. Although some autophagy modulators, such as rapamycin and chloroquine, are used to regulate autophagy in anticancer therapy, since this process also plays roles in both tumor suppression and promotion, the precise mechanism of autophagy in cancer requires further study. In this review, we will summarize the mechanism of autophagy under stressful conditions and its roles in tumor suppression and promotion in cancer and in cancer stem-cells.
have any gender related studies been done? Any data on safety in women v men? Dosing in women v men (or other genders!)?
You could ask him if other mTOR suppressors such as TRF/intermittent fasting or supplements known to suppress mTor would mitigate the possible rebound effect.
Also, ask him if there is any actual proof the excessive rebound actually occurs.
I am not too worried about mTOR rebound. Rapamycin is not the only thing that suppresses mTor. Intermittent fasting, TRF and some supplements also suppress mTOR.
A quote from a Peter Attia podcast:" The only two interventions that really seem to offer longevity benefits are rapamycin supplementation and some degree of fasting
They both suppress mTOR (mechanistic target of rapamycin) and in turn increase autophagy
“Increasing studies have demonstrated that some diet-derived natural products, including curcumin, resveratrol, epigallocatechin gallate (EGCG), genistein, 3, 3-diindolylmethane (DIM) and caffeine, may inhibit mTOR signaling directly or indirectly (Table 1) [140-147].”
“TRE also works to naturally suppress the Mtor pathway. Not to the extent of intermittent fasting, but enough to noticeably turn the dial, if practiced routinely, ideally at a minimum 16/8 split (or one more stringent, such as an 18/6).”
“In contrast, the mechanistic target of rapamycin (mTOR), a nutrient-activated serine/threonine protein kinase, is activated during the fed state”
“It was shown mainly in vitro that IF directly improves cellular stress adaptation (mainly against ROS generation), decrease inflammation and DNA damage, decrease mTOR expression [31], and promote autophagy [34] which is similar to the effects of Rapamycin in some animal models”
“Inhibition of mTOR, Insulin, and IGF-1during IF may promote the growth and self-renewal of ISCs with repair of the epithelial damage.”
“Molecular theoretical models show potential beneficial effects of IF on the interruption of SARS-CoV-2 life cycle through inhibition of mTOR and promotion of autophagy”
“Updates of mTOR inhibitors - PMC)%20%5B140%2D147%5D.”
“Health Hack #28 - Time Restricted Eating | Real Food Eatery”
“Time-Restricted Eating: Benefits, Mechanisms, and Challenges in Translation - PMC(TRE%2C,calorie%20intake%20or%20diet%20quality.”
@Jennifer_Sudarsky1 & @desertshores
Sorry, I had the interview with him yesterday so I missed this one.
If I remember it correctly we touched a little bit about the effect of minimizing the rebound effect (@desertshores)
Cannot wait to see your interview! There was an interesting post today on Twitter re fasting. Would be nice to have interpretation of it.
Please ask him if his recommendations on the dose and frequency or anything else regarding rapamycin are different for people that have mild insulin resistance before starting rapamycin, and if so why?
When you are ready to release your interview with Mikhail Blagosklonny, would it be possible to also reference and point us to highlights in the interview?
I realize that the time and effort and preparation you have already put into these interviews is more than anyone might imagine. I understand if this may be too much to ask.
I dont want to miss a single word of your questions and what he has to say. But one of the side effects of old age, at least in my case, is the extreme difficulty I have in comprehending accents that differ from my own.
What does he think of the new paper on taurine?
There was study and posting recently showing arcabose is deactivated in oral and microbiota. Anyone remember that?