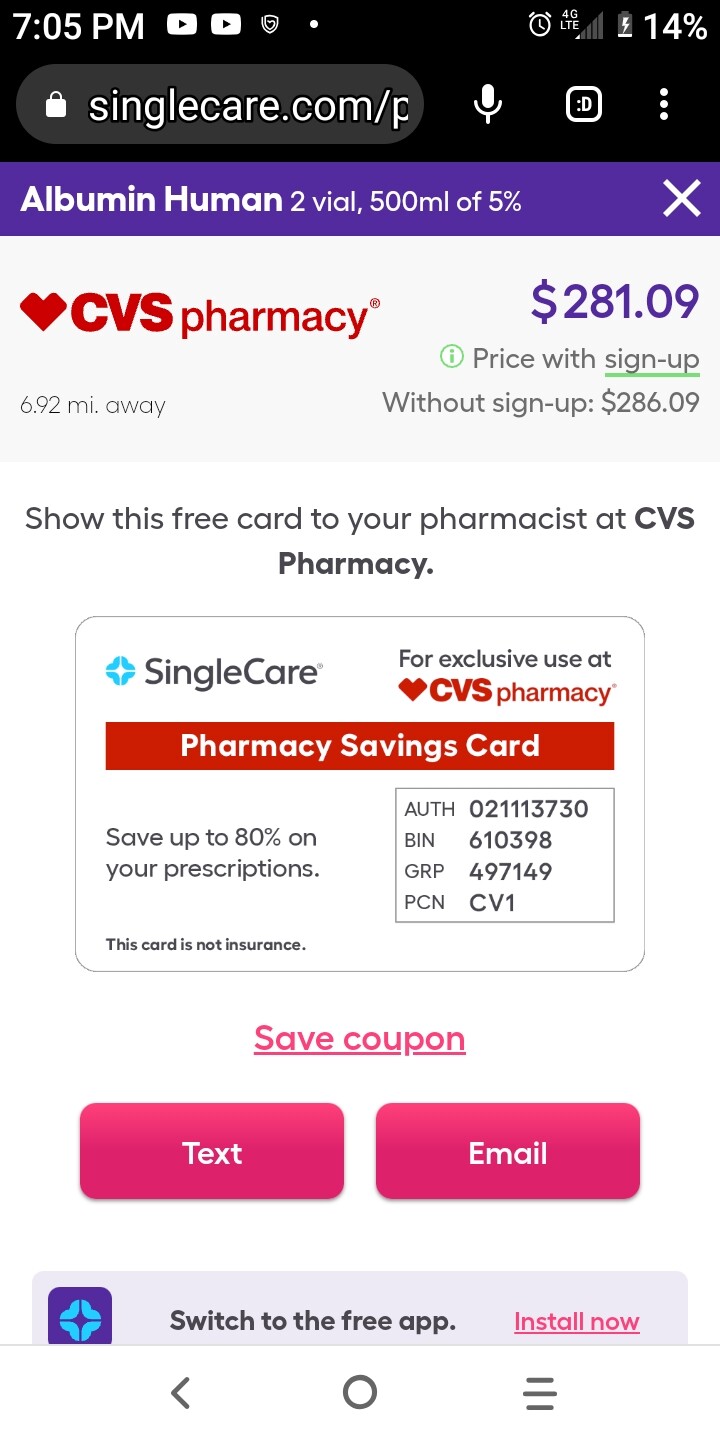
So - it seems we have two options; Albumin (which seems like we can get if we can get a prescription), and young blood plasma.
Its interesting… at point of purchase (i.e. what companies are currently paying people for a liter of blood plasma) it seems to be priced in the $100 to $200 range. That same liter (after processing… which I’ve yet to see defined exactly) it sells for $8,000 per liter from companies like the former Ambrosia.
If you want to do a frequent plasmapheresis, which I suspect might be weekly or every two weeks, most people are going to want to target paying at the lower end of the $100 to $8,000 per liter range.
Perhaps we need to start a non-profit organization for young blood donations that targets college athletes around the world, with an offer that gives these people a good income (perhaps paying $500 / liter), and also providing a priority access for the parents of the people doing the donating, for more incentive, and a viral marketing approach to get the word out. Develop a simple and viral phone app that teens and 20-somethings would find helpful, and the potential to tell friends about this income opportunity, so that they can easily get a bonus for signing up their friends.
To keep costs low, the customers (e.g. us ) could be offered subscription plans to get the young blood plasma mailed to you in cold containers every week, two weeks, etc. and you could either do the plasmapheresis process yourself or take the blood to a clinic where they do it for you.
This may also be a “for-profit” opportunity, but there is already lots of competition in companies buying blood plasma, and given the desire to get the price down as low as possible and also appeal to people’s better nature of helping older people (at a price most donor’s parents can afford), it seems like a non-profit or perhaps a public benefit (B- corp) might be a better organizational structure.
Payment for young blood is not allowed in many countries, I see: Blood for money: my journey in the industry buying poor Americans’ plasma | US healthcare | The Guardian
Hunting around more on the refurbished equipment side, @lara is right, it seems there are some much smaller devices that look more like a laser printer. You can put it on your office desk and use it while you are working on your computer 

https://bimedis.com/a-item/plasmapheresis-machines-haemonetics-pcs2-1987226