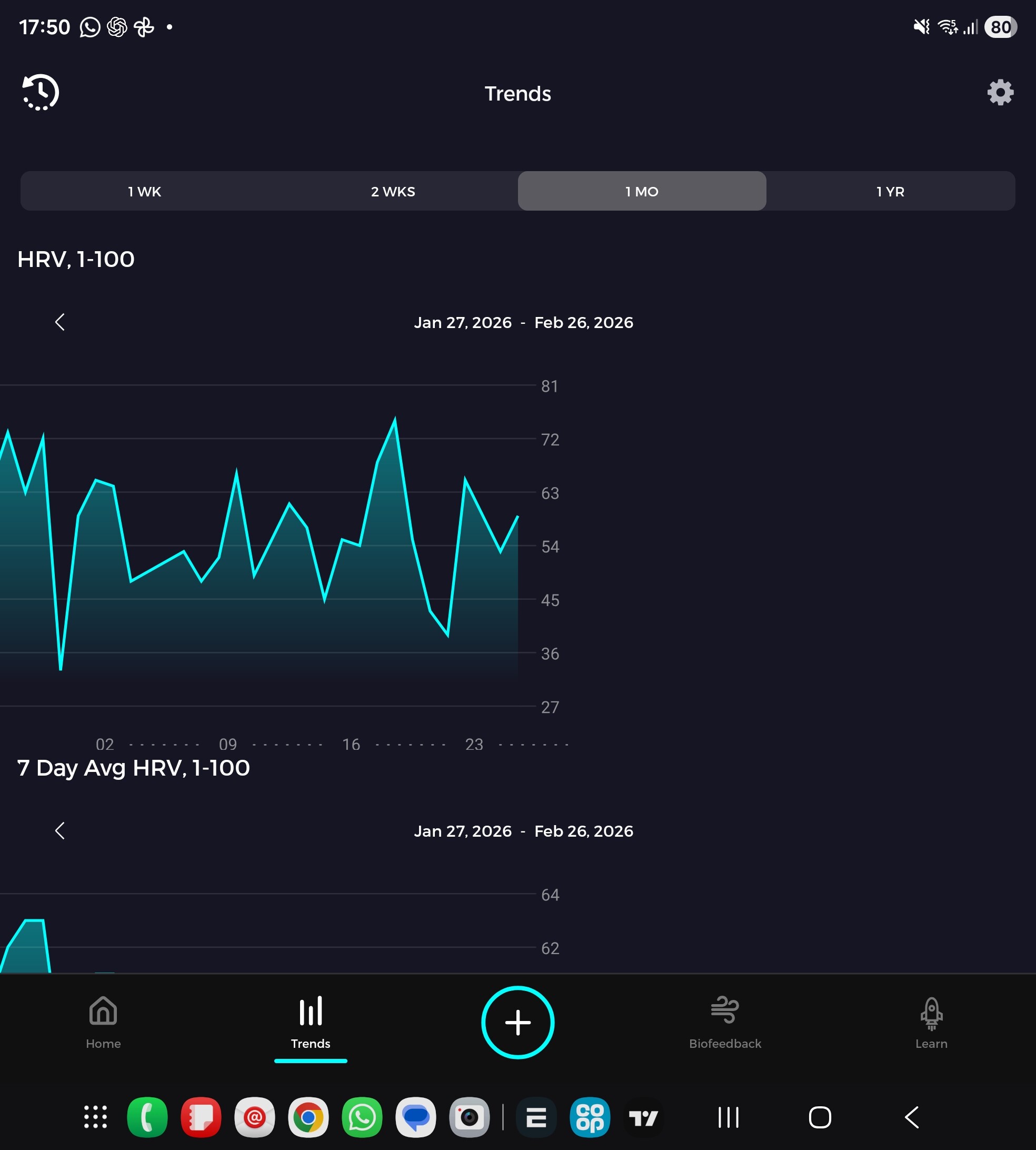
I. Executive Summary
The provided transcript centers on the physiological utility of the Heart Rate Variability Coefficient of Variation (HRV-CV) as a digital biomarker for healthspan and autonomic nervous system resilience. HRV-CV, defined mathematically as the seven-day standard deviation of HRV divided by its seven-day mean, quantifies day-to-day cardiac autonomic fluctuations. The primary thesis posits that a lower HRV-CV (greater stability) indicates superior adaptation to physiological and psychological stressors, whereas a higher HRV-CV reflects poor allostatic load management and elevated cardiovascular risk.
The discussion is anchored by a retrospective analysis of approximately 21,000 wearable device users, establishing that HRV-CV requires a minimum of five recording nights per week for statistical reliability. The data reveal that HRV-CV is highly responsive to modifiable behavioral inputs—specifically alcohol consumption, physical activity, sleep duration, and sleep consistency. Crucially, the authors claim HRV-CV demonstrates greater sensitivity to acute behavioral perturbations (like alcohol and sleep debt) than absolute HRV or resting heart rate (RHR).
Demographically, the data outline divergent biological trends. Females consistently exhibit lower (more favorable) HRV-CV across the lifespan compared to males, despite well-documented autonomic variations linked to the menstrual cycle. In males, HRV-CV remains flat until approximately age 40, after which it rises continuously, aligning with population-level increases in cardiovascular disease burden. Furthermore, a higher body mass index (BMI) correlates with elevated HRV-CV in both sexes.
From a translational perspective, the speakers propose HRV-CV as a novel metric for risk stratification, suggesting it provides early insight into insulin insensitivity, metabolic dysfunction, and maladaptation to training loads. The core actionable takeaway is that minimizing HRV-CV via behavioral consistency, strategic functional overreaching, and appropriate tapering can optimize physiological resilience. However, the cross-sectional nature of the primary dataset limits causal inferences. While HRV-CV is a promising metric for intra-individual monitoring, longitudinal clinical trials are required to validate it as an independent, predictive biomarker of mortality and disease progression.
II. Insight Bullets
- HRV-CV is defined as the seven-day standard deviation of an individual’s HRV divided by their seven-day mean HRV.
- A low HRV-CV score indicates high autonomic stability and favorable adaptation to ongoing physiological and psychological loads.
- A minimum of five nights of sleep-derived HRV data within a seven-day window is required to generate a statistically reliable HRV-CV calculation.
- Shift workers and acute care physicians exhibit severely elevated HRV-CV (high 20s to low 30s) compared to the general population, reflecting the massive physiological toll of circadian disruption.
- Elite endurance athletes typically demonstrate highly stable HRV-CV metrics below 10%.
- Alcohol consumption drastically inflates HRV-CV, exhibiting a negative physiological impact nearly double that of other measured behavioral variables.
- HRV-CV demonstrates greater mathematical sensitivity to sleep duration, sleep consistency, and alcohol intake than absolute HRV or resting heart rate.
- Females maintain a lower (superior) HRV-CV across their lifespan compared to males, even when controlling for behavioral variables like sleep debt and alcohol consumption.
- In males, HRV-CV is relatively stable from ages 18 to 40, after which it rises linearly, mirroring age-related cardiovascular disease onset.
- In females, HRV-CV displays a U-shaped trajectory, declining until age 50 before rising, accommodating the autonomic noise generated by menstrual cycle fluctuations.
- Elevated Body Mass Index (BMI) positively correlates with higher HRV-CV instability in both biological sexes.
- Late-night eating acts as an acute metabolic stressor that suppresses overnight HRV and destabilizes HRV-CV, acting as a potential proxy indicator for impaired insulin sensitivity.
- Chronically elevated HRV-CV in the absence of severe training loads implies poor allostatic load management or incipient metabolic dysfunction.
- Functional overreaching in athletic training temporarily increases HRV-CV, which should rapidly normalize following an appropriate recovery taper.
- “Vagal tank theory” posits that a healthy autonomic system requires parasympathetic withdrawal in response to acute stressors, combined with a rapid return to baseline.
- Absolute HRV naturally declines by roughly 20 percent per decade; thus, age-matched HRV-CV serves as a superior longitudinal metric for individual tracking.
- The “African-American paradox” demonstrates that higher absolute HRV does not universally correlate with lower cardiovascular morbidity, underscoring the limitations of inter-individual HRV comparisons.
III. Adversarial Claims & Evidence Table
| Specific Claim | What they cited | Verified status + PubMed/DOI Link | Evidence Grade (A-E) | Verdict |
|---|---|---|---|---|
| 5 days of data is the minimum required to accurately estimate 7-day HRV-CV. | Retrospective analysis of 21,000 Whoop users. | Verified. Grosicki et al., 2026 | Level C | Strong Support |
| Higher HRV-CV is associated with older age (>40 in men), higher BMI, and male sex. | Wearable data dataset published in Am J Physiol Heart Circ Physiol. | Verified. Grosicki et al., 2026 | Level C | Strong Support |
| Improved sleep consistency directly lowers HRV-CV and improves autonomic markers. | Internal platform data / Expert observation. | Verified. Sleep consistency interventions improve HRV and RHR. Fuller et al., 2026 | Level B | Strong Support |
| Late-night eating suppresses overnight HRV due to insulin insensitivity and metabolic load. | Observational cohort data. | Plausible. Late eating disrupts peripheral clocks and impacts blood pressure/cortisol, though direct HRV impact varies. Gutierrez et al., 2025 | Level C | Plausible |
| GLP-1 agonists will drastically improve HRV-CV by improving insulin sensitivity. | Speculation based on systemic metabolic benefits. | Contradicted. Clinical data frequently show GLP-1s increase heart rate and decrease absolute HRV due to sympathomimetic effects. Data needed. | Level E | Safety Warning |
| Absolute phenotypic HRV is a predictor of all-cause mortality, but comparing between individuals is flawed. | The “African American Paradox” and ectopic beats in the elderly. | Verified. Phenotypic HRV links to mortality, but genetic predisposition does not. Inter-individual comparisons are heavily confounded. Nolte et al., 2024 | Level A | Strong Support |
IV. Actionable Protocol (Prioritized)
High Confidence Tier (Level A/B Evidence)
- Establish a 5-Day Minimum Baseline: To utilize HRV-CV for actionable programming, ensure wearable device compliance for at least 5 out of 7 nights. Less data renders the coefficient statistically invalid for load management.
- Prioritize Sleep-Wake Consistency: Circadian alignment through consistent sleep and wake times is a primary driver of autonomic stability. Target a sleep consistency score above 80 percent to lower HRV-CV and improve baseline parasympathetic tone.
- Strict Alcohol Moderation: Alcohol acts as an acute systemic toxin that severely deregulates cardiac autonomic control. Limit intake completely during functional overreaching blocks to prevent artificial spikes in HRV-CV that mask true training adaptation.
Experimental Tier (Level C/D Evidence)
- HRV-CV Guided Periodization: Utilize the 7-day HRV-CV trend to dictate training volume. If HRV-CV is rising (exceeding baseline by 10-15 percent) alongside subjective fatigue, implement a deload week. Wait for HRV-CV to stabilize before introducing a new functional overload.
- Time-Restricted Eating (Early Window): Shift the feeding window to avoid caloric intake within 3 hours of sleep. This prevents nocturnal postprandial glucose excursions and subsequent sympathetic nervous system activation, theoretically stabilizing overnight HRV parameters.
Red Flag Zone (Safety Data Absent / Contradicted)
- GLP-1 Agonist Presumptions: Do not assume GLP-1 receptor agonists will universally improve autonomic metrics. Current literature indicates these compounds can exert sympathomimetic effects, raising resting heart rate and suppressing absolute HRV. Tracking HRV-CV while initiating a GLP-1 requires careful baseline recalibration.
- Inter-Individual HRV Comparisons: Discard the practice of comparing absolute HRV numbers against other individuals. Due to genetic variance, survival bias in ectopic beats among the elderly, and paradoxes in specific demographics, HRV is strictly an intra-individual biomarker.
V. Technical Mechanism Breakdown
Autonomic Resilience and Allostatic Load
HRV-CV serves as a mathematical proxy for allostatic load—the cumulative wear and tear on biological systems responding to chronic stress. A healthy autonomic nervous system operates with high vagal tone (parasympathetic dominance) at rest. When challenged, the system rapidly withdraws vagal influence (the “vagal brake”), allowing sympathetic dominance to raise heart rate and cardiac output. High HRV-CV indicates a failure of the system to efficiently re-engage the vagal brake after a stressor, leading to erratic day-to-day autonomic signaling and delayed recovery kinetics.
Circadian Misalignment and Glycemic Variability
The transcript correctly highlights late-night eating as a disruptor of HRV. Mechanistically, insulin secretion and peripheral tissue insulin sensitivity follow strict diurnal rhythms dictated by core clock genes (CLOCK and BMAL1) in the pancreas and liver. Caloric load introduced during the biological night—when insulin sensitivity is nadir—causes exaggerated postprandial glucose excursions. This metabolic inflexibility triggers a counter-regulatory stress response: the hypothalamic-pituitary-adrenal (HPA) axis elevates cortisol, and sympathetic output increases to manage the metabolic load. This systemic sympathetic activation overrides nocturnal parasympathetic tone, resulting in acute HRV suppression and increased HRV-CV instability.
Good paper. One question goes to accurate measurements. An expert on an Attia deep dive, for example, claims that iPhone HRV measurements are not valid either in absolute terms or even as a trend monitor.
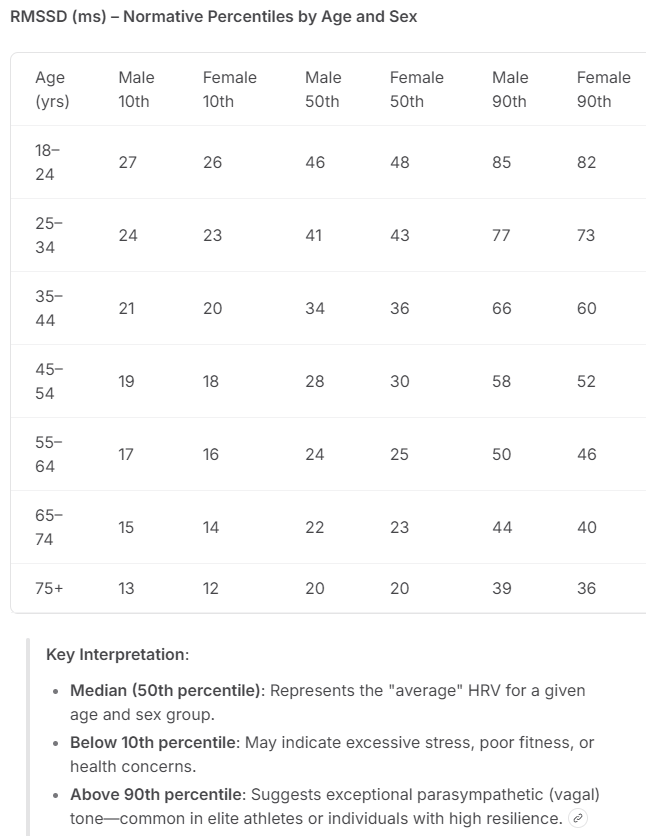
Separately, here is a 10th, 50th, and 90thj percentile table by age and sex, using the RMSSD model. Apple uses the SDNN method.
.
This only really works as a metric if you are not drinking alcohol not only during the measurement period, but also for a few days beforehand. It is also heavily influenced by the state of the autonomous nervous system and that can vary.
I get two HRV measurements. One from fitbit which is when I am asleep and the other from Polar H10 and elite (RMSSD) which I do when I have woken and am lying in bed.
I am not sure whether the sleeping HRV is that useful, but to be honest I think HRV is interesting, but not something to target that much.
What I can say with certainty is that my AM HRV is a good predictor of how my workouts will go for that day. The higher the HRV, the better the workout. I experience a few outliers but the correlation is high enough that you want to bet on the number.
I have not examined the IVs against HRV as a DV but I suspect sleep and sleep quality is well accounted for.in terms of variance.
Another consideration applies to those who take ARBs (and some other drugs). Telmisartan 80 mg/day, taken in the evening might increase AM HRV 10-20%. It is debatable as to whether this change reflects a beneficial change or is an artifact. I believe it is the former.