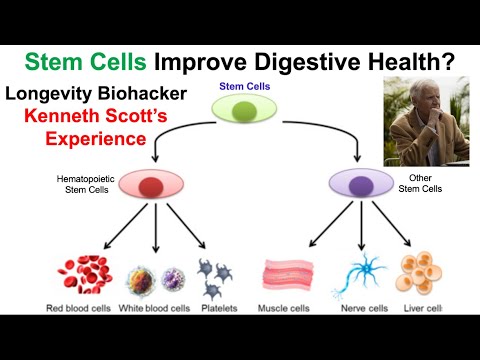
what percent of a person’s stem cells are left by age 40?
Studies suggest that by age 40, a person retains only about 25-30% of the stem cells they had at birth. Stem cell populations gradually decline with aging for the following reasons:
• Decreased self-renewal: Stem cells have a limited ability to self-renew, and this ability decreases with age. As stem cells divide over time, they lose the capacity to replicate themselves perfectly. This results in a slower replenishment of the stem cell pool.
• Increased senescence: Stem cells also undergo senescence, or aging-related changes, that inhibit their function. Senescent stem cells remain in the body but are no longer capable of differentiating into new cells. With accumulating senescent changes, fewer functional stem cells remain by age 40 and older ages.
• Environmental damage: Exposure to environmental stressesors, like inflammation, toxins, and radiation, can damage stem cells. The longer stem cells are in an aging body, the more likely they are to sustain damage that disrupts their self-renewal and differentiation capacity. This contributes to the decline of the remaining stem cell pool over the lifespan.In summary, for these reasons the percentage of remaining stem cells decreases substantially by age 40. Most stem cell loss occurs early in life, and fewer self-renewing, undamaged, and non-senescent stem cells remain at later ages. This contributes to age-related changes and decline of organ/tissue function. Understanding how to promote stem cell preservation and regeneration remains an active area of research for healthy longevity.
supposedly, zoledronate can reduce damage to stem cells
> how much does telomere shortening contribute to stem cell loss by age 40?
Telomere shortening likely contributes to stem cell loss by age 40, but it is not the only factor or necessarily the primary contributor. Here are some key points:
• Telomeres shorten with each cell division, and stem cells divide regularly to self-renew and produce new cells. By age 40, after many years of life and cell division, telomeres in stem cells have undergone substantial shortening. This telomere shortening is associated with stem cell senescence and reduced function.
• However, telomere length is not the only determinant of stem cell aging. Other factors like DNA/mitochondrial damage, epigenetic changes, and altered signaling/gene expression also contribute to age-related stem cell decline. These additional influences likely play a significant role in stem cell loss by age 40, along with telomere shortening.
• In addition, some stem cell populations have higher telomerase activity that helps maintain telomere length. Not all stem cells will have critically short telomeres by age 40. The degree of telomere shortening will depend on the specific stem cell type and factors regulating telomere maintenance in those cells. So while telomere shortening contributes to stem cell loss over time, it is not solely responsible or necessarily the primary contributor, especially by age 40.In summary, telomere shortening likely plays a role in stem cell loss by age 40 due to the cell divisions stem cells undergo. However, other drivers of stem cell aging are also important, and different stem cell populations have varying telomere dynamics. Telomere length is one influence on stem cell decline with aging, but not the only determinant. More research is needed to understand the relative impacts of telomere shortening and other factors on stem cell loss at different ages.
> is telomere length in PBMCs a valid proxy for telomere length in stem cells?
Telomere length in peripheral blood mononuclear cells (PBMCs) can provide some indication of whole-body telomere dynamics, but may not be a perfect proxy for telomere length in stem cells. Here are some key points:
• PBMC telomere length correlates with telomere length in other tissues and cell types, including stem cells. As telomeres shorten systemically with aging, PBMC telomeres can give a sense of the overall telomere shortening rate.
• However, telomere length can vary between different tissues and cell types. Stem cells have higher telomerase activity to maintain their telomeres, so stem cell telomeres may shorten at a slower rate than PBMC telomeres. Other factors like oxidative stress and inflammation also influence telomere length, and can affect PBMCs and stem cells differently.
• In addition, PBMCs are a heterogeneous mix of cell types, and telomere length can differ between these subsets of cells. The specific composition of PBMCs may further influence how well they reflect stem cell telomere length. Isolating specific PBMC subsets or stem cells directly could provide a more precise measure of telomere length for comparison.In summary, while PBMC telomere length can provide a general sense of whole-body telomere dynamics, it may not perfectly reflect telomere length in stem cells. Stem cells have higher telomerase and their telomeres could shorten at different rates than PBMC telomeres. The heterogeneous mix of PBMCs could also affect their correlation with stem cell telomere length. Direct measurement of telomere length in specific PBMC subsets or stem cells would likely give a more accurate comparison, though this is not always feasible. PBMC telomere length should be interpreted with these limitations in mind.
how much does asymmetry in stem cell division preserve stem cell quantity?
Asymmetric stem cell division, where a stem cell produces one copy of itself and one differentiated cell, helps preserve stem cell quantity. By dividing asymmetrically, stem cells can self-renew while also producing mature cells as needed. This balances self-renewal and differentiation, and prevents uncontrolled exhaustion or depletion of the stem cell pool. Some key points:
• With asymmetric division, the number of stem cells stays the same or increases gradually with tissue growth. This maintains a stable pool of stem cells for maintenance and regeneration. Without asymmetric division, stem cells would progressively differentiate and be depleted.
• The rate of asymmetric division varies between tissues and is regulated based on need for differentiated cells. A higher rate of asymmetric division is needed when more differentiated cells are required for growth or damage repair. The rate slows when existing differentiated cells are sufficient. This allows stem cell quantity to be preserved while meeting tissue demands.
• Asymmetric division requires precise regulation to be effective. The process that partitions cellular components into the two daughter cells must be carefully controlled. If the partition is imbalanced, it could lead to over-production of stem cells or differentiated cells. Proper signaling and cellular machinery is necessary to regulate asymmetric division and its ratios.In summary, asymmetric stem cell division helps balance self-renewal and differentiation, stabilizing stem cell quantity. It allows for production of differentiated cells while preserving the stem cell pool. The rate of asymmetric division varies between tissues and with conditions, but effective regulation of the process is necessary to generate proper ratios of stem and mature cells. Asymmetric division is an important mechanism for sustaining healthy stem cell populations and tissue maintenance/regeneration.
How does ROS regulate asymmetrical stem cell division?
ROS (reactive oxygen species) can regulate asymmetric stem cell division in the following ways:
• Oxidative stress signaling: Low levels of ROS act as signaling molecules that trigger responses to oxidative stress. This ROS signaling can influence the proteins and pathways involved in regulating asymmetric division. For example, ROS may activate factors that promote differentiation versus self-renewal in the two daughter cells. This allows stem cells to sense and respond to redox state, influencing division asymmetry.
• Telomere shortening: ROS also contribute to telomere shortening with aging and cell division. As telomeres shorten, it alters the partitioning of telomeres in asymmetric division. This can lead to one daughter cell getting critically short telomeres, forcing it to differentiate. The other cell retains longer telomeres, allowing it to remain a self-renewing stem cell. So telomere dynamics and ROS-induced telomere shortening may impact division asymmetry.
• Epigenetic regulation: The epigenetic landscape of stem cells is regulated in part by ROS levels. As the histone code and DNA methylation change with redox state, it impacts which genes are expressed in the two daughter cells. This results in different epigenetic/differentiation programs emerging from asymmetric division. So epigenetic regulation via ROS could be another way that division asymmetry is modulated.In summary, ROS may regulate asymmetric stem cell division through oxidative stress signaling, influencing telomere dynamics, and shaping the epigenetic landscape. Low levels of ROS are involved in establishing the self-renewal vs. differentiation programs adopted by the two daughter cells. Effective regulation of redox state and responses to ROS is necessary for proper asymmetric division and balanced self-renewal/differentiation. However, excessive ROS can impair stem cell function, so ROS must be maintained at appropriate levels.