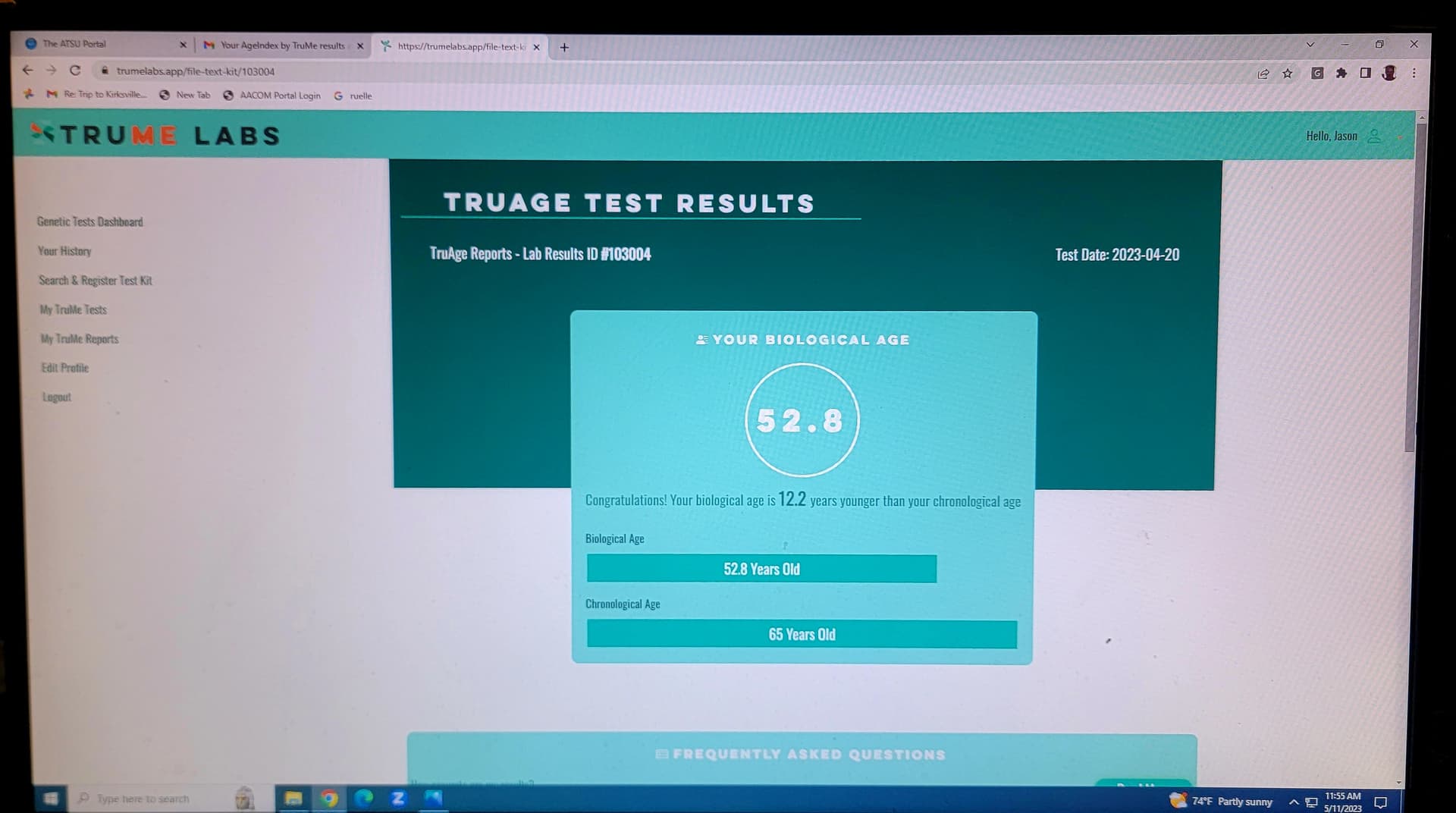
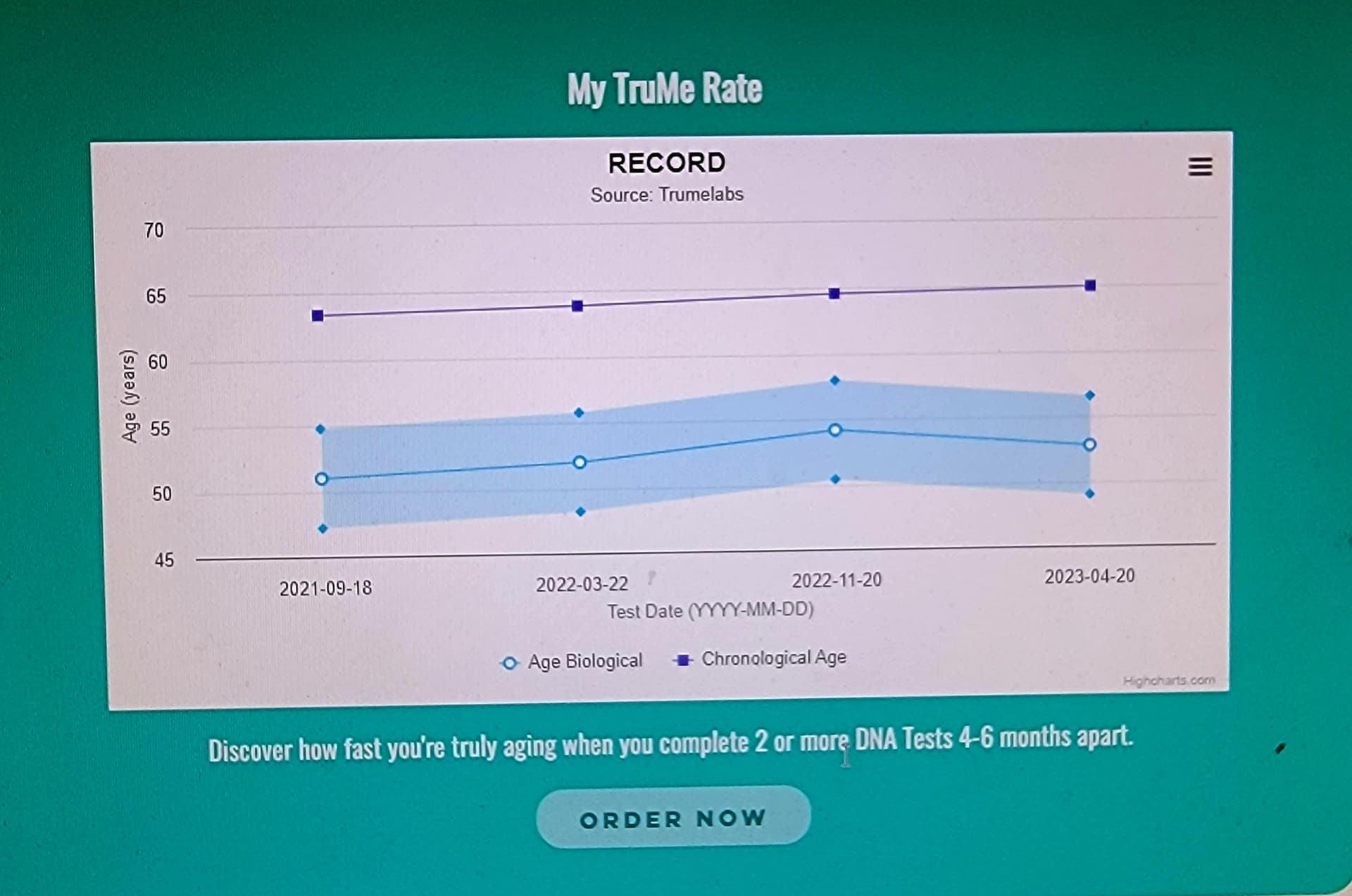
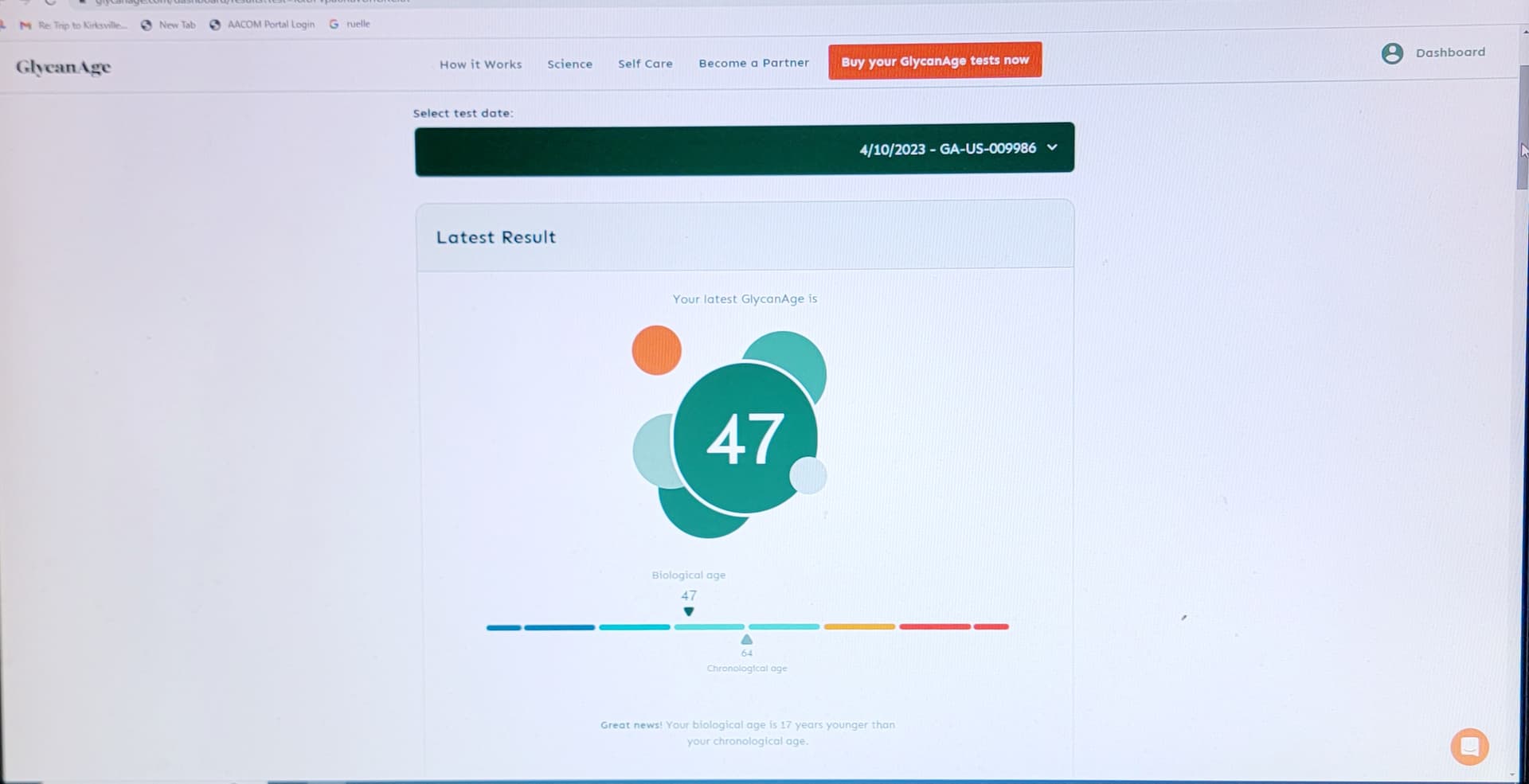
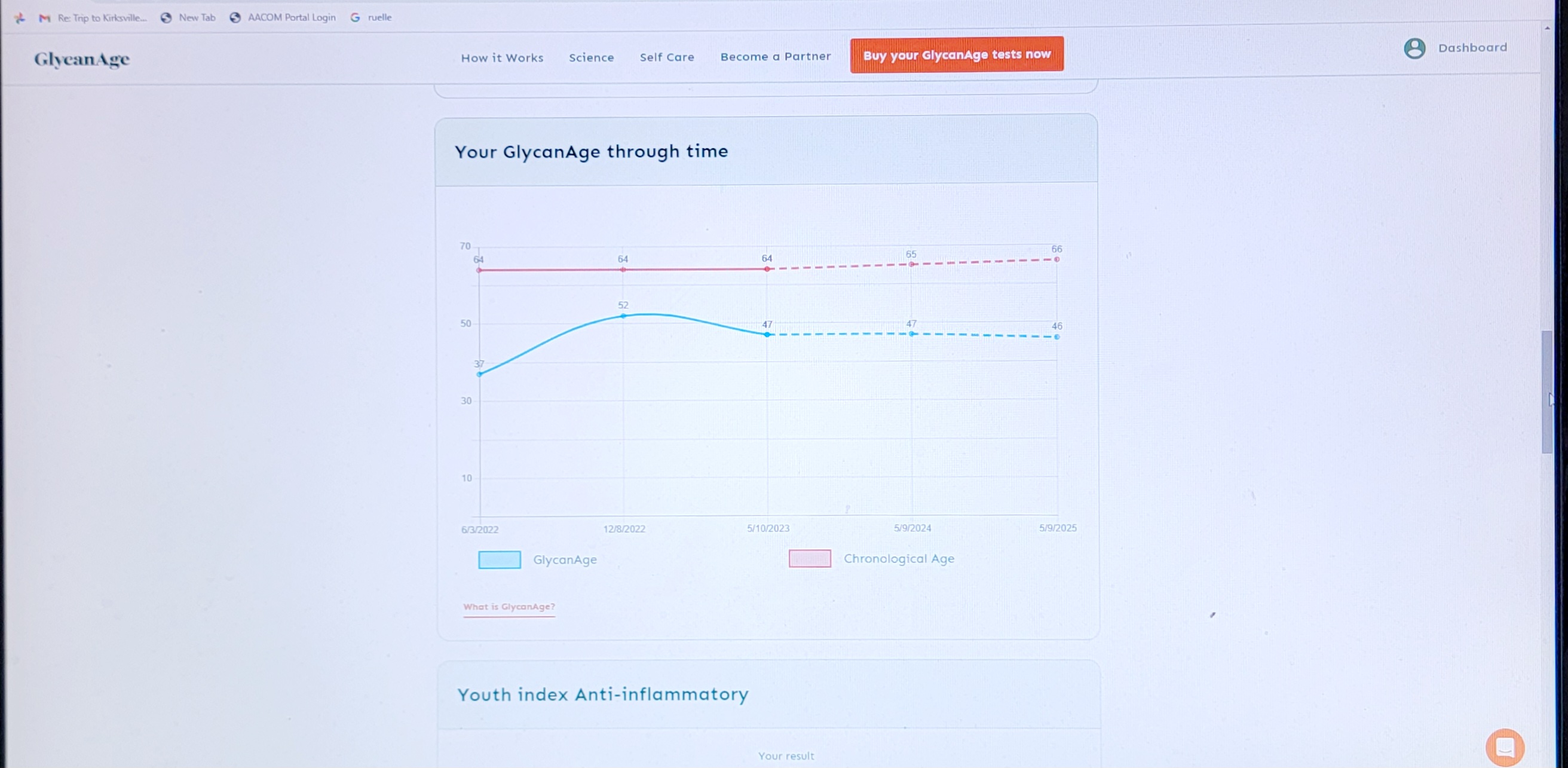
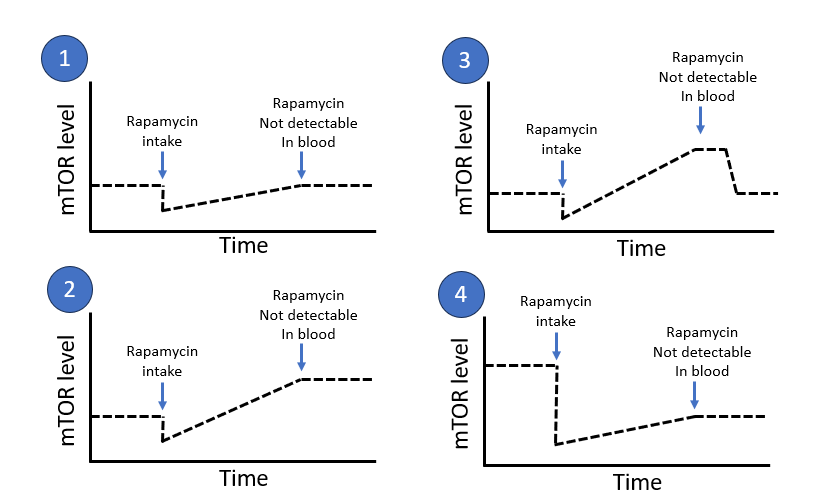
I think its good to add the first paragraph here to what you (@CTStan) quoted and it’s about very high intermittent doses Blagosklonny is talking about.
In theory, high intermittent dose of rapamycin (for example, 30 mg every 3 weeks) may produce a high peak level to ensure that even rapamycin-resistant cells will be targeted. (Probably, everolimus is better for this purpose because of short half-life). A high peak concentration may affect neurons, protected by the blood brain barrier, and stem cells in their niches. A high single dose of rapamycin was shown to maintain lower body weight by shifting the set point long-term in rats [25].
However, intermittent therapy may have some disadvantages. Such schedules include drug-free periods. During these periods, mTOR can be over-activated in compensation and may, in theory, cause acute harmful events. (I believe that rebound of mTOR in endothelial cells may increase thrombosis, arterial permeability and arterial spasm)
Source: Doses and schedules of rapamycin for longevity: does aging exist or only age-related diseases? - Dr. Blagosklonny, M.D., Ph.D.
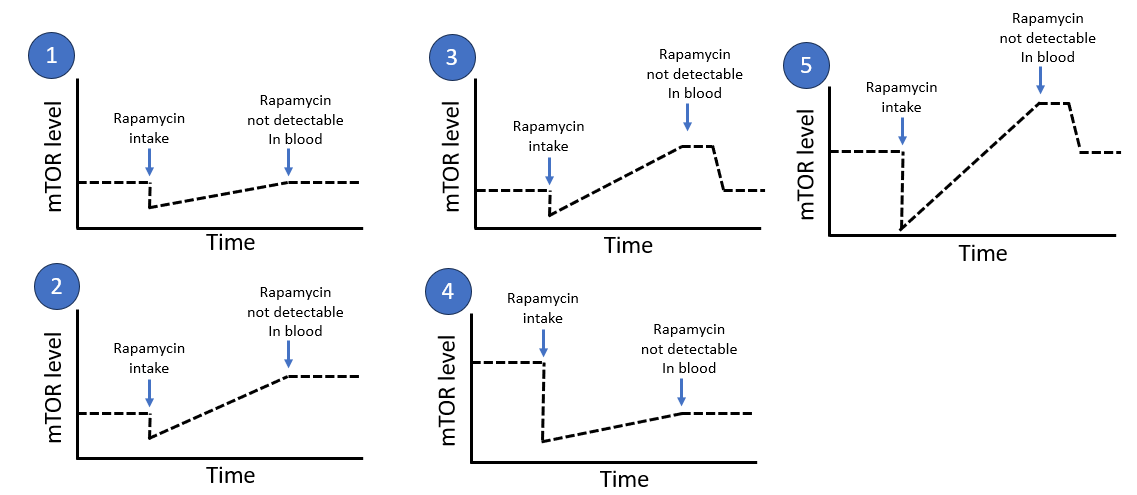
So I would like to add one more scenario 5 here which seems more aligned with what he is talking about as I interpret it. It’s about creating a deep peak but the risk is that it bumps up in the other direction (high mTOR) when it comes back and after that normalizes. It’s almost like scenario 3 but you push more in scenario 5.
For some years ago I dig a lot in the fasting topic and the same thing exists there. But the fasting community seldom talk about mTOR. They talk mostly about activating autophagy (which mTOR inhibition does). But in that community they also got very concerned about the potential cancer stimulating effect of fasting because it was not that simple that fasting only prevents cancer or improves cancer treatment. It can also make cancer grow. It’s a powerful double edge sword. Below are some quick quotes from some studies that I looked into during that time. This is a very interesting topic but I would say that this is so much bigger than just the longevity interventions fasting and Rapamycin. This double edge sword effect is connected to a lot of longevity interventions which many people use. So it’s not that easy to just stop taking Rapamycin and stop doing fasting if a person is concerned about the cancer effect. There are most likely lots of other longevity interventions to also stop doing because many are connected with activating a catabolic state with high ampk and high autophagy and low anabolism (low mtor etc).
autophagy plays a paradoxical role in tumorigenesis, depending on the stage of tumor development. Early in tumorigenesis, autophagy is a tumor suppressor via degradation of potentially oncogenic molecules. However, in advanced stages, autophagy promotes the survival of tumor cells by ameliorating stress in the microenvironment. … Autophagy is reported to stop tumor cell metastasis ([103], [104]), but other authors suggest that autophagy favors metastasis ([105], [106]).
Source:The Double-Edge Sword of Autophagy in Cancer: From Tumor Suppression to Pro-tumor Activity - PubMed
Autophagy can either cause cancer cell death or promote tumor survival during colorectal cancer.
Source:Autophagy is a double-edged sword in the therapy of colorectal cancer - PubMed
However, excessive autophagy may lead to apoptosis. Furthermore, abnormal autophagy may lead to carcinogenesis and promote tumorigenesis in normal cells. In tumor cells, autophagy may provide the energy required for excessive proliferation, promote the growth of cancer cells, and evade apoptosis caused by certain treatments, including radiotherapy and chemotherapy, resulting in increased treatment resistance and drug resistance. On the other hand, autophagy leads to an insufficient nutrient supply in cancer cells and the destruction of energy homeostasis, thereby inducing cancer cell apoptosis. Therefore, understanding the mechanism of the double-edged sword of autophagy is crucial for the treatment of cancer.
Source: Autophagy-related signaling pathways are involved in cancer (Review) - PubMed
In cancer, autophagy is highly context-specific depending on the cell type, tumour microenvironment, disease stage and external stimuli. Recently, the emerging role of autophagy as a double-edged sword in cancer has gained much attention. On one hand, autophagy suppresses malignant transformation by limiting the production of reactive oxygen species and DNA damage during tumour development. Subsequently, autophagy evolved to support the survival of cancer cells and promotes the tumourigenicity of cancer stem cells at established sites.
Source: Is targeting autophagy mechanism in cancer a good approach? The possible double-edge sword effect - PubMed
Autophagy can suppress or promote tumors depending on the developmental stage and tumor type, and modulating autophagy for cancer treatment is an interesting therapeutic approach currently under intense investigation. Nutritional restriction is a promising protocol to modulate autophagy and enhance the efficacy of anticancer therapies while protecting normal cells.
Source: Autophagy and intermittent fasting: the connection for cancer therapy? - PubMed
The modulation of autophagy plays dual roles in tumor suppression and promotion in many cancers. In addition, autophagy regulates the properties of cancer stem-cells by contributing to the maintenance of stemness, the induction of recurrence, and the development of resistance to anticancer reagents. Although some autophagy modulators, such as rapamycin and chloroquine, are used to regulate autophagy in anticancer therapy, since this process also plays roles in both tumor suppression and promotion, the precise mechanism of autophagy in cancer requires further study. In this review, we will summarize the mechanism of autophagy under stressful conditions and its roles in tumor suppression and promotion in cancer and in cancer stem-cells.
Source: The Roles of Autophagy in Cancer - PubMed
![]()
![]()