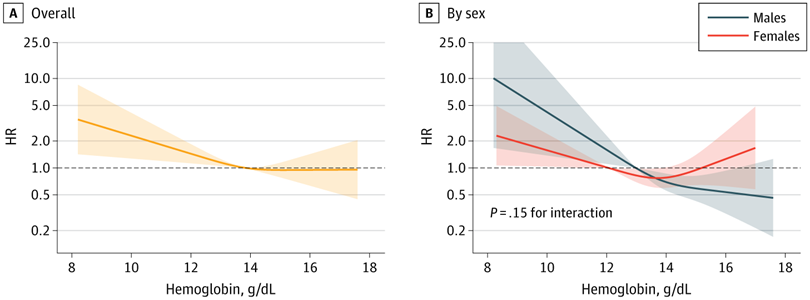
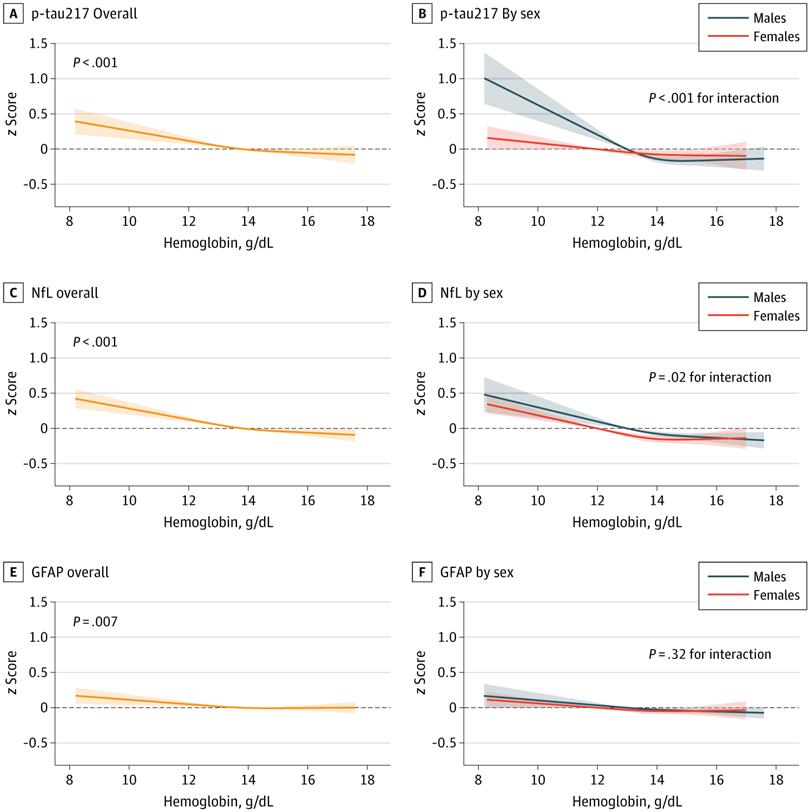
This has been an interesting topic because of looking at various people’s different circumstances and in particular the way that TRT skews iron metabolism.
I may be unique in doing weekly full panel blood tests (ie the discount full panel that any lab operates, which does not always include ferritin, but mainly does). Hence I have my ferritin figures since 2022. And here they are:
420 337 358 329 332 404 371 412 325 204.6 364 202 337 176.2 176.2 319 172.9 205 253 205 145 175 157.9 165.7 171.4 190.9 180.4 173.4 222 176.7 ng/ml 298 291 mgc/L 202.5 187.8 174.9 140.4 155.4 170.5 123.2 150 152.6 147.6 168.7 177 201 165 203 168 165 164 154 142 ng/ml 164.16 129 126 134.42 118 116.07 115 120.45 96 going down? 102.5 93 79.02 95 122.54 166 109 95 115.63 161 112 105 97 110 96 99 112 92 68.3 99.5 103.21 96 101.3 108 93 101 56 52.3 56.1 71.3 45.38 83.3 58.06 78.7 76.82 77.4 57.91 71.7 47.66 24.4 60.19 51.9 63.7 56.42 69.8 35.96 67.7 74.41 73.8 72.33 75.9 56.04 63 65.76 55 61 50.79 67.2 63.64 48.6 40.8 24.22 52.63 39.92 38.59 36.08 35.83 33.15 28.84 34.3 20.2 37.40 44.3 30.1 36.2 28.08 19.07 22.25 34.25 89 50.74 34.89 32.01 32.52 25.96 12.43 47 30.71 47.65 47.85 39.01 30.71 33.86 39.53 53 48 39.31 67 44.92 41.91 61 46.96 43.87 58.89 76 60.23 48.7 49.65 50.6 55.1 62 70 72 59.75 37.8 56.23 44.94 35.76 22.75 low iron 37.72 37.14 48 40.31 48.85 41.76 40.66 48.44 45.79 36.34 71 62 67.78 57.72 39.36 30.35 59.77 60.27 76 58.63 70.55 55 46 54.3
The first thing you can say from those is that ferritin is quite variable week to week. It think it probably moves in a sort of range of 15 (the units are the same number in US and Europe even if the units vary because the numerator and denominator vary by the same number).
It would need a bit more work on looking at the variability between ferritin tests to see how variable it is on a day or between days, but clearly you cannot take it as a biomarker like height.
I cannot find who gave the link to this website for some reason: Iron Status Interpreter
Edit 31/3 - it was @Kebossit on a renal thread who I thank.
However, that is a useful start on looking at iron status. I have not tried to work out how reliable it is.
It has changed since I last looked at it as it used to refer to Peter Attia saying ferritin should be in a range of something like 30-400.
Clearly there is a minimum iron level below which people have symptoms like hair loss, but obviously if there is enough iron in haemoglobin then the symptoms which result from a shortage of oxygen do not appear. Hence you cannot just rely on ferritin to judge this.
We know that GPs go for 30 and neurologists go for 70 (because of the dopamine pathway in the brain) - which is why RLS is a potential symptom of iron deficiency.
A few weeks ago I did a chatGPT review of iron and mitophagy. It is a bit long and the conclusion is simply that less iron is better for mitophagy. It has a lot of links to original papers. I have created a share which is:
There is some work in human cells and biopsies from humans
There are, however, as far as I can tell no actual trials into seeing the effects of keeping iron bouncing along the bottom.
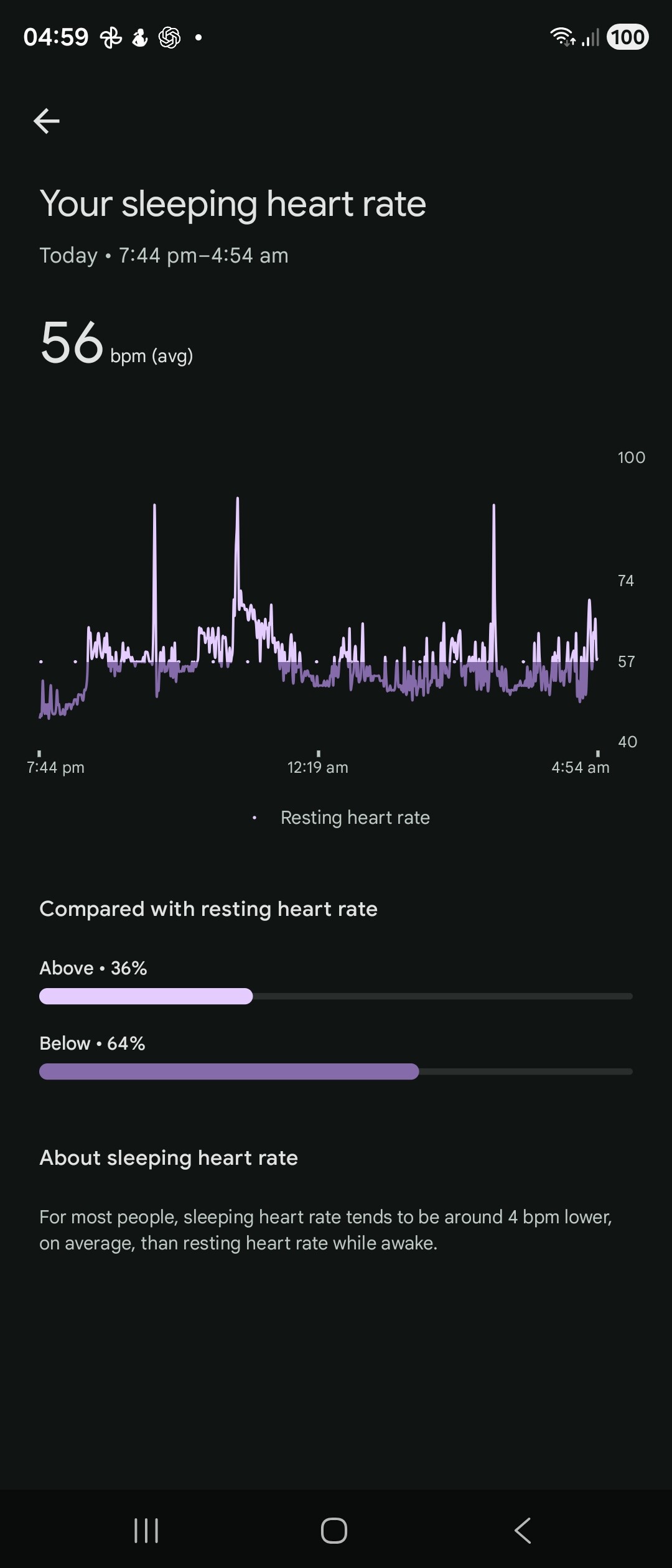
Obviously I am doing that myself in a biohacking manner. I have a hypothesis that a highly variable heart rate whilst sleeping is symptomatic of increased autophagy. The only evidence for this, however, is from my own experimentation.
I do think, however, the evidence is that for mitophagy lower ferritin is better, but realistically there are problems from low iron. I am not persuaded that the GP threshold of 30 is right and clearly if there is a nephrologist threshold of 10 then that is wrong. I do think it is important to maintain enough iron for dopamine metabolism, but that gives really an upper limit of 70 and one need to consider symptoms.
This was last night’s sleep tracker. I fasted yesterday and my Monday iron figures were:
Hb 145 g/l Ferritin 54.3 mcg/l CRP 0.4 mg/l Albumin 41.7 g/l MCV 92.5 fl Iron 7.1 mcmol/l TSat 13.1% TIBC 54.3 mcmol/l
TSat is low which is a sign of low iron. These things move quite rapidly, however.