The full text link doesn’t work for me, but I can see the date of the paper and it says 1996?
You need to let it load for a while. That website has these studies hosted on a very slow server.
But I will upload it here for now:
TREATMENT OF SKIN AGING WITH TOPICAL ESTROGENS – JOLANTA B_ SCHMIDT; MARTINA BINDER; GABRIELE DEMSCHIK; – International Journal of Dermatology, #9, – 10_1111_j_1365-4362_1996_tb03701_x – 4458c31f904775bcf58e79.pdf (5.2 MB)
I have extreme ongoing elastin production at age 64, it seems. Passed every relevant test I found via Grok-AI for it. Any questions you might have for me?
Yea what is your diet, lifestyle, exercise, supplement, drug protocol
I’m still interested in diet, lifestyle, exercise, supplement and drug protocol if you get this message. Also curious if you have any particular health issues/diseases/illnesses that might be unknowingly causing some kind of elastin generating effect.
Still interested if you see this message.
Since elastin is critical for a healthy old age and there is currently no way to restore elastin in a systemic way, what we are currently left with is still a good thing. Fixing or improving CVD, Cancer, Stroke, COPD, etc. outcomes are quite possible in the next couple of decades.
While fixing the top 4 causes of death in the aged is not going to increase maximal life span, I’m still hoping to be on that train ![]()
Fixing the elastin problem is significantly harder than any of those.
Which is why there is so little research in this area. Even the great AdG is not working on this. Why not? it is the hardest thing to fix compared to the 4 major killers related to aging.
No fix for the elastin problem on the horizon.pdf (290.2 KB)
I think this is a splicing thing.
Damage accumulation in long-living macromolecules (especially extracellular matrix (ECM) proteins, nuclear pore complex (NPC) proteins, and histones) is a missing hallmark of aging. Stochastic non-enzymatic modifications of ECM trigger cellular senescence as well as many other hallmarks of aging affect organ barriers integrity and drive tissue fibrosis. The importance of it for aging makes it a key target for interventions. The most promising of them can be AGE inhibitors (chelators, O-acetyl group or transglycating activity compounds, amadorins and amadoriases), glucosepane breakers, stimulators of elastogenesis, and RAGE antagonists.
https://advanced.onlinelibrary.wiley.com/doi/10.1002/adhm.202400484
I’ve gone through the very latest studies on elastin, picked the most relevant for what we are interested in and summarized why each of these studies are important. Here is what I’ve found:
Why this seems important: Afzelin is a potential collagen type 1 and elastin restoring compound.
“Results: Afzelin restored UVA-impaired cell viability and reduced β-galactosidase, p53, and p21 while recovering Lamin B1. It lowered ROS levels and restored mitochondrial membrane potential (2.8-fold) via AMPK-AKT/mTOR-ULK1 and PINK1-Parkin activation. Combined with GAA (50 mM), afzelin showed strong synergy (Bliss = 67.6 ± 5.1). In vivo, co-treatment reduced epidermal thickness ( ∼37.3 %), restored collagen I and elastin, and suppressed p53/p21 expression.
Conclusion: Afzelin alleviates UVA-induced photodamage by activating autophagy and mitophagy. Together with the anti-aging triterpenoid GAA, it exerts synergistic anti-photoaging effects, supporting its potential as a natural autophagy-targeting agent for skin rejuvenation.”
Afzelin resists UVA damage through autophagy and synergizes with ganoderic acid A to skin photoaging Afzelin resists UVA damage through autophagy and synergizes with ganoderic acid A to skin photoaging - PubMed
Why this seems important: Potentially important genes for rejuvenating elastin which could be explored in genetic reprogramming or other methods of targeting.
“The analysis revealed five protein-coding genes significantly associated with collagen III (P value < 2.44E-6, R2 > 0.5). Two protein-coding genes, including collagen III, were positively correlated with fibronectin, and five protein-coding genes were positively correlated with elastin.”
Transcriptome-wide analysis of genes associated with collagen III in human skin Transcriptome-wide analysis of genes associated with collagen III in human skin - PubMed
Why this seems important: By analysing mechanisms of elastin downregulation in diseased states we might uncover insights in how to recover elastin production or halt degredation/downregulation in all humans.
“In the diagnosed group, proteins such as MMP7, POSTN, and CD163 exhibited characteristic high-level expression. Furthermore, compared with the suspected group, the diagnosed group showed significant downregulation of proteins related to elastin fibers, indicating a more severe degree of fibrosis in the lacrimal gland tissues.”
Autoimmune fibroinflammation in IgG4-related ophthalmic disease: TLR8-dependent signaling pathways and fibrotic remodeling revealed by proteomic profiling Autoimmune fibroinflammation in IgG4-related ophthalmic disease: TLR8-dependent signaling pathways and fibrotic remodeling revealed by proteomic profiling - PubMed
Why this seems important: Synthetic elastic protein combined with ascorbic acid appears to improve elastic fibre formation.
“…imbalances in the components of the extracellular matrix (ECM) during tissue engineering often result in the deterioration of the mechanical properties and physiological relevance of dermal substitutes, in part due to the detrimental effect of ascorbic acid (AA) on elastic fibre biosynthesis. The objective of this study is to investigate the potential of a synthetic elastic protein (SEP) to improve ECM remodelling and to restore the equilibrium between collagen and elastin in reconstructed dermal tissues. Primary fibroblasts monolayers were treated with increasing concentrations of SEP in the presence or not of AA without affecting cytotoxicity. Western blot and immunofluorescence analyses showed that in the presence of AA - which typically reduced elastin synthesis- SEP improved elastic fibre formation without affecting type I collagen assembly.”
A Recombinant Elastic Peptide Rescues Elasticity From a Self-Assembled Dermal Sheet Model Treated With Ascorbic Acid A Recombinant Elastic Peptide Rescues Elasticity From a Self-Assembled Dermal Sheet Model Treated With Ascorbic Acid - PubMed
Why this seems important: Diabetes downregulates extracellular matrix remodeling genes and impairs elastin deposition after injury, suggesting that chronic inflammatory and metabolic signaling suppresses elastogenesis.
“…T1D and T2D mice displayed upregulation of genes associated with canonical chemokine/monocyte-mediated inflammatory pathways, and downregulation of genes associated with extracellular matrix remodeling… T1D and T2D delayed activation of M2-like (CD206+) macrophages in the heart, and impaired normal collagen and elastin deposition after MI.”
Distinct immunometabolic signatures of type 1 versus type 2 diabetes in a murine model of myocardial infarction Distinct immunometabolic signatures of type 1 versus type 2 diabetes in a murine model of myocardial infarction - PubMed
Why this seems important: Estrogen, and the agonism of various receptors appears to cause, among other seemingly good things, elastin synthesis.
“This review examines the mechanisms through which estrogen, acting via nuclear receptors (ERα and ERβ) and the membrane receptor G protein-coupled estrogen receptor (GPER), regulates pelvic floor connective tissue homeostasis through both genomic and non-genomic pathways. Key regulatory effects include the promotion of collagen and elastin synthesis, inhibition of matrix metalloproteinase (MMP) activity, modulation of fibroblast function-including mitigation of cellular senescence and enhancement of migratory and anti-apoptotic capacities-as well as integration with mechanical signaling through the integrin-YAP/TAZ axis.”
Multidimensional regulation of estrogen signaling in pelvic floor connective tissue homeostasis and remodeling Multidimensional regulation of estrogen signaling in pelvic floor connective tissue homeostasis and remodeling - PubMed
Why this seems important: Supraperiosteal implantation of polycaprolactone (PCL) microspheres appears to significantly promote elastin fiber formation.
“H&E and Masson’s trichrome staining revealed significantly enhanced collagen ingrowth in PCL microspheres compared to HA. Immunofluorescence staining showed substantial infiltration of type I and type III collagen within the PCL microspheres, with a predominance of type I collagen. EVG staining indicated that PCL microspheres significantly promoted elastin fiber formation.”
A Rat Model Investigation of Enhanced Facial Rejuvenation via PCL Microsphere-Induced Superior Collagen Neogenesis in the Supraperiosteal Plane A Rat Model Investigation of Enhanced Facial Rejuvenation via PCL Microsphere-Induced Superior Collagen Neogenesis in the Supraperiosteal Plane - PubMed
Why this seems important: Activation of the STING inflammatory pathway drives elastin disruption in abdominal aortic aneurysm, and its inhibition preserves elastin structure, suggesting STING signalling may be a key regulator of elastin degradation in inflammatory vascular aging.
“The STING signaling pathway was significantly activated in AAA tissues… Sting mutation slowed AAA formation, as confirmed by reduced AAA incidence, maximal abdominal aortic diameter, elastin disruption, collagen deposition, and inhibited immune cell infiltration…”
Inhibition of STING pathway attenuates experimental abdominal aortic aneurysm progression Inhibition of STING pathway attenuates experimental abdominal aortic aneurysm progression - PubMed
Why this seems important: Eggshell membrane appears to have some sort of skin structure regenerative property, seemingly related to collagen and elastin formation. Potentially biased industry funded study but worth further investigation.
“This study demonstrates that a 300 mg intake of fresh membrane MKARE® positively affects mechanical skin parameters compared to the group that consumed 8,000 mg of hydrolyzed collagen or the placebo group. These improvements suggest that MKARE® consumption regenerates the skin structure, primarily due to positive changes in collagen and elastin formation, which are key contributors to the improvement of skin firmness, elasticity, and barrier protection.”
Comparative effects of MKARE® eggshell membrane and hydrolyzed collagen as nutricosmetics on skin biophysical properties: a randomized clinical trial Comparative effects of MKARE® eggshell membrane and hydrolyzed collagen as nutricosmetics on skin biophysical properties: a randomized clinical trial - PubMed
Why this seems important: Vitamin B6 decreased elastin degradation. While I don’t think many people have vitamin B6 deficiencies and I don’t think vitamin B6 megadosing will solve elastin degradation, we may be able to create novel B6 like compounds or figure out what B6 is doing in the body to figure out new targets for elastin protection and/or regeneration.
“B6 supplementation significantly attenuated AD, as evidenced by reduced AD incidence, improved survival, preservation of aortic architecture, and decreased elastin degradation. B6 also exerted antioxidant effects, alleviating oxidative stress, which was associated with inhibition of smooth muscle cell phenotypic switching and reduced ECM degradation.”
Vitamin B6 attenuates aortic dissection by inhibiting pathological remodeling of the aortic extracellular matrix Vitamin B6 attenuates aortic dissection by inhibiting pathological remodeling of the aortic extracellular matrix - PubMed
Why this seems important: We already know that retinoids can have a positive effect on elastin production, but they are often irritating. Hydroxypinacolone 9-cis retinoate is a novel retinoid that may be able to be applied more often without irritation offering a more robust elastin and collagen rejuvenating effect for more effective aesthetic antiaging effect in the skin.
“Photoaging, driven by chronic ultraviolet radiation (UVR), disrupts skin structure and function. Traditional retinoids enhance extracellular matrix (ECM) regeneration but cause irritation. Hydroxypinacolone 9-cis retinoate (9-cis HPR), a derivative of 9-cis retinoic acid, selectively activates RARα and RXRα, improving efficacy and tolerability. In a UVR-induced SKH-1 mouse photoaging model, 9-cis HPR reduced erythema, desquamation, and loss of elasticity while promoting collagen and elastin production. Single-cell RNA sequencing and spatial transcriptomics revealed restoration of fibroblast, basal cell, and melanocyte proportions, suppression of myofibroblast differentiation, and upregulation of ECM-related genes (e.g., Col1a2, Col3a1, Elastin). Additionally, 9-cis HPR inhibited melanogenesis by downregulating melanogenesis-related genes (Tyr, Dct, Tyrp1), melanosome biogenesis genes (Mlana, Pmel), and the melanocyte proliferation gene Kit, likely via ROS suppression. Cell-cell interaction analysis showed that 9-cis HPR promoted fibroblast-driven repair via NPY-NPY1R, PTN-SDC2, and POSTN-ITGA/BV signaling, while inhibiting KITL-KIT-mediated melanocyte proliferation. In a single-blind, split-face clinical trial involving 31 Chinese women, 0.03% 9-cis HPR applied daily for 4 weeks demonstrated comparable or superior improvements in wrinkles, elasticity, hydration, dermal density, and radiance versus 0.3% retinol, without observed irritation. These findings support 9-cis HPR as a safe and effective retinoid that mitigates photoaging through ECM restoration, inflammation modulation, and pigmentation control.”
Hydroxypinacolone 9-cis Retinoate Mitigates UV-Induced Photoaging by Modulating ECM, Fibroblasts, Inflammation, and Melanogenesis Hydroxypinacolone 9-cis Retinoate Mitigates UV-Induced Photoaging by Modulating ECM, Fibroblasts, Inflammation, and Melanogenesis - PubMed
Why this seems important: The use of hydroxytyrosol loaded soluble microneedles upregulated elastin expression alongside a range of beneficial effects to reduce photoaging. This seems like a truly amazing treatment!
“Western blot analysis further revealed that HT MNs upregulated the expression of collagen type I alpha 1 (COL1A1), elastin (ELN), hyaluronan synthase 2 (HAS2), and filaggrin (FLG), while downregulating matrix metalloproteinase 1. Overall, these findings suggest that HT MNs effectively mitigate UV-induced photoaging through antioxidant, anti-senescence, and extracellular matrix (ECM)-regulating mechanisms, underscoring their potential as a novel transdermal anti-photoaging therapy.”
Anti-Photoaging Effect of Soluble Microneedles Loaded with Hydroxytyrosol Anti-Photoaging Effect of Soluble Microneedles Loaded with Hydroxytyrosol - PubMed
Why this seems important: This dual hyaluronic acid compound was shown to increase elastin and collagen production alongside numerous other beneficial effects promoting skin rejuvenation.
“This study investigated the multimodal rejuvenation potential of a dual hyaluronic acid compound (DHC), composed of low- and high-molecular-weight HA integrated within a minimally cross-linked hybrid complex. In vitro assays using dermal fibroblasts, melanoma cells, and macrophages demonstrated that DHC enhanced fibroblast viability, collagen I/III and elastin production, antioxidant enzyme activity, and wound-healing capacity while reducing senescence markers.”
Regenerative Skin Remodeling by a Dual Hyaluronic Acid Hybrid Complex in Multimodal Preclinical Models Regenerative Skin Remodeling by a Dual Hyaluronic Acid Hybrid Complex in Multimodal Preclinical Models - PubMed
Why this seems important: Tirzepatide, a GLP1/GIP receptor agonist, appears to have powerful anti-inflammatory effects. Part of this appears to protect elastin integrity. I also hypothesize that tirzepatide and this class of drugs prevent some damage from glycation which would also protect elastin.
“This study offers initial experimental evidence that tirzepatide (TZP), a novel dual GIP and GLP-1 receptor agonist, significantly reduces formation and rupture, maintains medial elastin integrity, and inhibits VSMC phenotype switching and vascular inflammation in a BAPN-induced mouse model of TAAD. These protective effects are partially mediated by inhibiting NLRP3 inflammasome activation, as indicated by decreased expression of NLRP3, Caspase-1, and pro-inflammatory cytokines like IL-1β.”
Tirzepatide mitigates thoracic aortic aneurysm and dissection by alleviating the loss of the contractile phenotype in vascular smooth muscle cells and reducing vascular inflammation https://www.sciencedirect.com/science/article/abs/pii/S1537189126000017
Why this seems important: Silk fibroin restores elastin deposition by reversing fibroblast senescence and suppressing ROS–MAPK–AP-1–MMP signaling, identifying cellular aging and oxidative stress as upstream regulators of elastic fiber loss.
“Skin aging is characterized by a progressive decline in regenerative capacity, primarily driven by fibroblast senescence, oxidative stress, chronic inflammation, and the degradation of type I/III collagen, culminating in an extracellular matrix (ECM) imbalance. Current injectable fillers-such as hyaluronic acid, collagen, and PLLA-provide temporary structural support but fail to address the underlying cellular senescence or restore ECM homeostasis, highlighting the need for regenerative biomaterials. Silk fibroin (SF), a natural protein, self-assembles into a β-sheet-rich scaffold that structurally supports fibroblasts in depositing collagen and elastin, thereby improving the skin’s ECM, accelerating wound healing, and promoting tissue regeneration. However, its role in modulating fibroblast senescence and ECM remodeling remains unclear. This study demonstrates that SF provides a suitable microenvironment for the adhesion and proliferation of fibroblasts, reducing the accumulation of SASP factors and facilitating the transition of fibroblasts from a senescent to a functional state. Furthermore, SF improves the skin microenvironment by reducing reactive oxygen species (ROS) and matrix metalloproteinase (MMP) expression through modulation of the ROS-MAPK-AP-1-MMP signal pathway, thereby delaying collagen degradation in aged skin. These findings reveal that SF uniquely rejuvenates fibroblasts and restores ECM homeostasis through a non-inflammatory mechanism, distinguishing it from conventional fillers that rely on inflammatory pathways for collagen induction. This work establishes SF as a next-generation injectable biomaterial with dual targeting of cellular senescence and ECM imbalance, offering a transformative strategy for regenerative dermatology and personalized anti-aging approaches.”
Silk Fibroin Counteracts Fibroblast Senescence to Restore ECM Homeostasis in Aged Skin Silk Fibroin Counteracts Fibroblast Senescence to Restore ECM Homeostasis in Aged Skin - PubMed
Why this seems important: IL-16 appears to be incredibly important not just for preventing the degradation of elastin and collagen but for a wide variety of vital health functions for longevity.
“IL-16 deficiency increased the necrotic core and reduced fibrous cap thickness in the plaques. IL-16 deletion accelerated the degradation of intraplaque collagen and elastin, increased matrixmetalloproteinase activity, and reduced TIMP-3 expression. Transplantation of wild-type IL-16 bone marrow into IL-16 knockout mice successfully attenuated the plaque instability caused by IL16 deficiency.”
Interleukin-16 upregulates tissue inhibitor of metalloproteinase 3 to promote atherosclerotic plaque stability Interleukin-16 upregulates tissue inhibitor of metalloproteinase 3 to promote atherosclerotic plaque stability - PubMed
A NEW THEORY OF HOW WE MAY FIX THE ELASTIN PROBLEM:
Tropoelastin Coacervation as the First Step in Elastic Fiber Assembly
The soluble precursor to elastin, tropoelastin, has an intrinsic ability to self-associate into dense droplets through coacervation. This process is driven by the cooperative interactions of hydrophobic domains within the protein, where exposure of these regions to water leads to an entropically favorable clustering as water molecules are released, lowering the system’s free energy. External factors such as protein concentration, ionic strength, pH, and temperature influence the propensity of tropoelastin to undergo this phase separation, which is essential for organizing monomers into multimers prior to crosslinking and fiber formation. Coacervation is therefore considered an endothermic, entropically driven phenomenon that arranges the molecules for subsequent crosslinking by lysyl oxidase.
Source: Coacervation of tropoelastin - PubMed
How Coacervation Could Be Guided for Longevity and Aesthetic Elastin Regenerative Purposes
From a regenerative or anti-aging perspective, the coacervation step represents a control point in extracellular matrix assembly. In vivo, coacervates align on cell surfaces and may interact with components like glycosaminoglycans and microfibrillar proteins that modulate droplet behavior and fusion, suggesting that cellular and matrix factors can influence coacervation dynamics.
Source: Checking your browser - reCAPTCHA
To harness this for tissue rejuvenation, one theoretical strategy is to use engineered scaffolds or oligomers that mimic the mechanical and biochemical cues of the native extracellular matrix.
These scaffolds could:
- Provide nucleation surfaces that localize and orient tropoelastin coacervates, reducing the critical concentration needed for assembly;
- Present complementary hydrophobic or charged motifs that stabilize droplet interactions and guide fusion into higher-order structures;
- Incorporate binding domains or crosslinking sites that facilitate subsequent enzymatic stabilization into mature fibers.
Synthetic elastin-like polypeptides (ELPs) are an example of such design principles. ELPs are repetitive polymers that emulate the tropoelastin sequence and retain phase transition behavior similar to coacervation, making them candidates for scaffolds or injectable matrices in regenerative engineering. In tissue engineering, ELP‑based materials have been used to create hydrogels or modify existing scaffolds to promote cell proliferation and elastic matrix deposition.
Collagen and Coacervate Guided Scaffolds
Collagen regeneration presents analogous challenges because it also assembles hierarchically in the extracellular space, though via a different mechanism. Combined scaffolds that integrate collagen and elastin motifs have been explored in dermal and bone regeneration contexts, demonstrating that structural support materials can promote tissue integration, new matrix deposition, and cell infiltration.
Source: Collagen-Elastin-Like Polypeptide-Bioglass Scaffolds for Guided Bone Regeneration - PubMed
Bridging Fundamental Mechanisms and Therapeutic Goals
Understanding coacervation at the molecular level, including how hydrophobic and hydrophilic domains drive phase separation and how environmental factors modulate it, provides a foundation for rational design of scaffolds that assist or accelerate fiber assembly.
In longevity or aesthetic applications, materials that guide coacervation and enhance enzymatic crosslinking could, in theory, improve the quality and resilience of elastic and collagenous tissues in the skin, vasculature, and other organs by restoring structural integrity lost with aging. This remains an area of research rather than an established therapy, but the biomolecular insights and engineered approaches (like ELPs and composite scaffolds) point toward tangible paths to influence extracellular matrix regeneration.
The Future of Topical and Injectable Anti-Aging Aesthetic Products
We are significantly closer to practical topical and injectable aesthetic treatments that influence elastin and collagen regeneration than to broad systemic rejuvenation therapies. For example, a recombinant humanized elastin gel applied to skin was shown to restore dermal elastic fibers and improve skin elasticity and extracellular matrix remodeling in animal models of aging, outperforming some traditional antioxidant treatments.
Source: Checking your browser - reCAPTCHA
Other research has shown that designed elastin‑like polypeptides can be absorbed through the skin and improve measures of skin elasticity and reduce wrinkles in human participants over weeks of topical use.
Source: https://pubmed.ncbi.nlm.nih.gov/36628198/
These approaches operate locally, offering aesthetic benefits such as improved elasticity, firmness, or reduction of wrinkles. They tend to require less systemic exposure and can be designed to target skin tissue specifically, simplifying safety considerations and regulatory hurdles compared with systemic interventions.
Systemic Longevity Rejuvenation Products
Systemic regenerative therapies aimed at elastin and collagen homeostasis throughout the body face greater complexity and uncertainty. Elastic and collagenous tissues in internal organs like lungs, arteries, and heart valves have tightly regulated architecture and biomechanical roles.
Systemic rejuvenation would require not only delivering regenerative cues throughout the body but also precise control over when and where new extracellular matrix is formed. Differences in tissue turnover rates, local biochemical environments, and cell types across organs make this challenging.
Rigorous in vivo studies in animal models and comprehensive clinical trials in humans would be necessary to ensure both efficacy and safety before any systemic product could be widely recommended.
Safety Considerations
Uncontrolled stimulation of elastin or collagen synthesis carries inherent risks. While enhanced production in skin might improve appearance, similar upregulation in other tissues could contribute to negative remodeling.
In lungs and other organs, aberrant elastin deposition or disorganized fiber networks are associated with tissue stiffening and functional impairment, as seen in chronic respiratory diseases where extracellular matrix remodeling worsens mechanical function.
Source: https://respiratory-research.biomedcentral.com/articles/10.1186/s12931-023-02376-8
Even in skin, simply increasing elastin or collagen quantity is not enough; proper organization and integration into the existing matrix are critical. Age‑related decline in elastin is characterized by fragmentation and disorganization of fibers, which contributes to laxity and loss of mechanical integrity. This implies that any therapy should not only stimulate synthesis but also support correct fiber architecture and facilitate the removal of degraded elastin and collagen, effectively clearing damaged matrix components in a senolytic-like process.
Source: https://www.nature.com/articles/s41598-025-14393-2
For both aesthetic and longevity applications, safety strategies might include local targeting to avoid affecting deeper or internal tissues, controlled dose and timing of active agents, and monitoring of tissue responses over time. These considerations become even more crucial for systemic approaches, where off‑target effects could potentially harm organ function rather than promote rejuvenation.
Potential Additional Longevity Benefit from ECM Efficiency
Improving the organization and integration of elastin and collagen fibers could reduce the body’s need to constantly synthesize new protein for tissue maintenance. Well-structured and crosslinked extracellular matrix would maintain tissue function with less turnover, lowering the demand on fibroblasts and other matrix-producing cells.
This reduced protein synthesis could, in turn, modestly decrease systemic mTOR activation, supporting improved autophagy, cellular homeostasis, and stress resilience. Optimizing ECM efficiency may therefore offer a subtle but meaningful avenue for longevity, complementing strategies that target dietary protein or other metabolic pathways.
SUNDAY 1ST OF MARCH 2026 UPDATE ON THE LATEST RELEVANT ELASTINOGENESIS STUDIES:
Why this seems important: This study highlights a molecular mechanism, prolyl hydroxylation, that directly affects elastin’s structural stability. By increasing local rigidity and reducing flexibility, hydroxylation may help elastin fibers resist degradation and maintain proper assembly, which are key factors in tissue resilience and aging. From a translational standpoint, this points to future research directions such as enhancing prolyl hydroxylase activity, designing hydroxyproline-mimicking molecules, or optimizing tropoelastin assembly in regenerative therapies. Understanding and manipulating this process could eventually enable strategies to slow elastin breakdown and rejuvenate elastic tissues in the skin, vasculature, and other organs.
This likely would only slow elastin aging, not repair damage.
“We perform classical atomistic molecular dynamics simulations on elastin models of different length, originating from the tropoelastin monomer, namely, short elastin motifs, domain 18, and full-length tropoelastin, in the presence of various combinations of hydroxyproline in place of proline residues, to study the effects of hydroxylation in elastin. Our findings demonstrate that prolyl hydroxylation strengthens local hydration effects, particularly by enhancing protein-water hydrogen bonding. This gives rise to local rigidity, characterized by reduced accessible conformational states and fluctuations in the local environment of the elastin molecules. Consequently, the structural ensembles of elastin are altered, and the protein dynamics are dampened, potentially contributing to reduced enzymatic degradation and abnormal assembly and crosslinking behavior of elastin-like proteins, as observed in experimental studies. These findings, in conjunction with existing literature, underscore the critical role of prolyl hydroxylation in elastin function and its potential role in adapting to the varying mechanical needs of different connective tissues. Additionally, they illuminate the process of native elastin assembly and offer valuable insights into the potential involvement of hydroxylation in the degeneration of elastin in aging and disease. Based on these findings, strategically placed hydroxyproline residues may offer a means of engineering elastin-based materials with desired properties. Nevertheless, further research is needed to elucidate the precise mechanisms by which hydroxylation regulates elastin properties, which could provide valuable insights for applications in tissue engineering and regenerative medicine.”
Functions of Prolyl Hydroxylation in Elastin: https://www.jbc.org/article/S0021-9258(26)00193-6/fulltext
Why this seems important: Arterial stiffness, a major driver of vascular aging, can be mitigated by compounds that enhance elastin integrity and reduce vascular inflammation. Extracts from Garlic (Allium sativum), Amla (Phyllanthus emblica), Dill (Anethum graveolens), Centella asiatica, Carica papaya, Labisia pumila, Moringa oleifera, and bioactives like polyphenols, ginsenosides, curcumin, L-citrulline, saponins, flavonoids, betalain, and tyrosine promote elastin neo-synthesis, improve endothelial function, and limit oxidative stress.
Pharmaceuticals such as GLP-1 receptor agonists, SGLT-2 inhibitors, and DPP4 inhibitors further protect elastin by reducing glucose-driven AGE damage, suppressing inflammation, and enhancing vascular remodeling.
Targeted research could specifically analyze which components of plant extracts are having the greatest neoelastinogenesis effect to gather information for further combinatorial study, and for the research and development of novel compounds specifically targeted at neoelastinogenesis.
“By reducing arterial stiffness, plant extracts or derived bioactive compounds not only improved vascular relaxation by enhancing Nitric Oxide production and/or antioxidant defences, but also inhibited inflammation-induced aortic remodelling and promoted elastin neo-synthesis. Polyphenols have often been identified as the main effective compounds involved in these beneficial effects. However, only a few studies explained the mechanisms associated.”
The Conventional and Alternative Therapeutic Approaches in Arterial Stiffness Management https://www.mdpi.com/1999-4923/18/2/166
Why this seems important: A traditional botanical extract (Guang Chenpi) enriched in flavonoids like hesperidin, nobiletin, and tangeretin upregulated elastin (elna) gene expression and improved regenerative responses in zebrafish, while also enhancing antioxidant defenses and synergizing with ergothioneine and polydeoxyribonucleotide (PDRN) to protect against UV‑induced skin damage.
In vivo, GCE boosted elastin and collagen gene expression, improved wound regeneration, strengthened the skin barrier, and reduced oxidative stress without toxicity at low doses, pointing to natural compounds that could be studied for activating endogenous elastogenesis and extracellular matrix repair pathways.
“GCE also upregulated collagen and elastin gene expression, improved blood circulation, and suppressed melanogenesis.”
Guang Chenpi Extract as a Multifunctional Phytotherapeutic: Enhanced Effects with Ergothioneine and Polydeoxyribonucleotide on Redox Homeostasis and Tissue Resilience Guang Chenpi Extract as a Multifunctional Phytotherapeutic: Enhanced Effects with Ergothioneine and Polydeoxyribonucleotide on Redox Homeostasis and Tissue Resilience - PubMed
Why this seems important: Chronic lung inflammation can lead to two divergent outcomes: emphysema, characterized by progressive alveolar destruction, or interstitial fibrosis, marked by excessive collagen deposition and tissue stiffening. Percolation theory frames these outcomes by showing that the density and spatial distribution of collagen and elastin crosslinks act as critical determinants of tissue fate. Enzymatic crosslinks, primarily mediated by lysyl oxidase (LOX/LOXL1-4) and transglutaminases (TG2), along with non-enzymatic crosslinks such as AGEs (pentosidine, glucosepane, CML), dictate whether the extracellular matrix (ECM) maintains mechanical connectivity or becomes prone to catastrophic failure. Loss or degradation of desmosine/isodesmosine elastin crosslinks drives downward percolation transitions in emphysema, fragmenting tissue architecture, whereas excessive crosslinking and matrix stiffening induce rigidity percolation in fibrosis, forming self-reinforcing fibrotic networks. These insights highlight how local ECM alterations propagate systemically and suggest that the balance between crosslink formation and degradation governs tissue resilience.
Extending beyond the lung, this principle may apply to other elastin- and collagen-rich organs, including skin, arteries, and joints. Longevity strategies that modulate LOX activity, selectively stabilize elastin crosslinks, or break pathological crosslinks with AGE breakers or TG2 inhibitors could preserve tissue elasticity and slow structural aging, while avoiding maladaptive fibrosis.
“The percolation perspective ultimately reframes our understanding of chronic lung disease from a purely biochemical problem to one involving critical transitions in physical network properties. Crosslinking serves as the molecular mechanism controlling these transitions, making it both a marker of disease state and a therapeutic target. By recognizing that emphysema and fibrosis represent opposite sides of percolation thresholds, we can develop more rational, mechanism-based approaches to preventing and treating these devastating diseases. The challenge ahead is to translate these insights into clinical tools that can detect approaching transitions and interventions that can stabilize lung tissue within the homeostatic range between degradative and rigidity percolation thresholds.”
Percolation Forces in Lung Inflammation: Determining the Path to Emphysema or Fibrosis https://www.mdpi.com/2227-9059/14/2/281
Why this seems important: Strategies that successfully guide adult tissue to deposit and organize functional elastin and collagen are exceptionally rare, yet they are central to maintaining elasticity in skin, arteries, and other connective tissues with age. The vascular graft study demonstrates that a carefully designed scaffold, combining mechanical reinforcement, porous architecture, and bioactive cues, can drive in situ tissue remodeling and promote robust elastin and collagen deposition in a fully adult environment. This provides a proof of principle that adult neoelastinogenesis can be achieved when structural and biochemical signals are appropriately combined, suggesting a translational pathway for longevity and aesthetic interventions aimed at preserving tissue elasticity and mitigating age-related stiffening.
“This study demonstrates the successful development and evaluation of a biofunctional, mechanically reinforced electrospun vascular graft designed for hemodialysis access applications. The grafts supported robust host integration, characterized by cellular infiltration, endothelial coverage, and ECM deposition of both collagen and elastin —hallmarks of early vascular remodeling. Compared with commercial PTFE grafts, the electrospun scaffolds exhibited superior tissue ingrowth, reduced acellularity, and clear evidence of vascular-like neotissue formation, highlighting their potential to improve long-term patency.”
Biofunctionalized Vascular Access Graft Improves Patency and Endothelialization in a Porcine Arteriovenous Model https://www.mdpi.com/2079-4983/17/2/65
Why this seems important: Pentapeptide-18 functions similarly to botox in that it reduces the repetitive mechanical stress caused by facial muscle contractions, which normally disrupts collagen and elastin alignment. Unlike botox, it works subtly by modulating neurotransmitter release rather than fully paralyzing muscles, allowing natural expression while supporting more orderly dermal fiber deposition. This creates an environment where fibroblasts can lay down new collagen and elastin with higher structural fidelity, potentially enhancing neoelastinogenesis. In addition, pentapeptide-18 interacts with key skin-aging pathways including TGF-β, TNF-α, MAPK, PI3K/AKT, and NF-κB, helping regulate inflammation, oxidative stress, and extracellular matrix remodeling. By combining mechanical stress reduction with direct molecular support of ECM integrity, it represents a promising, non-invasive strategy for both aesthetic anti-aging and long-term dermal resilience.
“Skin aging is predominantly associated with the progressive breakdown of the dermal extracellular matrix (ECM), leading to visible manifestations such as wrinkle development caused by repetitive facial muscle movements and a gradual reduction in collagen and elastin synthesis. This degeneration is intensified by external factors including ultraviolet exposure, tobacco use, and nutritional imbalance, which interfere with major intracellular signaling pathways-namely TGF-β, TNF-α, MAPK, PI3K/AKT, and NF-κB-that collectively regulate inflammatory responses, oxidative stress, and ECM remodeling.”
Pentapeptide-18 as an anti-aging candidate: Spectroscopic characterization and molecular interaction analysis https://www.sciencedirect.com/science/article/abs/pii/S1476927126000770
Why this seems important: PHOA shows that chronic overactivation of inflammatory and proteolytic pathways, particularly PGE2-driven upregulation of MMPs and VEGF, can disrupt elastin and collagen structure, leading to tissue laxity and ECM remodeling. The observed differences in prevalence among racial groups suggest that underlying genetic variation may influence susceptibility to elastin and collagen degradation. Deeper analysis of these aggregate genetic differences could reveal molecular mechanisms that govern elastin and collagen homeostasis, offering insights that might be leveraged to enhance neoelastinogenesis and improve connective tissue resilience in aging populations.
“Histologically, sebaceous gland hyperplasia was most commonly described (72%), followed by inflammatory infiltrates (50%), tarsal plate fibrosis or thickening (44%), mucin deposition (28%), and alterations in elastin fibers (11%)…
… Further studies targeting disease specific cytokines, VEGF and MMP are needed to explore additional medical therapies.”
Primary hypertrophic osteoarthropathy presenting with ptosis and floppy eyelids: a review of ophthalmic manifestations, histopathology, and pathophysiology https://www.tandfonline.com/doi/full/10.1080/01676830.2026.2627478
Why this seems important: miR-378a-5p protects VSMCs’ contractile phenotype and reduces MMP2-mediated elastin and collagen degradation in the aortic wall. By limiting extracellular matrix breakdown, it slows structural deterioration associated with AAA formation. While this does not directly induce elastin regeneration, maintaining matrix integrity is a critical prerequisite for any subsequent neoelastinogenesis strategies, since intact scaffolding is necessary for new elastin fibers to form and integrate effectively.
“Compared with the antagomir-NC + Ang II group, increased collagen disruption and elastin degradation were observed in the abdominal aortas of the antagomir-378a-5p + Ang II group"
miRNA‑378a‑5p attenuates the development of abdominal aortic aneurysm via ABLIM1‑MKL1 signaling pathways International Journal of Molecular Medicine
Why this seems important: This study highlights that TRPML1 plays a central role in regulating extracellular matrix turnover by controlling the release of matrix metalloproteinases (MMPs) from lung fibroblasts and macrophages. Loss of TRPML1 reduces MMP exocytosis, which leads to decreased collagen and elastin degradation, causing accumulation of these ECM proteins and a fibrosis-like phenotype. This suggests that TRPML1 activity primarily influences the degradation side of elastin and collagen homeostasis rather than directly promoting new elastin formation. For neoelastinogenesis, the implication is that maintaining or enhancing TRPML1-mediated MMP release could help clear damaged or misassembled elastin, potentially creating a tissue environment more permissive for regeneration by reducing ECM crowding and abnormal cross-linking, but it does not directly stimulate elastin synthesis itself.
“In addition to the reduction of several important collagenases, the role of MMP12 in elastin degradation may also be a critical factor in the development of the observed phenotype. Indeed, increased elastin levels have been reported previously to play a role in fibrosis and fibrosis development (Blaauboer et al, 2014; Upagupta et al, 2018; Mariani et al, 1995; Enomoto et al, 2013; Yombo et al, 2023; Hoff et al, 1999; Hansen et al, 2016). In healthy lungs, elastin contributes to proper lung function and the elasticity of lung tissue. Elastic fibers provide the elasticity needed during inhalation and ensure efficient gas exchange in the lung. While reduced elastin levels promote emphysema, excess amounts of ECM molecules such as collagen and elastin in PF are well documented to result in progressive fibrosis, scarring, and impaired lung function by reducing lung compliance, impacting ventilation, and thus compromising gas exchange”
TRPML1 suppresses pulmonary fibrosis by limiting collagen and elastin deposition TRPML1 suppresses pulmonary fibrosis by limiting collagen and elastin deposition | The EMBO Journal | Springer Nature Link
Why this seems important: This study demonstrates that extracellular vesicles (EVs) from human bone marrow mesenchymal stem cells carry microRNAs capable of suppressing fibrosis-related gene expression, including elastin, in hepatic stellate cells. The mechanism involves downregulating RhoA signaling, which reduces cellular activation and migration associated with fibrogenesis. For neoelastinogenesis, this is significant because it highlights a strategy to actively reduce excessive or pathological ECM deposition, including elastin, and to modulate the cellular environment toward a less fibrotic, more regenerative state. By clearing or restraining overproduced or misassembled elastin and collagen, MSC-EV microRNAs may create conditions that are more favorable for elastin regeneration, distinguishing this effect from simply slowing age-related degradation.
“This study identified five anti-fibrotic miRNAs enriched in MSC-EVs and elucidated the underlying mechanism of the anti-fibrotic action of MSCs.”
Extracellular vesicle microRNAs from human bone marrow MSCs suppress fibrogenesis of hepatic stellate cells by downregulation of RhoA signaling https://academic.oup.com/stmcls/advance-article-abstract/doi/10.1093/stmcls/sxag006/8489158
My plan over 2026 is to continually review all of the latest studies on elastin to hopefully uncover actionable interventions for neoelastinogenesis, and to come up with plausible source backed theories to be tested for proper elastin regeneration.
Could you please share the link to the product you get and your supplier? I have done mesotherapy on myself and would love to try the Radiesse alternative you mentioned ![]()
The only way elastogenisis can be “restarted” is with gene therapy IMHO, maybe CRISPER with an off switch. AI may be able to come up with a solution at some point but one would need to feed it a LOT of data on this problem.
The first thing you need to understand is how elastin made and why (more importantly) that process stops at the age of 13.
Once you come to grips with that, you will have a better idea of why this is such a difficult problem, which very few researchers are working on. Because it is well known in dermatology at the highest (honest) levels, any company looking to do real research on this has a hard time getting funding.
Very minor superficial repair in the skin is not a real solution, that’s not even a lead to a systemic solution.
I do differentiate between skin focused therapies and systemic therapies.
It’s not just the creation of new elastin that is needed, because too much is part of problems like fibrosis. What is needed is the right configurations of elastin in the right areas.
As mentioned in the theory above, certain scaffolds are probably needed to guide this formation.
Something like CRISPR would be even better of course.
I’ll continue doing these periodic reviews of the latest literature to uncover information. Hopefully we gain a clearer picture as I continue this process, and hopefully anyone with any connections to science and studies takes that information and runs with it.
If I find anything particularly interesting I’ll consider fundraising a study, but that would be very far down the road if it happens.
I was going to consistently look at the latest studies on collagen and elastin but I’ve had a lot going on in my personal life and my interests in longevity had moved to different areas. However when I come across something interesting I will still bring attention to it here.
Collagen and elastin are part of the extracellular matrix, something from my view is not focused on enough in the longevity space.
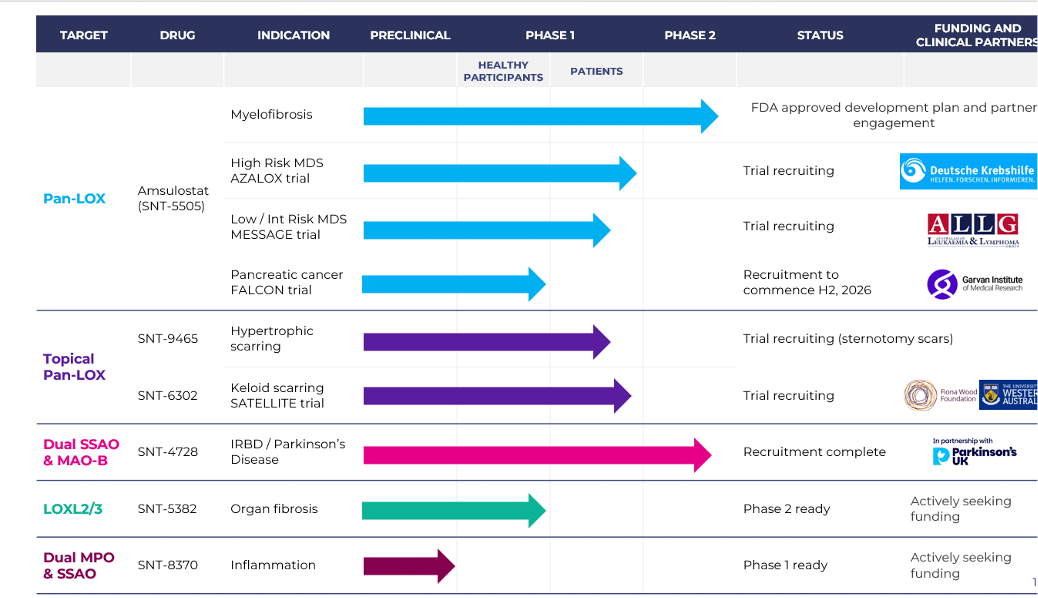
An Australian biotech company that is particularly focused on the extracellular matrix was brought to my attention: Pipeline – Syntara
“Currently, our clinical development efforts are primarily focused on evaluating novel drugs targeting a subset of oxidase enzymes known as lysyl oxidases. This family of enzymes are uniquely responsible for the development of fibrosis by cross-linking collagen and elastin, resulting in hardened, poorly-degradable scar-like tissue.”
While their interventions are currently targeted at scar tissue and various other specific conditions I get a feeling that something in this collection of drugs they are developing are going to have beneficial off-target effects for elastin/collagen for skin aging, as well as overall vascular/systemic aging.
After three months, patients receiving SNT-6302 treatment showed significant improvements in scar vascularisation and extracellular matrix remodelling compared to placebo-treated patients. Advanced imaging analysis provides compelling evidence that scars appeared structurally and biologically closer to normal, uninjured skin. Source: BCAL Diagnostics and Sonic Healthcare advance validation of BreastestPlus Version 2 for US market - Biotech
"In 2023, Professor Wood led clinical trials conducted at the University of Western Australia, identifying a drug developed by biotech company Syntara (ASX: SNT) as one that could reinvigorate elasticity of the skin in scar tissue.
Syntara’s pan LOX (lysyl oxidase) inhibitors caught her attention, with potential to inhibit the enzymes that create scar tissue. Research was put into clinical trials in 2023 with 42 patients volunteering to apply the topical cream to their scars in a 3-month study.
The results were highly encouraging, showing 66% reduction in LOX enzyme activity which is responsible for the cross linking of collagen fibres implicated in adverse scarring. Collagen was reduced by 30%, a result Professor Wood described as “an unprecedented change to the scar composition that we have not seen with any other form of treatment.”…
…Syntara’s pan LOX inhibitors, however, have shown the potential to permanently modify the scar by reducing the cross linking of collagen fibres and collagen, holding out the potential to revert scars back to their pre-burn elasticity and prevent bad scars forming."
https://thesentiment.com.au/syntara-prepares-for-new-clinical-trials-led-by-professor-fiona-wood-to-reinvigorate-elasticity-into-burn-scars/
From their website:
Syntara using pan-LOX inhibitors for skin scarring
Lysyl oxidases are a family of enzymes that play a critical role in scar formation and maintenance. Lysyl oxidases stabilize the main component of scar tissue, by the cross linking of collagen and elastin fibres, and drive scar stiffness and appearance. The process by which the inhibition of LOX enzymes can improve scars is:
- Inhibition of lysyl oxidases in the skin reduces cross-linking of extracellular matrix proteins (collagen and elastin).
- Collagen stability is reduced and remodeling is
- Consequently, scar tissue will be reduced and of diminished stiffness, with improved mechanical properties and appearance.
Syntara is developing topical and oral drugs inhibiting all lysyl oxidase family members with potential anti-fibrotic application in severe fibrotic related skin scarring indications
The company’s drug discovery, known as SNT-6302, has shown promising clinical results in inhibiting the LOX enzymes that play a critical role in the development of scar tissue. SNT-6302 was discovered by the company’s research team at the company’s Frenchs Forest laboratories. The project was awarded initial National Health and Medical Research Council (NHMRC) funding for some of the pre-clinical development work done in collaboration with researchers at the University of Western Australia, led by distinguished surgeon Professor Fiona Wood.
UWA researchers published the pre-clinical studies performed in collaboration with Syntara in Nature Communications showing the topically applied pan-LOX inhibitors reduced collagen deposition and cross-linking and improved scar appearance without reducing tissue strength.
In 2023 the phase 1c trial of SNT-6302 reported it had met its primary safety objective and two secondary biomarker endpoints in patients with established scars. 42 Australian patients with any type of scar older than 1 year and at least 10cm2 in size were recruited in the double‐blind three‐month study and applied either SNT‐6302 or placebo cream three times a week.
Applications of SNT‐6302 cream resulted in a mean 66% reduction in LOX activity when measured 2 days after the last dose (p<0.001) compared to baseline and to placebo group. LOX is responsible for the cross linking of collagen fibres implicated in adverse scarring. Professor Wood commented, “This exploratory clinical study has significantly enhanced our understanding of the role of LOX enzymes in scarring and the scar process itself. SNT‐6302 safely inhibits these key enzymes to a significant degree and leads directly to an unprecedented change to the scar composition that we have not seen with any other form of treatment. We estimate that up to 50% of the excess collagen in these patients’ scars has been removed and while the length of this Phase 1c safety study was not sufficient to change the appearance of an established scar the remodelling process will be ongoing and I’m confident we would see an improvement in scar appearance and physical characteristics if we observed them for longer.
Skin Scarring – Syntara